Abstract
Background
Tandem occlusions can complicate medical and endovascular stroke treatment. To identify these occlusions, computed tomography angiography (CTA) represents the best imaging modality. However, CTA is still not initially performed in some patients not admitted directly to stroke centers. Early identification of an additional occlusion of the proximal extracranial internal carotid artery may improve the best suitable treatment strategy. The purpose of this study was to find a valuable threshold of thrombus attenuation in a non-contrast head CT (NCCT) scan to facilitate a safe diagnosis of tandem occlusions.
Materials and methods
Consecutive patients with acute middle cerebral artery (MCA) occlusions who underwent endovascular treatment were identified from our registry of neuroendovascular interventions. Thrombus attenuations of the affected MCA and contralateral vessel were measured by NCCT. To compare individual baseline blood attenuations, the difference between the thrombus attenuation and the contralateral MCA attenuation (referred to as ΔTM) was calculated.
Results
Three hundred and twenty-five patients were included. There was a highly significant difference between mean thrombus attenuation with isolated MCA occlusion and additional extracranial internal carotid artery (ICA) occlusion (49.9 ± 8 vs. 56.2 ± 10 Hounsfield units (HU); P < 0.001). The area under the receiver operating characteristic curve of ΔTM was 0.72. The optimal threshold value was 13.5 HU, with a sensitivity of 67.5% and a specificity of 68.6%.
Conclusion
Despite a significant difference in thrombus attenuation in MCA occlusions with an additional extracranial ICA occlusion compared with isolated MCA occlusions, a relevant threshold of thrombus attenuation was not found.
Similar content being viewed by others
Introduction
Endovascular treatment of acute intracranial artery occlusions has evolved rapidly. There are various treatment options, which include thrombolytic therapy, aspiration, or mechanical thrombectomy with a stent retriever. Information on the presence of thrombus expansion, e.g., tandem internal carotid artery (ICA)/middle cerebral artery (MCA) occlusion, is important for the outcome and material selection when performing mechanical recanalization [1,2,3]. In the anterior circulation, the hyperdense vessel sign on unenhanced (computed tomography) CT is an early and highly reliable indicator of the occlusive thrombus within the affected vessel [4]. However, the initial non-contrast head CT (NCCT) scan does not provide information on an additional proximal artery occlusion. A significantly higher thrombus attenuation was shown in patients with MCA occlusion and additional extracranial ICA occlusion when compared with MCA occlusion only [5].
Most hospitals performing CT in stroke patients usually implement computed tomography angiography (CTA) to confirm an occlusion and determine its location. However, based on our daily clinical practice, some stroke patients only undergo NCCT in the referring hospital. This is because of the lack of proper implementation of the CTA in the stroke imaging protocol or avoiding contrast administration due to kidney dysfunction in the patient. Information on thrombus attenuation may be a suitable tool to predict the expansion of the thrombus and, therefore, may aid in selecting the most suitable treatment modality, medically and/or endovascularly. Even if CTA is available, differentiation between true ICA occlusions and pseudo-occlusions can sometimes be difficult for the radiologist [6]. And even with duplex ultrasound, it can be hard to differentiate between subtotal and total occlusions of the ICA [7]. Consequently, measurement of thrombus attenuation may improve true ICA occlusion detection on unenhanced CT scans. Accordingly, reliable prediction of an additional extracranial ICA occlusion to improve stroke patient care is required. This retrospective study was conducted to assess the clinical benefits of attenuation measurement in patients with acute MCA occlusion as well as the value of a threshold for the diagnosis of an additional extracranial ICA occlusion.
Materials and methods
The research in this study was conducted according to the principles of the Declaration of Helsinki. The local ethics committee approved the research protocol (12/17).
We performed a retrospective review of all consecutively endovascularly treated patients at our institution between 2009 and 2017. Inclusion criteria were an endovascular treated M1 segment occlusion and NCCT before digital subtraction angiography (DSA).
The imaging protocol for patients with suspected stroke in our department consists of a native CCT and CT angiography, followed by CT perfusion, which was performed on an Aquilion 32 Slice CT scanner (Toshiba Medical Systems, Tokyo, Japan) in helical mode (0.5-mm thickness, 120 kV). The native scan was reconstructed in the axial, sagittal, and coronal planes (0.5-mm thickness).
