Abstract
Background
Previous studies have focused on the non-inferiority of RPG compared with conventional port gastrectomy (CPG); however, we assumed that some candidates might derive more significant benefit from RPG over CPG.
Methods
We retrospectively analyzed the clinicopathological and perioperative parameters of 1442 patients with gastric cancer treated by gastrectomy between 2009 and 2022. The C-reactive protein level on postoperative day 3 (CRPD3) was used as a surrogate parameter for surgical trauma. Patients were grouped according to the extent of gastrectomy [subtotal gastrectomy (STG) or total gastrectomy (TG)] and lymph node dissection (D1+ or D2). The degree of surgical trauma, bowel recovery, and hospital stay between RPG and CPG was compared among those patient groups.
Results
Of 1442 patients, 889, 354, 129, and 70 were grouped as STGD1+, STGD2, TGD1+, and TGD2, respectively. Compared with CPG, RPG significantly decreased CRPD3 only among patients in the STGD1+ group (CPG: n = 653, 84.49 mg/L, 95% CI 80.53–88.45 vs. RPG: n = 236, 70.01 mg/L, 95% CI 63.92–76.09, P < 0.001). In addition, the RPG method significantly shortens bowel recovery and hospital stay in the STGD1+ (P < 0.001 and P < 0.001), STGD2 (P < 0.001 and P < 0.001), and TGD1+ (P = 0.026 and P = 0.007), respectively. No difference was observed in the TGD2 group (P = 0.313 and P = 0.740).
Conclusions
The best candidates for RPG are patients who undergo STGD1+, followed by STGD2 and TG D1+, considering the reduction in CRPD3, bowel recovery, and hospital stay.
Similar content being viewed by others
Introduction
Minimally invasive surgery (MIS) has radically transformed surgical outcomes by reducing surgical trauma, improving patient recovery, and shortening the lengths of hospital stays [1,2,3,4,5,6,7]. To minimize invasiveness, reduced port laparoscopic surgery (RPS) techniques are applied more frequently [8]. Recent technical developments were initially developed for the surgical treatment of benign diseases; however, they have allowed RPS techniques to be applied to treat malignant diseases, such as colorectal and gastric cancers [9,10,11,12]. The use of reduced port gastrectomy (RPG) is increasing [11, 13,14,15], and prior studies have reported the non-inferiority of RPG compared with conventional port gastrectomy (CPG) regarding lymph node dissection (LND), bleeding, and pain after surgery [14]. However, the widespread adoption of RPG remains limited by the lack of specialized instruments, a constrained operating view, restricted instrument movement, and longer operating times [14]. Furthermore, RPG can be applied to various indications; however, the patient subgroups that would benefit most from RPG remain unclear. The goals of RPG are minimized surgical stress and expedited recovery. Therefore, quantitative parameters must be identified to measure the effects of RPG and assess surgical trauma to determine which patients would benefit most from RPG over CPG.
Notably, several established clinical laboratory markers evaluate systemic inflammation in daily clinical practice, including C-reactive protein (CRP) and the cytokines interleukin 1, interleukin 6, and tumor necrosis factor-alpha [16, 17]. CRP is the most specific and sensitive marker for assessing the extent of surgery-induced tissue injury and is well suited for evaluating whether RPG can reduce surgical stress [18]. The ability to predict surgical stress using a quantitative parameter, such as CRP, would enable surgeons to select either CPG or RPG depending on which gastrectomy approach would be most beneficial for each patient.
