Abstract
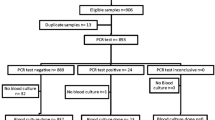
We retrospectively examined the yield of a cerebrospinal fluid (CSF) multiplex real-time PCR assay of febrile young infants undergoing a full sepsis work-up. Eighty infants were included in the study: Forty-nine (61%) neonates and 31 (39%) 29–90 day-old patients were included in the study. A viral pathogen was detected in 59% (47/80) of the samples, human enterovirus in 53% (42/80) and Human parechovirus in 6% (5/80). The CSF of nearly half of the subjects with CNS infection was without pleocytosis; all CSF cultures were negative. Multiplex PCR CSF testing enhances the diagnosis of pathogen-specific viral CNS infection among febrile young infants.

Similar content being viewed by others
References
Baraff LJ et al (Jul. 1993) Practice guideline for the management of infants and children 0 to 36 months of age with fever without source. Agency for Health Care Policy and Research. Ann Emerg Med 22(7):1198–1210. https://doi.org/10.1016/s0196-0644(05)80991-6
Martinez E et al (May 2015) Prevalence and predictors of bacterial meningitis in young infants with fever without a source. Pediatr Infect Dis J 34(5):494–498. https://doi.org/10.1097/INF.0000000000000629
M. D. Baker and J. R. Avner, “Management of Fever in Young Infants: Evidence Versus Common Practice,” Pediatrics, vol. 138, no. 6, 2016, doi: https://doi.org/10.1542/peds.2016-2085
Kadish HA, Loveridge B, Tobey J, Bolte RG, Corneli HM (Feb. 2000) Applying outpatient protocols in febrile infants 1-28 days of age: can the threshold be lowered? Clin Pediatr (Phila) 39(2):81–88. https://doi.org/10.1177/000992280003900202
A. T. Cruz et al., “Accuracy of Complete Blood Cell Counts to Identify Febrile Infants 60 Days or Younger With Invasive Bacterial Infections,” JAMA Pediatr., vol. 171, no. 11, p. e172927, 06 2017, doi: https://doi.org/10.1001/jamapediatrics.2017.2927
Baker MD, Bell LM (May 1999) Unpredictability of serious bacterial illness in febrile infants from birth to 1 month of age. Arch. Pediatr. Adolesc. Med. 153(5):508–511. https://doi.org/10.1001/archpedi.153.5.508
Byington CL et al (Jun. 2004) Serious bacterial infections in febrile infants 1 to 90 days old with and without viral infections. Pediatrics 113(6):1662–1666. https://doi.org/10.1542/peds.113.6.1662
Bonadio W et al (May 2016) Meta-analysis to determine risk for serious bacterial infection in febrile outpatient neonates with RSV infection. Pediatr Emerg Care 32(5):286–289. https://doi.org/10.1097/PEC.0000000000000799
Bulbul L, Bulbul A, Ocak S, Elitok GK (2018) Etiology and causative pathogens of febrile neonates. Pediatric Infect Dis 3(1):1–7
Seiden JA, Zorc JJ, Hodinka RL, Shah SS (Feb. 2010) Lack of cerebrospinal fluid pleocytosis in young infants with enterovirus infections of the central nervous system. Pediatr Emerg Care 26(2):77–81. https://doi.org/10.1097/PEC.0b013e3181ce2fad
Tan NWH, Lee EY, Khoo GMC, Tee NWS, Krishnamoorthy S, Choong CT (Apr. 2016) Cerebrospinal fluid white cell count: discriminatory or otherwise for enteroviral meningitis in infants and young children? J Neuro-Oncol 22(2):213–217. https://doi.org/10.1007/s13365-015-0387-2
M. R. Precit et al., “Cerebrospinal Fluid Findings Are Poor Predictors of Appropriate FilmArray Meningitis/Encephalitis Panel Utilization in Pediatric Patients,” J. Clin. Microbiol., vol. 58, no. 3, Feb. 2020, doi: https://doi.org/10.1128/JCM.01592-19
Chakrabarti P, Warren C, Vincent L, Kumar Y (Oct. 2018) Outcome of routine cerebrospinal fluid screening for enterovirus and human parechovirus infection among infants with sepsis-like illness or meningitis in Cornwall, UK. Eur J Pediatr 177(10):1523–1529. https://doi.org/10.1007/s00431-018-3209-8