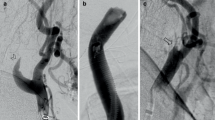
Thrombus attenuation and intraluminal attenuation of the contralateral MCA were measured in manually placed regions of interest perpendicular to the vessel on sagittal reconstructions of the CT scans (Fig. 1). To consider individual baseline blood attenuation in the statistical analysis, differences between the thrombus attenuation and the intraluminal attenuation of the contralateral MCA (referred to as ΔTM) were calculated. Additional extracranial ICA occlusions were noted on CTA and confirmed as true tandem occlusions on DSA.
Statistical analysis was performed using SPSS 25 (IBM, NY, USA). The χ2 test was applied to determine differences in the frequencies. Differences between the means were tested using Student’s t test. For both the attenuation and ΔTM, receiver operating characteristic (ROC) curves were obtained and analyzed to determine the optimal threshold by calculating the sensitivity and specificity. The significance level was defined as P < 0.05.
Results
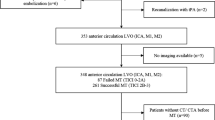
Data of 393 consecutive patients with acute occlusion of the M1 segment of the MCA who underwent endovascular treatment were analyzed retrospectively. Sixty-eight patients were excluded for the following reasons: initial CT scans were performed externally in the referring hospital (n = 55), retrospective analysis of the images was not possible (n = 4), an MRI was performed initially rather than a CT scan (n = 5), and the presence of a metal artifact close to the occluded MCA (n = 3). Spontaneous recanalization occurred in one patient.
From the remaining 325 patients, 162 were male and 163 female, with a mean age of 71 ± 13 years. Of these patients, data from 176 patients on the thrombus attenuation and angiographic results of mechanical thrombectomy with stent retrievers have been published previously [5]. The mean incidence rate of additional extracranial ICA occlusion was 25.5%. The highest rate was found in patients aged between 40 and 49 years (39%).
The mean thrombus attenuation was 6 Hounsfield units (HU) lower (95% CI 4–8 HU) in patients with isolated MCA occlusions (49.9 ± 8 HU) compared with patients with additional ICA occlusion (56.2 ± 10 HU) (Fig. 2a). These values are higher than the respective intraluminal densities of the contralateral MCA (39.2 ± 7 HU for isolated MCA occlusion, 38.2 HU for patients with tandem occlusion). This results in the mean difference ΔTM of 10.6 ± 8 HU for isolated occlusions and ΔTM of 17.8 ± 11 HU for patients with tandem occlusions. Thus, the ΔTM of patients with isolated MCA occlusions is significantly lower by 7 HU than for patients with tandem occlusions (95% CI 5–10 HU) (Fig. 2c). A total of 8 patients, belonging to both patient groups, had a higher attenuation of contralateral MCA than of that of the clots, resulting in a negative ΔTM. Calcified thrombi were observed in 8 (3.3%) patients with isolated MCA occlusions and in 2 (2.4%) patients with tandem occlusions.
a, c Box plot demonstrating the mean and distribution of the attenuation values (density) among patients with an isolated MCA occlusion (no ICA) and additional occlusion of the extracranial ICA (ICA). Graph a shows statistics for the absolute attenuation values, whereas graph c demonstrates statistics of the relative values (ΔTM). b, d Receiver operating characteristic (ROC) curves and corresponding area under the curve (AUC) for the risk score of additional extracranial ICA occlusion. Graph b depicts statistics for the absolute attenuation values, whereas graph c shows statistics for relative values (ΔTM)
ROC analysis (Fig. 2b, d) of thrombus attenuation in patients with an M1 occlusion and additional extracranial ICA occlusion showed an area under the curve (AUC) of 0.71 and an optimal threshold of 53.5 HU, with a sensitivity of 62.8% and a specificity of 68.7%. Furthermore, ROC analysis of the ΔTM of the thrombus attenuations and contralateral MCA attenuations showed an AUC of 0.72 and an optimal threshold of 13.5 HU. The resulting sensitivity was 67.5%, whereas the specificity was 68.6%. Logistic regression analyses (Fig. 3) of ΔTM > 25 HU showed a 50% probability of an additional extracranial ICA occlusion.