Methods
Patients
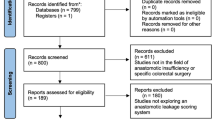
A total of 2469 patients with gastric cancer were treated with gastrectomy by a single surgeon (Hyoung-Il Kim) at Yonsei Cancer Center between 2009 and 2022. We collected retrospective data on clinicopathological and perioperative parameters to examine their impacts on surgical trauma. In this study, we assumed that the level of CRP on postoperative day 3 (CRPD3) is a surrogate parameter for the total surgical stress associated with the procedures [19,20,21,22]. To focus our analysis on ideal MIS conditions, we excluded patients with a history of (1) open gastrectomy or an MIS gastrectomy that was converted to an open gastrectomy (n = 516); (2) proximal gastrectomy, completion total gastrectomy (TG), or miscellaneous gastrectomy (n = 125); (3) neoadjuvant chemotherapy (n = 67); or (4) intraoperative bleeding over 1000 cc, over 15 days of hospital stay, Clavien–Dindo classification grade 3 or higher, or missing CRPD3 values (n = 319). The remaining 1442 patients were enrolled in the final statistical analysis (Fig. 1). Informed consent was waived because of the study’s retrospective nature, and the analysis used anonymous clinical data. This study was approved by the Institutional Review Board of Yonsei University College of Medicine, Korea (IRB No. 4-2022-1543).
Port insertion protocol for CPG and RPG
CPG was performed through a 10 mm port located in the umbilical area for the laparoscope, a 12 mm port located in the right lower quadrant area, and the left paramedian area. A 5 mm port is located in both the right upper quadrant area and the left upper quadrant area. RPG was performed using an umbilical incision with a 12 mm port at the RUQ as an assist port. The umbilical incision size for both the CPG and RPG approaches remained consistently 25–30 mm. In CPG, a 10 mm port was initially employed for laparoscopy and extended to 25–30 mm at the conclusion of the procedure for specimen extraction. Conversely, for RPG, a 25–30 mm incision was initially created from the beginning to accommodate the multiport configuration.
Special instruments, SurgiTractor (St0306kb, SurgiCore Co., Ltd, Korea), ArtiSential fenestrated forceps, and a medium–large clip applier (AUF01-L and ACA01-L, LivsMed, Seongnam, Korea) were used for RPG in laparoscopic gastrectomy [23]. For robotic RPG, Single-Site system and associated instruments [Cadiere forceps and wristed needle drive (478055 and 478115, Intuitive Surgical, Sunnyvale, CA, USA)] were used through Single-Site port (478065, intuitive Surgical, Sunnyvale, CA, USA) as in a previously published study [24].
CRPD3 analyses by subgroup and associations with perioperative parameters
The patients were categorized according to gastrectomy type [subtotal gastrectomy (STG) or TG] and the extent of LND (D1+ or D2), resulting in four patient groups: STGD1+ (n = 889), STGD2 (n = 354), TGD1+ (n = 129), and TGD2 (n = 70). Patients were further subdivided based on whether they received RPG (n = 305, 21.15%) or CPG (n = 1137, 78.85%). Figure 2 illustrates surgical trauma following RPG or CPG according to the extent of LND and gastrectomy. We analyzed CRPD3 values for all groups and estimated the contributions of several perioperative parameters to CRPD3 values using linear regression analysis.
Statistics
Descriptive statistics are presented as the mean and standard deviation for continuous variables and the number and percent for categorical variables. Student’s t test and the Chi-square test or Fisher exact test were performed to evaluate differences in continuous and categorical variables. Linear regression was applied to assess associations between postoperative serum CRP levels and potential risk factors. Variables with P < 0.05 in the univariate analysis were examined in a multivariate linear regression analysis to estimate the contributions of perioperative parameters to CRPD3. All statistical analyses were conducted using SAS software version 9.4 (SAS Institute, Cary, NC) and R package (v. 4.0.4, http://www.r-project.org/). The graphs were produced using GraphPad Prism software version 10.0.2 for Mac (GraphPad Software, San Diego, CA, USA). Reported P-values are two-sided, and statistical significance was set at P < 0.05.