Lumley SF, Pritchard D, Dutta A, Matthews PC, Cann K (2018) Multiplex PCR reveals high prevalence of enterovirus and HHV6 in acellular paediatric cerebrospinal fluid samples. J Inf Secur 77(3):249–257. https://doi.org/10.1016/j.jinf.2018.05.008
Ramers C, Billman G, Hartin M, Ho S, Sawyer MH (May 2000) Impact of a diagnostic cerebrospinal fluid enterovirus polymerase chain reaction test on patient management. JAMA 283(20):2680–2685. https://doi.org/10.1001/jama.283.20.2680
Blaschke AJ et al (Apr. 2018) Retrospective evaluation of infants aged 1 to 60 days with residual cerebrospinal fluid (CSF) tested using the FilmArray meningitis/encephalitis(ME) panel. J Clin Microbiol 56(7):e00277–e00218. https://doi.org/10.1128/JCM.00277-18
Bonadio WA, Stanco L, Bruce R, Barry D, Smith D (Jul. 1992) Reference values of normal cerebrospinal fluid composition in infants ages 0 to 8 weeks. Pediatr Infect Dis J 11(7):589–591. https://doi.org/10.1097/00006454-199207000-00015
A. Piralla, B. Mariani, M. Stronati, P. Marone, and F. Baldanti, “Human enterovirus and parechovirus infections in newborns with sepsis-like illness and neurological disorders,” Early Hum. Dev., vol. 90 Suppl 1, pp. S75–77, Mar. 2014, doi: https://doi.org/10.1016/S0378-3782(14)70023-4
Yun KW et al (Oct. 2012) Enteroviral meningitis without pleocytosis in children. Arch Dis Child 97(10):874–878. https://doi.org/10.1136/archdischild-2012-301884
Schnadower D et al (Jul. 2011) Sterile cerebrospinal fluid pleocytosis in young febrile infants with urinary tract infections. Arch Pediatr Adolesc Med 165(7):635–641. https://doi.org/10.1001/archpediatrics.2011.104
Martínez Planas A et al (2012) Low prevalence of invasive bacterial infection in febrile infants under 3 months of age with enterovirus infection. Clin Microbiol Infect Off Publ Eur Soc Clin Microbiol Infect Dis 18(9):856–861. https://doi.org/10.1111/j.1469-0691.2011.03671.x
Rittichier KR et al (2005) Diagnosis and outcomes of enterovirus infections in young infants. Pediatr Infect Dis J 24(6):546–550. https://doi.org/10.1097/01.inf.0000164810.60
King RL, Lorch SA, Cohen DM, Hodinka RL, Cohn KA, Shah SS (Sep. 2007) Routine cerebrospinal fluid enterovirus polymerase chain reaction testing reduces hospitalization and antibiotic use for infants 90 days of age or younger. Pediatrics 120(3):489–496. https://doi.org/10.1542/peds.2007-0252
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
All the authors have no conflict of interest to declare.
Data and material are available.
Ethics approval
The study was approved by the Bnai Zion IRB No BNZ −19-005.
All patients gave their consent to participate in the study and the data to be published.
All patients gave their consent to the data to be published.We give a full consent to publish at the European Journal of Clinical Microbiology & Infectious Diseases.Authors’ contributions –.
All authors whose names appear on the submission.
1) Made substantial contributions to the conception or design of the work; or the acquisition, analysis, or interpretation of data; or the creation of new software used in the work;
2) drafted the work or revised it critically for important intellectual content;
3) Approved the version to be published;
4) Agree to be accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Nassrallah, B., Bamberger, E., Cohen, S. et al. The yield of CSF molecular testing in febrile neonates. Eur J Clin Microbiol Infect Dis 40, 1553–1557 (2021). https://doi.org/10.1007/s10096-021-04168-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10096-021-04168-8