Discussion
The presence of a hyperdense vessel sign in NCCT scans is a surrogate of an arterial occlusion by thrombus and represents an early ischemic change in acute stroke [8,9,10]. However, an NCCT scan alone cannot exclude an additional extracranial occlusion. In this study, we investigated whether measurement of thrombus attenuation in MCA occlusions is suitable for predicting an additional extracranial ICA occlusion. The early identification of tandem occlusion in acute stroke could be relevant to decide whether intravenous thrombolysis as bridging therapy may be required. To date, there is a continuous discussion about the increased risk of intracranial hemorrhage due to the bridging therapy and the additional administration of antiplatelet drugs (e.g., aspirin, clopidogrel, or tirofiban) in patients with tandem lesions when carotid stenting is needed [11, 12].
Our retrospective analysis confirmed that there is a significantly higher difference in thrombus attenuations and ΔTM in patients with additional occlusion of the extracranial ICA than in patients with isolated MCA occlusion [5]. The AUC for ΔTM was slightly higher than the curve for absolute HU values. Therefore, the mean differences in thrombus attenuations should be used for analysis.
Calcified thrombi were observed in a similar distribution in both analyzed patient groups with approximately 3%. These results are in line with previous reports [13, 14]. However, in eight cases, a negative ΔTM was found, which is attributable to a higher attenuation of contralateral MCA compared with those of the clots. A high lipid-rich composition might explain this preliminary observation but requires further exploration in larger sample–sized studies. Thrombus attenuation depends on the hematocrit and components such as cellular debris and lipids [15,16,17]. Furthermore, septic components of thrombi may influence the attenuation of thrombi [18].
The attenuation of red blood cell (RBC)–rich thrombi has been recently reported to be higher than that of fibrin-predominant thrombi [19, 20]. Cardioembolic thrombi have been hypothesized to contain a higher percentage of fibrin and fewer RBCs than non-cardioembolic thrombi [16, 21]. As arteriosclerotic events most likely cause tandem occlusions, our finding of higher thrombus attenuation in tandem occlusions is in agreement with these results. In contrast to these observations, Brinjikji et al. [20] found no significant difference in the proportion of RBC-rich thrombi between cardioembolic and large artery atherosclerosis etiologies. Further, Marder et al. [22] did not find differences in the histological composition of thrombus whether derived from cardiac or arterial sources in their series of 25 cases. However, larger prospective histopathological studies are required for confirmation of these findings.
The present study has several limitations. First, the study is restricted by its retrospective design. Second, due to the imbalance of the compared groups, the sample size was limited for a stroke study.
Conclusion
Despite a significant difference in thrombus attenuation in MCA occlusions with an additional extracranial ICA occlusion compared with isolated MCA occlusions, a relevant threshold of thrombus attenuation was not found. Therefore, measurement of thrombus attenuation in MCA occlusions is not suitable for predicting an additional extracranial ICA occlusion. To identify such tandem occlusions, CTA remains the best clinical practice.
Data availability
Not applicable.
Abbreviations
- MCA:
-
Middle cerebral artery
- ICA:
-
Internal carotid artery
- NCCT:
-
Non-contrast head CT scan
- CTA:
-
Computed tomography angiography
- HU:
-
Hounsfield unit
References
Papanagiotou P, Roth C, Walter S, Behnke S, Grunwald IQ, Viera J, Politi M, Körner H, Kostopoulos P, Haass A, Fassbender K, Reith W (2011) Carotid artery stenting in acute stroke. J Am Coll Cardiol 58:2363–2369. https://doi.org/10.1016/j.jacc.2011.08.044