Results
Patients
The clinical characteristics of patients in the CPG and RPG subgroups are compared in Table 1. No significant differences were observed between these two groups for clinical features, including sex (P = 0.059), body mass index (BMI; CPG: 23.88 ± 3.32 kg/m2 vs. RPG: 23.66 ± 3.05 kg/m2, P = 0.315), American Society of Anesthesiologists Physical Status (ASA-PS; P = 0.346), operating time (minutes) (CPG: 166.31 ± 57.70 vs. RPG: 161.80 ± 39.93, P = 0.115), diabetes mellitus (DM, P = 0.302), non-tuberculosis (TB) pulmonary disease (P > 0.999), nephrology disease (P = 0.491), liver disease (P = 0.816), cerebral disease (P = 0.457), or TB (P = 0.770). However, significant differences between groups were observed for hypertension (HTN, P = 0.020) and cardiology disease (P = 0.046). According to pathologic reports, patients who underwent CPG had more advanced forms of gastric cancer than those who underwent RPG, as assessed by T-stage (P = 0.003), N-stage (P = 0.042), and total stage (P = 0.002). As a result, the CPG group had more extensive LND (P < 0.001), gastrectomy (P < 0.001), and omentectomy (P = 0.002) than the RPG group.
CRPD3, bowel recovery, and hospital stay in each subgroup
The RPG method significantly decreased CRPD3 values compared to the CPG method. (CPG: n = 1137, 90.13 mg/L, 95% CI 86.87–93.30 vs. RPG: n = 305, 75.49 mg/L, 95% CI 63.95–81.69, P < 0.001) (Fig. 3a). In subgroup analysis, the RPG method significantly decreased CRPD3 values in the STGD1+ group (CPG: n = 635, 84.49 mg/L, 95% CI 80.53–88.45 vs. RPG: n = 236, 70.01 mg/L, 95% CI 63.92–76.09, P < 0.001, Fig. 3b). No significant advantages of using RPG over CPG were observed for the STGD2 (CPG: n = 305, 92.51 mg/L, 95% CI 86.48–98.55 vs. RPG: n = 49, 91.03 mg/L, 95% CI 73.06–109.01, P = 0.861), TGD1+ (CPG: n = 118, 101.11 mg/L, 95% CI 90.66–111.56 vs. RPG: n = 11, 101.50 mg/L, 95% CI 60.72–142.28, P = 0.983), or TGD2 (CPG: n = 61, 117.35 mg/L, 95% CI 102.02–132.70 vs. RPG: n = 9, 102.79 mg/L, 95% CI 60.33–145.25 P = 0.495) groups.
Subgroup analysis identifying the advantage of reduced port gastrectomy. a CRPD3 reduction. b Subgroup analysis of CRPD reduction. c Bowel recovery. d The hospital stay. CPG conventional port gastrectomy, RPG reduced port gastrectomy, cSTGD1+ conventional port subtotal gastrectomy with D1+ dissection, rSTGD1+ reduced port subtotal gastrectomy with D1+ dissection, cSTGD2 conventional port subtotal gastrectomy with D2 dissection, rSTGD2 reduced port subtotal gastrectomy with D2 dissection, cTGD1+ conventional port total gastrectomy with D1+ dissection, rTGD1+ reduced port total gastrectomy with D1+ dissection, cTGD2 conventional port total gastrectomy with D2 dissection, rTGD2 reduced port total gastrectomy with D2 dissection
The RPG method significantly shortens bowel recovery in the STGD1+ (CPG: n = 651, 2.86 days, 95% CI 2.81–2.92vs. RPG: n = 236, 2.66 days, 95%CI, 2.57–2.74, P < 0.001, Fig. 3c), STGD2(CPG: n = 305, 3.03 days, 95% CI 2.95–3.12 vs. RPG: n = 48, 2.44 days, 95% CI 2.22–2.65, P < 0.001), and TGD1+ (CPG: n = 118, 3.05 days, 95% CI 2.92–3.18 vs. RPG: n = 11, 2.55 days, 95% CI 2.08–3.01, P = 0.026) groups. There was no significant benefit of RPG compared with CPG for the TGD2 group (CPG: n = 61, 2.92 days, 95% CI 2.74–3.10 vs. RPG: n = 9, 2.67 days, 95% CI 2.12–3.21, P = 0.313).
