Abstract
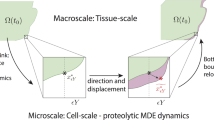
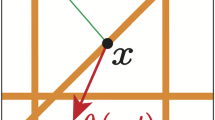
Here we introduce a model of solid tumour growth coupled with a multiscale biomechanical description of the tumour microenvironment, which facilitates the explicit simulation of fibre–fibre and tumour–fibre interactions. We hypothesise that such a model, which provides a purely mechanical description of tumour–host interactions, can be used to explain experimental observations of the effect of collagen micromechanics on solid tumour growth. The model was specified to mouse tumour data, and numerical simulations were performed. The multiscale model produced lower stresses than an equivalent continuum-like approach, due to a more realistic remodelling of the collagen microstructure. Furthermore, solid tumour growth was found to cause a passive mechanical realignment of fibres at the tumour boundary from a random to a circumferential orientation. This is in accordance with experimental observations, thus demonstrating that such a response can be explained as purely mechanical. Finally, peritumoural fibre network anisotropy was found to produce anisotropic tumour morphology. The dependency of tumour morphology on the peritumoural microstructure was reduced by adding a load-bearing non-collagenous component to the fibre network constitutive equation.











Similar content being viewed by others
Notes
FEB3: Finite Element Bioengineering in 3D, publicly available from https://bitbucket.org/vasvav/feb3-finite-element-bioengineering-in-3d.
References
Ambrosi D, Mollica F (2002) On the mechanics of a growing tumor. Int J Eng Sci 40:1297–1316
Ambrosi D, Mollica F (2004) The role of stress in the growth of a multicell spheroid. J Math Biol 48:477–499
Ambrosi D, Preziosi L (2009) Cell adhesion mechanisms and stress relaxation in the mechanics of tumours. Biomech Model Mechanobiol 8:397–413
Balay S, Gropp WD, McInnes LC, Smith BF (1997) Efficient management of parallelism in object oriented numerical software libraries. In: Bruaset AM, Langtangen HP, Arge E (eds) Modern software tools in scientific computing. Birkhäuser Press, Berlin, pp 163–202
Barocas V, Tranquillo R (1997) An anisotropic biphasic theory of tissue-equivalent mechanics: the interplay among cell traction, fibrillar network deformation, fibril alignment, and cell contact guidance. J Biomech Eng 119:137–145
Bathe KJ (1996) Finite element procedures. Prentice Hall, Upper Saddle River, NJ
Billiar KL, Sacks MS (2000a) Biaxial mechanical properties of the native and glutaraldehyde-treated aortic valve cusp: part I—experimental results. J Biomech Eng 122:23–30
Billiar KL, Sacks MS (2000b) Biaxial mechanical properties of the native and glutaraldehyde-treated aortic valve cusp: part II—a structural constitutive model. J Biomech Eng 122:327–335
Butcher DT, Alliston T, Weaver V (2009) A tense situation: forcing tumour progression. Nature 9:108–122
Byrne H (2010) Dissecting cancer through mathematics: from the cell to the animal model. Nat Rev Cancer 10:221–230
Casciari J, Sotirchos S, Sutherland R (1992) Variations in tumor cell growth rates and metabolism with oxygen concentration, glucose concentration, and extracellular pH. J Cell Physiol 151:386–394
Chandran PL, Barocas VH (2006) Affine versus non-affine fibril kinematics in collagen networks: theoretical studies of network behavior. J Biomech Eng 128:259–270
Chandran PL, Barocas VH (2007) Deterministic material-based averaging theory model of collagen gel micromechanics. J Biomech Eng 129:137–147
Chen CS, Mrksich M, Huang S, Whitesides GM, Ingber DE (1997) Geometric control of cell life and death. Science 276:1425–1428
Cheng G, Tse J, Jain RK, Munn LL (2009) Micro-environmental mechanical stress controls tumor spheroid size and morphology by suppressing proliferation and inducing apoptosis in cancer cells. PLoS One 4:e4632
Conklin MW, Keely PJ (2012) Why the stroma matters in breast cancer. Cell Adhes Migration 6:249–260
Cristini V, Lowengrub J (2010) Multiscale modeling of cancer: an integrated experimental and mathematical modeling approach, chaps 1 and 3, 1st edn. Cambridge University Press, Cambridge
Discher DE, Janmey P, Wang YL (2005) Tissue cells feel and respond to the stiffness of their substrate. Science 310:1139–1143
Frieboes H, Chaplain M, Thompson A, Bearer E, Lowengrub J, Cristini V (2011) Physical oncology: a bench-to-bedside quantitative and predictive approach. Cancer Res 71:298–302
Helmlinger G, Netti PA, Lichtenbeld HC, Melder RJ, Jain RK (1997) Solid stress inhibits the growth of multicellular tumor spheroids. Nat Biotechnol 15:778–783
Hori M, Nemat-Nasser S (1999) On two micromechanics theories for determining micro–macro relations in heterogeneous solids. Mech Mater 31:667–682
Hughes TJR (2000) The finite element method: linear static and dynamic finite element analysis, 1st edn. Mineola, NY, Dover Publications
Jackson TL, Byrne H (2002) A mechanical model of tumor encapsulation and transcapsular spread. Math Biosci 180:307–328