Stampfl S, Ringleb PA, Möhlenbruch M, Hametner C, Herweh C, Pham M, Bösel J, Haehnel S, Bendszus M, Rohde S (2014) Emergency cervical internal carotid artery stenting in combination with intracranial thrombectomy in acute stroke. AJNR Am J Neuroradiol 35:741–746. https://doi.org/10.3174/ajnr.A3763
Rubiera M, Ribo M, Delgado-Mederos R, Santamarina E, Delgado P, Montaner J, Alvarez-Sabín J, Molina CA (2006) Tandem internal carotid artery/middle cerebral artery occlusion: an independent predictor of poor outcome after systemic thrombolysis. Stroke 37:2301–2305. https://doi.org/10.1161/01.STR.0000237070.80133.1d
Tomsick TA, Brott TG, Chambers AA, Fox AJ, Gaskill MF, Lukin RR, Pleatman CW, Wiot JG, Bourekas E (1990) Hyperdense middle cerebral artery sign on CT: efficacy in detecting middle cerebral artery thrombosis. AJNR Am J Neuroradiol 11:473–477
Yilmaz U, Roth C, Reith W, Papanagiotou P (2013) Thrombus attenuation does not predict angiographic results of mechanical thrombectomy with stent retrievers. AJNR Am J Neuroradiol 34:2184–2186. https://doi.org/10.3174/ajnr.A3565
Diouf A, Fahed R, Gaha M, Chagnon M, Khoury N, Kotowski M, Guilbert F, Landry D, Raymond J, Roy D, Weill A (2018) Cervical internal carotid occlusion versus pseudo-occlusion at CT angiography in the context of acute stroke: an accuracy, interobserver, and intraobserver agreement study. Radiology 286:1008–1015. https://doi.org/10.1148/radiol.2017170681
Ohm C, Bendick PJ, Monash J, Bove PG, Brown OW, Long GW, Zelenock GB, Shanley CJ (2005) Diagnosis of total internal carotid occlusions with duplex ultrasound and ultrasound contrast. Vasc Endovasc Surg 39:237–243. https://doi.org/10.1177/153857440503900304
Morita S, Ueno E, Masukawa A, Suzuki K, Machida H, Fujimura M (2010) Hyperattenuating signs at unenhanced CT indicating acute vascular disease. RadioGraphics 30:111–125. https://doi.org/10.1148/rg.301095052
Pressman B, Tourje E, Thompson (1987) An early CT sign of ischemic infarction: increased density in a cerebral artery. Am J Roentgenol 149:583–586 . https://doi.org/10.2214/ajr.149.3.583
Mair G, Boyd EV, Chappell FM, von Kummer R, Lindley RI, Sandercock P, Wardlaw JM (2015) Sensitivity and specificity of the hyperdense artery sign for arterial obstruction in acute ischemic stroke. Stroke 46:102–107. https://doi.org/10.1161/STROKEAHA.114.007036
van de Graaf RA, Chalos V, del Zoppo GJ, van der Lugt A, Dippel DWJ, Roozenbeek B (2018) Periprocedural antithrombotic treatment during acute mechanical thrombectomy for ischemic stroke: a systematic review. Front Neurol 9:238. https://doi.org/10.3389/fneur.2018.00238
Zhu F, Anadani M, Labreuche J, Spiotta A, Turjman F, Piotin M, Steglich-Arnholm H, Holtmannspötter M, Taschner C, Eiden S, Haussen DC, Nogueira RG, Papanagiotou P, Boutchakova M, Siddiqui AH, Lapergue B, Dorn F, Cognard C, Killer-Oberpfalzer M, Mangiafico S, Ribo M, Psychogios MN, Labeyrie M-A, Mazighi M, Biondi A, Anxionnat R, Bracard S, Richard S, Gory B, TITAN (Thrombectomy in Tandem Lesions) Investigators (2020) Impact of antiplatelet therapy during endovascular therapy for tandem occlusions: a collaborative pooled analysis. Stroke:STROKEAHA119028231. https://doi.org/10.1161/STROKEAHA.119.028231
Walker BS, Shah LM, Osborn AG (2014) Calcified cerebral emboli, a “do not miss” imaging diagnosis: 22 new cases and review of the literature. Am J Neuroradiol 35:1515–1519. https://doi.org/10.3174/ajnr.A3892
Cerase A, Grazzini I (2015) Early relapsing calcified cerebral embolism. J Stroke Cerebrovasc Dis 24:e125–e126. https://doi.org/10.1016/j.jstrokecerebrovasdis.2015.01.038
Kirchhof K, Welzel T, Mecke C, Zoubaa S, Sartor K (2003) Differentiation of white, mixed, and red thrombi: value of CT in estimation of the prognosis of thrombolysis—phantom study. Radiology 228:126–130. https://doi.org/10.1148/radiol.2273020530