Similar to the above results, the RPG method significantly shortens the hospital stay in the STGD1+ (CPG: n = 653, 5.53 days, 95% CI 5.41–5.66 vs. RPG: n = 236, 4.39 days, 95% CI 4.21–4.56, P < 0.001, Fig. 3d), STGD2 (CPG: n = 305, 5.67 days, 95% CI 5.50–5.84 vs. RPG: n = 49, 4.80 days, 95% CI 4.33–5.26, P < 0.001), and TGD1+ (CPG: n = 118, 6.47 days, 95% CI 6.11–6.82 vs. RPG: n = 11, 4.82 days, 95% CI 3.88–5.76, P = 0.007) groups. No significant advantages of using RPG over CPG were observed for the TGD2 group (CPG: n = 56, 5.52 days, 95% CI 5.11–5.92 vs. RPG: n = 9, 5.33 days, 95% CI 4.00–6.67, P = 0.740).
Quantitative estimation of the impacts of perioperative parameters on CRPD3
On univariate analysis (Table 2), the following factors were significantly associated with increasing CRPD3: male sex, age ≥ 65 years, BMI ≥ 25 kg/m2, ASA-PS ≥ 2, CPG, D2 dissection, T-stage > 1, TG, total omentectomy, node metastasis, HTN, and DM. Multivariate analysis was performed to estimate each parameter’s contributions to changes in CRPD3. Significant factors associated with increasing CRPD3 were male sex (P < 0.001, reference value female sex), BMI ≥ 25 kg/m2 (P < 0.001, reference value BMI < 25 kg/m2), CPG (P = 0.0109, reference value RPG), D2 dissection (P = 0.0174, reference value D1+ dissection), TG (P < 0.001, reference value STG), total omentectomy (P = 0.042, reference value partial omentectomy), HTN (P < 0.001), and DM (P = 0.0138). Finally, linear regression was performed to estimate CRPD3 levels after minimally invasive gastrectomy to treat gastric cancer (Table 3). Every variable included in the linear regression model was significant: male sex (32.56 ± 2.79 mg/L, P < 0.001), BMI ≥ 25 kg/m2 (10.21 ± 2.94 mg/L, P < 0.001), RPG (− 8.97 ± 3.39 mg/L, P = 0.008), D2 dissection (9.26 ± 3.10 mg/L, P = 0.003), TG (14.79 ± 4.97 mg/L, P < 0.001), total omentectomy (9.53 ± 4.67 mg/L, P = 0.041), HTN (11.84 ± 2.98 mg/L, P < 0.001), and DM (9.66 ± 3.85 mg/L, P = 0.012). The pie chart shows the proportional contribution of each parameter on the CRPD3. (Electronic Supplementary Material, Fig. 1).
Discussion
Since the initial application of RPS techniques for gastrectomy [11], many advantages of the method have been reported [25,26,27,28,29]. However, a detailed understanding of the patient groups most suited to receive RPG is yet to be determined. This study identified CRPD3 as an easily measured clinical laboratory parameter that could estimate surgical trauma and predict postoperative recovery. Serum CRP is an acute-phase reactant that responds to inflammation and tissue damage. The postoperative serum CRP concentration is a quantitative index that integrates the cumulative effects of preoperative comorbidities, surgical invasion, surgical duration, anesthetic management, and analgesia [30, 31]. CRPD3 has been used in many previous studies as a quantitative indicator of surgical stress that predicts the postoperative recovery course [19,20,21,22], and CRPD3 values correlate with the length of hospital stay, short-term recovery, and postoperative complications [32]. This study’s results corroborate with previous reports. The levels of CRP were highest on postoperative day 3. Therefore, we selected CRPD3 as a surrogate parameter to reflect the degree of surgical damage (Electronic Supplementary Material, Fig. 2a). Pearson’s correlation coefficients analysis of CRPD3 values correlated with the time of first gas passing and the length of the hospital stay (Electronic Supplementary Material, Fig. 2b and 2c).
