Abstract
Background. SmithKline Beecham synthesized camptothecin analogs and identified nogitecan hydrochloride (topotecan) with a broad spectrum of antitumor activity and less toxicity than camptothecin. Because preclinical and overseas clinical data indicated the antitumor effect of nogitecan hydrochloride with a 5-day repeat-dose schedule, we carried out phase I studies in Japan to determine the maximum tolerated dose (MTD), pharmacokinetics, and antitumor effect of nogitecan hydrochloride.
Methods. Phase I studies of nogitecan hydrochloride given by single and 5-day repeat dosing were carried out in patients with various solid tumors at 15 medical institutions in Japan. Pharmacokinetic evaluations were performed for both single and 5-day repeated dosing.
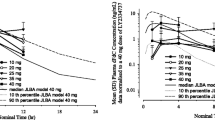
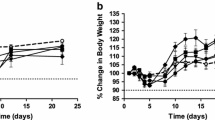
Results. The dose-limiting factor (DLF) was reversible leucopenia, and the maximum tolerated dose (MTD) was higher than 22.5 mg/m2 in the single-dose study. In the 5-day repeat-dose study, the DLF was also reversible leucopenia, and the MTD was estimated to be 1.5 mg/m2 per day. The plasma concentration of nogitecan hydrochloride increased with increasing dose, and the half-life after single dosing ranged from 3 to 5 h. There was no evidence of accumulation or delayed excretion during 5-day repeat dosing.
Conclusion. Based on these results and the finding that there were responders among patients treated at 1.5 mg/m2 per day by 5-day repeat dosing in overseas studies, 5-day repeat dosing of 1.2 mg/m2 per day, one dose level lower than the MTD, was selected for phase II studies in Japan.
Similar content being viewed by others
Author information
Authors and Affiliations
Additional information
Received: July 30, 2001 / Accepted: February 28, 2002
About this article
Cite this article
Kobayashi, K., Hino, M., Fukuoka, M. et al. Phase I studies of nogitecan hydrochloride for Japanese. Int J Clin Oncol 7, 177–186 (2002). https://doi.org/10.1007/s101470200026
Issue Date:
DOI: https://doi.org/10.1007/s101470200026