Niesten JM, van der Schaaf IC, van Dam L, Vink A, Vos JA, Schonewille WJ, de Bruin PC, Mali WPTM, Velthuis BK (2014) Histopathologic composition of cerebral thrombi of acute stroke patients is correlated with stroke subtype and thrombus attenuation. PLoS One 9:e88882. https://doi.org/10.1371/journal.pone.0088882
Velasco Gonzalez A, Buerke B, Görlich D, Fobker M, Rusche T, Sauerland C, Meier N, Jeibmann A, McCarthy R, Kugel H, Sporns P, Faldum A, Paulus W, Heindel W (2020) Clot analog attenuation in non-contrast CT predicts histology: an experimental study using machine learning. Transl Stroke Res 11:940–949. https://doi.org/10.1007/s12975-019-00766-z
Hernández-Fernández F, Rojas-Bartolomé L, García-García J, Ayo-Martín Ó, Molina-Nuevo JD, Barbella-Aponte RA, Serrano-Heras G, Juliá-Molla E, Pedrosa-Jiménez MJ, Segura T (2017) Histopathological and bacteriological analysis of thrombus material extracted during mechanical thrombectomy in acute stroke patients. Cardiovasc Intervent Radiol 40:1851–1860. https://doi.org/10.1007/s00270-017-1718-x
Liebeskind DS, Sanossian N, Yong WH, Starkman S, Tsang MP, Moya AL, Zheng DD, Abolian AM, Kim D, Ali LK, Shah SH, Towfighi A, Ovbiagele B, Kidwell CS, Tateshima S, Jahan R, Duckwiler GR, Viñuela F, Salamon N, Villablanca JP, Vinters HV, Marder VJ, Saver JL (2011) CT and MRI early vessel signs reflect clot composition in acute stroke. Stroke 42:1237–1243. https://doi.org/10.1161/STROKEAHA.110.605576
Brinjikji W, Duffy S, Burrows A, Hacke W, Liebeskind D, Majoie CBLM, Dippel DWJ, Siddiqui AH, Khatri P, Baxter B, Nogeuira R, Gounis M, Jovin T, Kallmes DF (2017) Correlation of imaging and histopathology of thrombi in acute ischemic stroke with etiology and outcome: a systematic review. J Neurointervent Surg 9:529–534. https://doi.org/10.1136/neurintsurg-2016-012391
Maekawa K, Shibata M, Nakajima H, Mizutani A, Kitano Y, Seguchi M, Yamasaki M, Kobayashi K, Sano T, Mori G, Yabana T, Naito Y, Shimizu S, Miya F (2018) Erythrocyte-rich thrombus is associated with reduced number of maneuvers and procedure time in patients with acute ischemic stroke undergoing mechanical thrombectomy. Cerebrovasc Dis Extra 8:39–49. https://doi.org/10.1159/000486042
Marder VJ, Chute DJ, Starkman S, Abolian AM, Kidwell C, Liebeskind D, Ovbiagele B, Vinuela F, Duckwiler G, Jahan R, Vespa PM, Selco S, Rajajee V, Kim D, Sanossian N, Saver JL (2006) Analysis of thrombi retrieved from cerebral arteries of patients with acute ischemic stroke. Stroke 37:2086–2093. https://doi.org/10.1161/01.STR.0000230307.03438.94
Acknowledgements
Open Access funding enabled and organized by Projekt DEAL.
Code availability
SPSS 25 (IBM, NY, USA).
Author information
Authors and Affiliations
Contributions
Ruben Mühl-Benninghaus: conceptualization, data collection, analysis and interpretation, manuscript draft writing/editing
Julia Dressler: conceptualization, data collection, statistical analysis and interpretation
Alena Haußmann: conceptualization, data collection
Andreas Simgen: data collection, resources
Wolfgang Reith: project development, resources, supervision
Umut Yilmaz: project development, resources, supervision
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Ethics approval
All procedures performed in the studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki Declaration and its later amendments or comparable ethical standards (approved by the local ethics committee, Ethikkommission des Saarlandes (No.12/17)).
Consent to participate
Not applicable.
Consent for publication
Not applicable.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Mühl-Benninghaus, R., Dressler, J., Haußmann, A. et al. Utility of Hounsfield unit in the diagnosis of tandem occlusion in acute ischemic stroke. Neurol Sci 42, 2391–2396 (2021). https://doi.org/10.1007/s10072-020-04798-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10072-020-04798-4