We utilized CRPD3 as a surrogate measure that allowed us to quantitatively estimate the degree to which the RPG method could reduce total surgical trauma, representing a novel application of CRPD3. Using linear regression analysis, we determined that RPG instead of CPG could reduce CRPD3 by approximately 6% among patients undergoing gastrectomy (Electronic Supplementary Material, Fig. 1). However, the potential benefits of RPG diminish with more extensive gastrectomy and LND. Therefore, our study suggests that patients who require STGD1+ procedures represent the best candidates for RPG, particularly women without comorbidities undergoing SGTD1+ procedures with partial omentectomy.
Acute pain is a frequent postoperative occurrence that delays the healing process, increases complications and mortality, and increases nursing costs and the duration of hospitalization [33]. Laparoscopic or robotic instruments transmit a surgeon’s external movements within the intraperitoneal cavity. The trocar acts as a fulcrum that bears the force of movement against the peritoneal wall. The somatic pain related to trocar insertion is sharper and more acute than the visceral pain associated with resection [34]. We hypothesized that the RPG method helps reduce somatic pain by piercing two to three fewer ports than CPG. The effect of RPG on CRPD3 was significant only in the STGD1+ group (Fig. 3b); however, the impact of RPG on bowel recovery and hospital stay was significant in STGD1+, STGD2, and TGD1+ (Fig. 3c, d). Furthermore, several previous studies reported that RPG showed less operative pain than CPG [35, 36], which is presumed to have led to more ambulation in those patients. As a result, the effect of shortening the recovery period was extended to the STGD2 and TGD1+ groups, in which the reduction of CRPD3 was not significant.
In the TGD2 group, RPG showed no improvement in CRPD3, bowel recovery, and hospital stay. Visceral and somatic pain associated with surgical trauma increased with more extensive gastrectomy (partial vs. total) and LND (D1+ vs. D2) procedures [37] (Figs. 2, 3). We hypothesize that the difference between CPG and RPG is clinically negligible if total surgical trauma is extensive.
This study has some limitations. First, CRPD3 is a clinically meaningful but indirect parameter for assessing surgical trauma. Second, to develop an ideal model for determining the optimal patient group for RPG, we excluded patients with complex presentations (Clavien-Dindo ≥ 3, hospital stay ≥ 14 d). Third, we only analyzed surgical trauma without considering cosmetic effects, which are crucial for younger patients.
In conclusion, we dissected and quantitatively assessed the perioperative parameters contributing to CRPD3 and found that CRPD3 varies with the extent of gastrectomy, the range of LND, sex, comorbidities, and obesity. RPG significantly reduces CRPD3 and expedites bowel recovery and hospital stay. Patients with STGD1+ benefit the most, followed by those with STGD2 and TGD1+.
References
Chow A, Aziz O, Purkayastha S, et al. Single incision laparoscopic surgery for acute appendicitis: feasibility in pediatric patients. Diagn Therap Endosc. 2010;2010:1–3.
Hu Y, Huang C, Sun Y, et al. Morbidity and mortality of laparoscopic versus open D2 distal gastrectomy for advanced gastric cancer: a randomized controlled trial. J Clin Oncol. 2016;34(12):1350–7.
Katai H, Mizusawa J, Katayama H, et al. Survival outcomes after laparoscopy-assisted distal gastrectomy versus open distal gastrectomy with nodal dissection for clinical stage IA or IB gastric cancer (JCOG0912): a multicentre, non-inferiority, phase 3 randomised controlled trial. Lancet Gastroenterol Hepatol. 2020;5(2):142–51.
Kim H-H, Han S-U, Kim M-C, et al. Effect of laparoscopic distal gastrectomy vs open distal gastrectomy on long-term survival among patients with stage I gastric cancer. JAMA Oncol. 2019;5(4):506.
McGregor CGC, Sodergren MH, Aslanyan A, et al. Evaluating systemic stress response in single port vs. multi-port laparoscopic cholecystectomy. J Gastrointest Surg. 2011;15(4):614–22.
