Abstract
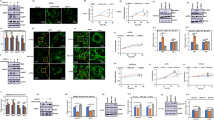
This study aimed to elucidate the roles of cilia- and flagella-associated protein 61 (CFAP61) in male infertility and its underlying mechanisms. CFAP61 expression levels in the testicular tissues of male patients with infertility were determined using quantitative real-time polymerase chain reaction, immunohistochemical assay, and western blotting. Moreover, the specific roles of CFAP61 in male infertility were evaluated using cell counting kit-8, 5-ethynyl-2'-deoxyuridine, flow cytometry, and enzyme-linked immunosorbent assays. Here, CFAP61 was expressed at low levels in the testicular tissues of male patients with infertility. Functionally, CFAP61 knockdown reduced the Leydig cell viability and testosterone secretion and enhanced apoptosis. A mechanistic study further revealed that silencing CFAP61 promoted the expression levels of mitogen-activated protein kinase (MAPK)/cyclooxygenase-2 (COX-2) signaling pathway-related proteins (p-extracellular signal-regulated kinase (p-ERK), p–c-Jun N-terminal kinase (p-JNK), p-P38, and COX-2). In conclusion, CFAP61 knockdown facilitated male infertility by suppressing Leydig cell viability and testosterone secretion and enhanced cell apoptosis by activating the MAPK/COX-2 pathway. Our data suggest CFAP61 as a potential therapeutic target for male infertility.




Similar content being viewed by others
Data availability
The datasets generated during and analyzed during the current study are available from the corresponding author on reasonable request.
References
Adegoke EO, Rahman MS, Pang MG (2020) Bisphenols threaten male reproductive health via testicular cells. Front Endocrinol (Lausanne) 11:624. https://doi.org/10.3389/fendo.2020.00624
Agarwal A, Baskaran S, Parekh N et al (2021) Male infertility. Lancet 397:319–333. https://doi.org/10.1016/s0140-6736(20)32667-2
Cacheiro-Llaguno C, Hernández-Subirá E, Díaz-Muñoz MD, Fresno M, Serrador JM, Íñiguez MA (2022) Regulation of cyclooxygenase-2 expression in human T cells by glucocorticoid receptor-mediated transrepression of nuclear factor of activated T cells. Int J Mol Sci 23. https://doi.org/10.3390/ijms232113275
Chen Y, Wang J, Xu D et al (2021) m(6)A mRNA methylation regulates testosterone synthesis through modulating autophagy in Leydig cells. Autophagy 17:457–475. https://doi.org/10.1080/15548627.2020.1720431
Chung JY, Park JE, Kim YJ, Lee SJ, Yu WJ, Kim JM (2022) Styrene cytotoxicity in testicular Leydig cells in vitro. Dev Reprod 26:99–105. https://doi.org/10.12717/dr.2022.26.3.99
Dudek P, Kozakowski J, Zgliczyński W (2017) Late-onset hypogonadism. Prz Menopauzalny 16:66–69. https://doi.org/10.5114/pm.2017.68595
Dymek EE, Smith EF (2007) A conserved CaM- and radial spoke associated complex mediates regulation of flagellar dynein activity. J Cell Biol 179:515–526. https://doi.org/10.1083/jcb.200703107
Fagerberg L, Hallström BM, Oksvold P et al (2014) Analysis of the human tissue-specific expression by genome-wide integration of transcriptomics and antibody-based proteomics. Mol Cell Proteomics 13:397–406. https://doi.org/10.1074/mcp.M113.035600
Frejborg E, Salo T, Salem A (2020) Role of Cyclooxygenase-2 in head and neck tumorigenesis. Int J Mol Sci 21. https://doi.org/10.3390/ijms21239246
Frungieri MB, Calandra RS, Mayerhofer A, Matzkin ME (2015) Cyclooxygenase and prostaglandins in somatic cell populations of the testis. Reproduction 149:R169-180. https://doi.org/10.1530/rep-14-0392