Jain RK, Martin JD, Stylianopoulos T (2014) The role of mechanical forces in tumor progression and therapy. Ann Rev Biomed Eng 16:321–346
Janmey PA (1998) The cytoskeleton and cell signaling: component localization and mechanical coupling. Physiol Rev 78:763–781
Kam Y, Rejniak K, Anderson A (2012) Cellular modeling of cancer invasion: integration of in silico and in vitro approaches. J Cell Physiol 227:431–438
Kim Y, Stolarska MA, Othmer HG (2011) The role of the microenvironment in tumor growth and invasion. Prog Biophys Mol Biol 106:353–379
Kirk BS, Peterson JW, Stogner RH, Carey GF (2006) libMesh: A C++ library for parallel adaptive mesh refinement/coarsening simulations. Eng Comput 22(3–4):237–254
Kroon M, Holzapfel GA (2008) A new constitutive model for multi-layered collagenous tissues. J Biomech 41:2766–2771
Liotta LA, Kohn EC (2001) The microenvironment of the tumour-host interface. Nature 411:375–379
Lubarda VA, Hoger A (2002) On the mechanics of solids with a growing mass. Int J Solids Struct 39:4627–4664
Malvern LE (1977) Introduction to the mechanics of a continuous medium, 1st edn. Prentice Hall, Upper Saddle River, NJ
Mpekris F, Angeli S, Pirentis A, Stylianopoulos T (2015) Stress-mediated progression of solid tumors: effect of mechanical stress on tissue oxygenation, cancer cell proliferation, and drug delivery. Biomech Model Mechanobiol. http://www.ncbi.nlm.nih.gov/pubmed/25968141
Netti PA, Berk DA, Swartz MA, Grodzinsky AJ, Jain RK (2000) Role of extracellular matrix assembly in interstitial transport in solid tumors. Cancer Res 60:2497–2503
Papageorgis P, Lambert A, Ozturk S, Gao F, Pan H, Manne U, Alekseyev Y, Thiagalingam A, Abdolmaleky H, Lenburg M, Thiagalingam S (2010) Smad signaling is required to maintain epigenetic silencing during breast cancer progression. Cancer Res 70:1916–1930
Paszek MJ, Zahir N, Johnson KR, Lakins JN, Rozenberg GI, Gefen A, Reinhart-King CA, Margulies SS, Dembo M, Boettiger D, Hammer DA, Weaver VM (2005) Tensional homeostasis and the malignant phenotype. Cancer Cell 8:241–254
Pelham RJ, Wang YL (1997) Cell locomotion and focal adhesions are regulated by substrate flexibility. PNAS 94:13661–13665
Preziosi L, Ambrosi D, Verdier C (2010) An elasto-visco-plastic model of cell aggregates. J Theor Biol 262:35–47
Provenzano PP, Eliceiri KW, Campbell JM, Inman DR, White JG, Keely PJ (2006) Collagen reorganization at the tumor-stromal interface facilitates local invasion. BMC Med 4:38
Rodriguez EK, Hoger A, McCulloch AD (1994) Stress-dependent finite growth in soft elastic tissues. J Biomech 27:455–467
Santner SJ, Dawson PJ, Tait L, Soule HD, Eliason J, Mohamed AN, Wolman SR, Heppner GH, Miller FR (2001) Malignant MCF10CA1 cell lines derived from premalignant human breast epithelial MCF10AT cells. Breast Cancer Res Treat 65:101–110
Silver FH, Horvath I, Foran DJ (2001) Viscoelasticity of the vessel wall: the role of collagen and elastin fibers. Crit Rev Biomed Eng 29:279–301
Stylianopoulos T, Martin JD, Chauhan VP, Jain SR, Diop-Frimpong B, Bardeesy N, Smith BL, Ferrone CR, Hornicek FJ, Boucher Y, Munn LL, Jain RK (2012) Causes, consequences, and remedies for growth-induced solid stress in murine and human tumors. PNAS 109:15101–15108
Stylianopoulos T, Martin JD, Snuderi M, Mpekris F, Jain SR, Jain RK (2013) Coevolution of solid stress and interstitial fluid pressure in tumors during progression: implications for vascular collapse. Cancer Res 73:3833–3841
Stylianopoulos T, Barocas VH (2007) Volume-averaging theory for the study of the mechanics of collagen networks. Comput Methods Appl Mech Eng 196:2981–2990
Veis A (1982) Collagen fibrillogenesis. Connect Tissue Res 10:11–24
Voutouri C, Mpekris F, Papageorgis P, Odysseos AD, Stylianopoulos T (2014) Role of constitutive behavior and tumor-host mechanical interactions in the state of stress and growth of solid tumors. PLoS One 9(e104):717
Yeung T, Georges PC, Flanagan LA, Marg B, Ortiz M, Funaki M, Zahir N, Ming W, Weaver V, Janmey PA (2005) Effects of substrate stiffness on cell morphology, cytoskeletal structure and adhesion. Cell Motil Cytoskelet 60:24–34
Acknowledgments
The authors from UCL would like to thank Dr. Rebecca Shipley (UCL IBME) for many useful discussions. The authors from UCL received funding from EPSRC grant “MIMIC”: EP/K020439/1, EU FP7 Virtual Physiological Human grant: “VPH-PRISM” (FP7-ICT-2011-9, 601040) and Marie-Curie Fellowship (FP7-PEOPLE-2013-IEF, 627025). The authors from UCY received funding from the European Research Council (FP7/2007-2013)/ERC Grant No. 336839-ReEngineeringCancer.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Wijeratne, P.A., Vavourakis, V., Hipwell, J.H. et al. Multiscale modelling of solid tumour growth: the effect of collagen micromechanics. Biomech Model Mechanobiol 15, 1079–1090 (2016). https://doi.org/10.1007/s10237-015-0745-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10237-015-0745-2