Ojima T, Nakamura M, Hayata K, et al. Short-term outcomes of robotic gastrectomy vs laparoscopic gastrectomy for patients with gastric cancer. JAMA Surg. 2021;156(10):954.
Vittimberga FJ Jr, Foley DP, Meyers WC, et al. Laparoscopic surgery and the systemic immune response. Ann Surg. 1998;227(3):326–34.
Cuschieri A. Single-incision laparoscopic surgery. J Minim Access Surg. 2011;7(1):3–5.
Lee JH, Ryu KW, Kim YW, et al. Staging laparoscopy in gastric cancer: a single port method. J Surg Oncol. 2003;84(1):50–2.
Remzi FH, Kirat HT, Kaouk JH, et al. Single-port laparoscopy in colorectal surgery. Colorect Dis. 2008;10(8):823–6.
Omori T, Oyama T, Akamatsu H, et al. Transumbilical single-incision laparoscopic distal gastrectomy for early gastric cancer. Surg Endosc. 2011;25(7):2400–4.
Ozdemir BA, Thomas RL, Soon Y. Single-port laparoscopic subtotal gastrectomy with DIalpha lymphadenectomy. Surg Innov. 2011;18(4):NP1-4.
Iida A, Goi T, Fujimoto D, et al. Initial results of reduced port laparoscopic gastrectomy for gastric cancer. Ann Laparosc Endosc Surg. 2017;2:16–16.
Inaki N, Tsuji T, Doden K, et al. Reduced port laparoscopic gastrectomy for gastric cancer. Transl Gastroenterol Hepatol. 2016;1:38–38.
Omori T, Fujiwara Y, Moon J, et al. Comparison of single-incision and conventional multi-port laparoscopic distal gastrectomy with D2 lymph node dissection for gastric cancer: a propensity score-matched analysis. Ann Surg Oncol. 2016;23(S5):817–24.
Baumann H, Gauldie J. Regulation of hepatic acute phase plasma protein genes by hepatocyte stimulating factors and other mediators of inflammation. Mol Biol Med. 1990;7(2):147–59.
Baigrie RJ, Lamont PM, Kwiatkowski D, et al. Systemic cytokine response after major surgery. Br J Surg. 1992;79(8):757–60.
Brewster N, Guthrie C, McBirnie J. CRP levels as a measure of surgical trauma: a comparison of different general surgical procedures. J R Coll Surg Edinb. 1994;39(2):86–8.
Dutta S, Fullarton GM, Forshaw MJ, et al. Persistent elevation of C-reactive protein following esophagogastric cancer resection as a predictor of postoperative surgical site infectious complications. World J Surg. 2011;35(5):1017–25.
Hübner M, Mantziari S, Demartines N, et al. Postoperative albumin drop is a marker for surgical stress and a predictor for clinical outcome: a pilot study. Gastroenterol Res Pract. 2016;2016:1–8.
Kim EY, Yim HW, Park CH, et al. C-reactive protein can be an early predictor of postoperative complications after gastrectomy for gastric cancer. Surg Endosc. 2017;31(1):445–54.
Lee S-H, Kim KH, Choi CW, et al. Reduction rate of C-reactive protein as an early predictor of postoperative complications and a reliable discharge indicator after gastrectomy for gastric cancer. Ann Surg Treat Res. 2019;97(2):65.
Lee CM, Park S, Park SH, et al. Short-term outcomes and cost-effectiveness of laparoscopic gastrectomy with articulating instruments for gastric cancer compared with the robotic approach. Sci Rep. 2023;13(1):9355.
Roh CK, Choi S, Seo WJ, et al. Comparison of surgical outcomes between integrated robotic and conventional laparoscopic surgery for distal gastrectomy: a propensity score matching analysis. Sci Rep. 2020;10(1):485.
Lee JH, Lee MS, Kim HH, et al. Comparison of single-incision laparoscopic distal gastrectomy and laparoscopic distal gastrectomy for gastric cancer in a porcine model. J Laparoendosc Adv Surg Tech A. 2011;21(10):935–40.