Fujisawa M (2006) Regulation of testicular function by cell-to-cell interaction. Reprod Med Biol 5:9–17. https://doi.org/10.1111/j.1447-0578.2006.00118.x
Grover SB, Antil N, Katyan A, Rajani H, Grover H, Mittal P, Prasad S (2020) Niche role of MRI in the evaluation of female infertility. Indian J Radiol Imaging 30:32–45. https://doi.org/10.4103/ijri.IJRI_377_19
Hashemi Goradel N, Najafi M, Salehi E, Farhood B, Mortezaee K (2019) Cyclooxygenase-2 in cancer: A review. J Cell Physiol 234:5683–5699. https://doi.org/10.1002/jcp.27411
Hu T, Meng L, Tan C et al (2023) Biallelic CFAP61 variants cause male infertility in humans and mice with severe oligoasthenoteratozoospermia. J Med Genet 60:144–153. https://doi.org/10.1136/jmedgenet-2021-108249
Huang T, Yin Y, Liu C et al (2020) Absence of murine CFAP61 causes male infertility due to multiple morphological abnormalities of the flagella. Sci Bull (Beijing) 65:854–864. https://doi.org/10.1016/j.scib.2020.01.023
Jing J, Ding N, Wang D et al (2020) Oxidized-LDL inhibits testosterone biosynthesis by affecting mitochondrial function and the p38 MAPK/COX-2 signaling pathway in Leydig cells. Cell Death Dis 11:626. https://doi.org/10.1038/s41419-020-02751-z
Li C, Zhang L, Ma T et al (2021) Bisphenol A attenuates testosterone production in Leydig cells via the inhibition of NR1D1 signaling. Chemosphere 263:128020. https://doi.org/10.1016/j.chemosphere.2020.128020
Liu S, Zhang J, Kherraf ZE et al. (2021) CFAP61 is required for sperm flagellum formation and male fertility in human and mouse. Development 148. https://doi.org/10.1242/dev.199805
Luo D, He Z, Yu C, Guan Q (2022) Role of p38 MAPK signalling in testis development and male fertility. Oxid Med Cell Longev 2022:6891897. https://doi.org/10.1155/2022/6891897
Ma A, Zeb A, Ali I et al (2021) Biallelic variants in CFAP61 cause multiple morphological abnormalities of the flagella and male infertility. Front Cell Dev Biol 9:803818. https://doi.org/10.3389/fcell.2021.803818
Monakova A, Sagaradze G, Basalova N, Popov V, Balabanyan V, Efimenko A (2022) Novel potency assay for MSC secretome-based treatment of idiopathic male infertility employed Leydig cells and revealed vascular endothelial growth factor as a promising potency marker. Int J Mol Sci 23. https://doi.org/10.3390/ijms23169414
Nordkap L, Jensen TK, Hansen Å M et al. (2016) Psychological stress and testicular function: a cross-sectional study of 1,215 Danish men. Fertil Steril 105:174–187.e171–172. https://doi.org/10.1016/j.fertnstert.2015.09.016
Oduwole OO, Huhtaniemi IT, Misrahi M (2021) The roles of luteinizing hormone, follicle-stimulating hormone and testosterone in spermatogenesis and folliculogenesis revisited. Int J Mol Sci 22. https://doi.org/10.3390/ijms222312735
Omolaoye TS, Omolaoye VA, Kandasamy RK, Hachim MY, Du Plessis SS (2022) Omics and male infertility: Highlighting the application of transcriptomic data. Life (Basel) 12. https://doi.org/10.3390/life12020280
Parmar BK, Verma UR, Vaishnav JA, Balakrishnan S (2022) Cyclooxygenase-2 plays a crucial role during myocardial patterning of developing chick. Int J Dev Biol 66:373–381. https://doi.org/10.1387/ijdb.220153sb
Sağraç D, Şenkal S, Hayal TB, Demirci S, Şişli HB, Asutay AB, Doğan A (2022) Protective role of cytoglobin and neuroglobin against the Lipopolysaccharide (LPS)-induced inflammation in Leydig cells ex vivo. Reprod Biol 22:100595. https://doi.org/10.1016/j.repbio.2021.100595