Kawamura H, Tanioka T, Kuji M, et al. The initial experience of dual port laparoscopy-assisted total gastrectomy as a reduced port surgery for total gastrectomy. Gastr Cancer. 2013;16(4):602–8.
Ahn SH, Son SY, Jung DH, et al. Pure single-port laparoscopic distal gastrectomy for early gastric cancer: comparative study with multi-port laparoscopic distal gastrectomy. J Am Coll Surg. 2014;219(5):933–43.
Omori T, Masuzawa T, Akamatsu H, et al. A simple and safe method for Billroth I reconstruction in single-incision laparoscopic gastrectomy using a novel intracorporeal triangular anastomotic technique. J Gastrointest Surg. 2014;18(3):613–6.
Usui S, Tashiro M, Haruki S, et al. Triple-incision laparoscopic distal gastrectomy for the resection of gastric cancer: comparison with conventional laparoscopy-assisted distal gastrectomy. Asian J Endosc Surg. 2014;7(3):197–205.
Dobson GP. Addressing the global burden of trauma in major surgery. Front Surg. 2015;2:43.
Watt DG, Horgan PG, McMillan DC. Routine clinical markers of the magnitude of the systemic inflammatory response after elective operation: a systematic review. Surgery. 2015;157(2):362–80.
Winsen M, McSorley ST, McLeod R, et al. Postoperative C-reactive protein concentrations to predict infective complications following gastrectomy for cancer. J Surg Oncol. 2021;124(7):1060–9.
Schug SA. 2011—the global year against acute pain. Anaesth Intensive Care. 2011;39(1):11–4.
Chiung Ta LuT, Gan P, Versace V. Fewer ports cut opioid use and length of stay in elective laparoscopic cholecystectomy. JSLS. 2021;25(2):e2020.00093. https://doi.org/10.4293/JSLS.2020.00093.
Kawamura H, Tanioka T, Shibuya K, et al. Comparison of the invasiveness between reduced-port laparoscopy-assisted distal gastrectomy and conventional laparoscopy-assisted distal gastrectomy. Int Surg. 2013;98(3):247–53.
Kim SM, Ha MH, Seo JE, et al. Comparison of reduced port totally laparoscopic distal gastrectomy (Duet TLDG) and conventional laparoscopic-assisted distal gastrectomy. Ann Surg Oncol. 2015;22(8):2567–72.
Macías AA, Finneran JJ. Regional anesthesia techniques for pain management for laparoscopic surgery: a review of the current literature. Curr Pain Headache Rep. 2022;26(1):33–42.
Acknowledgements
This research was supported by the Korea Health Technology R&D Project grant through the Korea Health Industry Development Institute (KHIDI), funded by the Ministry of Health & Welfare, Republic of Korea (Grant number: HI22C0767). The funding source played no role in the design or conduct of the study; the collection, analysis, or interpretation of data; the preparation, review, or approval of the manuscript; or the decision to submit the manuscript for publication. We would like to thank Editage (http://www.editage.co.kr) for editing and reviewing this manuscript for the English language. In the end, thanks to Mun Kee Choi, the illustrator, for visualization of surgical trauma by gastrectomy according to the extent of lymph node dissection and gastrectomy (Fig. 2).
Funding
This research was supported by the Korea Health Technology R&D Project grant through the Korea Health Industry Development Institute (KHIDI), funded by the Ministry of Health & Welfare, Republic of Korea (Grant number: HI22C0767). The funding source played no role in the design or conduct of the study; the collection, analysis, or interpretation of data; the preparation, review, or approval of the manuscript; or the decision to submit the manuscript for publication.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
All authors report no conflicts of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Chung, J.H., Hwang, J., Park, S.H. et al. Identifying the best candidates for reduced port gastrectomy. Gastric Cancer 27, 176–186 (2024). https://doi.org/10.1007/s10120-023-01438-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10120-023-01438-6