Sharma A, Minhas S, Dhillo WS, Jayasena CN (2021) Male infertility due to testicular disorders. J Clin Endocrinol Metab 106:e442–e459. https://doi.org/10.1210/clinem/dgaa781
Sun J, Lian X, Lv C et al (2023) Trps1 acts as a regulator of Sf-1 transcription and testosterone synthesis in mouse Leydig cells. Cell Biol Toxicol 8:2. https://doi.org/10.1007/s10565-023-09823-8
The Genotype-Tissue Expression (GTEx) project. (2013) Nat Genet 45:580–585. https://doi.org/10.1038/ng.2653
Tian WJ, Jeon SH, Cho HJ, Rajasekaran MR et al (2022) Effect of Li-ESWT on Testicular Tissue and Function in Androgen-Deficient Rat Model. Oxid Med Cell Longev 2022:5213573. https://doi.org/10.1155/2022/5213573
Urbanska P, Song K, Joachimiak E et al (2015) The CSC proteins FAP61 and FAP251 build the basal substructures of radial spoke 3 in cilia. Mol Biol Cell 26:1463–1475. https://doi.org/10.1091/mbc.E14-11-1545
Wang X, Shen CL, Dyson MT, Eimerl S, Orly J, Hutson JC, Stocco DM (2005) Cyclooxygenase-2 regulation of the age-related decline in testosterone biosynthesis. Endocrinology 146:4202–4208. https://doi.org/10.1210/en.2005-0298
Wang Z, Xie Y, Chen H et al (2021) Guilingji protects against spermatogenesis dysfunction from oxidative stress via regulation of MAPK and apoptotic signaling pathways in Immp2l mutant mice. Front Pharmacol 12:771161. https://doi.org/10.3389/fphar.2021.771161
Wu X, Zhou L, Shi J, Cheng CY, Sun F (2022) Multiomics analysis of male infertility†. Biol Reprod 107:118–134. https://doi.org/10.1093/biolre/ioac109
Xiong X, Wu Q, Zhang L et al (2022) Chronic stress inhibits testosterone synthesis in Leydig cells through mitochondrial damage via Atp5a1. J Cell Mol Med 26:354–363. https://doi.org/10.1111/jcmm.17085
Yang C, Li P, Li Z (2021a) Clinical application of aromatase inhibitors to treat male infertility. Hum Reprod Update 28:30–50. https://doi.org/10.1093/humupd/dmab036
Yang M, Guan S, Tao J et al (2021b) Melatonin promotes male reproductive performance and increases testosterone synthesis in mammalian Leydig cells†. Biol Reprod 104:1322–1336. https://doi.org/10.1093/biolre/ioab046
Zhao Y, Liu X, Qu Y et al (2021) The roles of p38 MAPK → COX2 and NF-κB → COX2 signal pathways in age-related testosterone reduction. Sci Rep 9:10556. https://doi.org/10.1038/s41598-019-46794-5
Zhou SH, Deng YF, Weng ZW, Weng HW, Liu ZD (2019) Traditional Chinese medicine as a remedy for male infertility: A review. World J Mens Health 37:175–185. https://doi.org/10.5534/wjmh.180069
Funding
This study received no funding.
Author information
Authors and Affiliations
Contributions
XC conceived and designed the study. WZ performed the experiment and drafted the manuscript. MJ performed the literature searching and data analysis. JQ contributed to the data analysis. XC and WZ discussed the results. XC and WZ revised the manuscript. All authors reviewed the manuscript.
Corresponding author
Ethics declarations
Ethics approval and consent participate
This study was reviewed and approved by the Ethic Committee of the Medical School of Nantong University (2020173). Informed consent was obtained from the patients involved.
Animal ethics
Not applicable.
Competing interests
The authors declare no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Zhu, W., Mao, J., Qin, J. et al. CFAP61 knockdown aggravates male infertility by inhibiting testosterone secretion by Leydig cells via the MAPK/COX-2 pathway. Funct Integr Genomics 23, 340 (2023). https://doi.org/10.1007/s10142-023-01271-1
Received:
Revised:
Accepted:
Published:
DOI: https://doi.org/10.1007/s10142-023-01271-1