Abstract
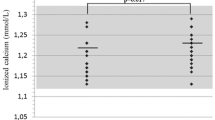
This current study aimed to evaluate the frequency of low bone mass, osteopenia, and osteoporosis in patients with myasthenia gravis (MG) and to investigate the possible association between bone mineral density (BMD) and plasma levels of bone metabolism markers. Eighty patients with MG and 62 controls BMD were measured in the right femoral neck and lumbar spine by dual-energy X-ray absorptiometry. Plasma concentrations of osteocalcin, osteopontin, osteoprotegerin, tumor necrosis factor (TNF-α), interleukin (IL)-1β, IL-6, dickkopf (DKK-1), sclerostin, insulin, leptin, adrenocorticotropic hormone, parathyroid hormone, and fibroblast growth factor (FGF-23) were analyzed by Luminex®. The mean age of patients was 41.9 years, with 13.5 years of length of illness, and a mean cumulative dose of glucocorticoids 38,123 mg. Patients had significant reduction in BMD of the lumbar, the femoral neck, and in the whole body when compared with controls. Fourteen percent MG patients had osteoporosis at the lumbar spine and 2.5% at the femoral neck. In comparison with controls, patients with MG presented lower levels of osteocalcin, adrenocorticotropic hormone, parathyroid hormone, sclerostin, TNF-α, and DKK-1 and higher levels of FGF-23, leptin, and IL-6. There was a significant negative correlation between cumulative glucocorticoid dose and serum calcium, lumbar spine T-score, femoral neck BMD, T-score, and Z-score. After multivariate analysis, higher TNF-α levels increased the likelihood of presenting low bone mass by 2.62. MG patients under corticotherapy presented low BMD and altered levels of bone markers.

Similar content being viewed by others
References
Farrugia ME, Vincent A (2010) Autoimmune mediated neuromuscular junction defects. Curr Opin Neurol 23:489–495
Kumar V, Kaminski HJ (2011) Treatment of myasthenia gravis. Curr Neurol Neurosci Rep 11:89–96
Grossman JM, Gordon R, Ranganath VK, Deal C, Caplan L, Chen W, Curtis JR, Furst DE, McMahon M, Patkar NM, Volkmann E, Saag KG (2010) American College of Rheumatology 2010 recommendations for the prevention and treatment of glucocorticoid-induced osteoporosis. Arthritis Care Res 62:1515–1526
Wakata N, Nemoto H, Sugimoto H (2004) Bone density in myasthenia gravis patients receiving long-term prednisolone therapy. Clin Neurol Neurosurg 106:139–141
Pouwels S, Boer A, Javaid MK, Jones DH, Verschuuren J, Cooper C, Leufkens HG, Vries F (2012) Fracture rate in patients with myasthenia gravis: the general practice research database. Osteoporos Int 24(2):467–476
Konno S, Suzuki S, Masuda M, Nagane Y, Tsuda E, Murai H, Imai T, Fujioka T, Suzuki N, Utsugisawa K (2015) Association between glucocorticoid-induced osteoporosis and myasthenia gravis: a cross-sectional study. PLoS One 10:1–7
Maricic M, Gluck O (2004) Densitometry in glucocorticoid-induced osteoporosis. J Clin Densitom 7:359–363
Gifre L, Ruiz-Gaspà S, Monegal A, Nomdedeu B, Filella X, Guañabens N, Peris P (2013) Effect of glucocorticoid treatment on Wnt signalling antagonists (sclerostin and Dkk-1) and their relationship with bone turnover. Bone 57:272–276
Jaretzki A, Barohn RJ, Ernstoff RM, Kaminski HJ, Keesey JG, Penn AS (2000) Myasthenia gravis. Recommendations for clinical research standards. Task Force of the Medical Scientific Advisory Board of the Myasthenia Gravis Foundation of America. Neurology 55:1–14
Burns TM, Conaway M, Sanders DB (2010) The MG composite. A valid and reliable outcome measure for myasthenia gravis. Neurology 74:1434–1440
Kanis JA, Delmas P, Burckhardt P, Cooper C, Torgerson D (1997) Guidelines for diagnosis and management of osteoporosis. The European Foundation for Osteoporosis and Bone Disease. Osteoporos Int 7:390–406
Yeh JH, Chen HJ, Chen YK, Chiu HC, Kao CH (2014) Increased risk of osteoporosis in patients with myasthenia gravis. A population-based cohort study. Neurology 83:1–5
Buehring B, Viswanathan R, Binkley N, Busse W (2013) Glucocorticoid-induced osteoporosis: an update on effects and management. J Allergy Clin Immunol 132:1019–1030
Pereira RMR, Carvalho JFC, Paula AP, Zerbini C, Domiciano DS, Gonçalves H, Danowski JS, Neto JFM, Mendonça LMC, Bezerra MC, Terreri MT, Imamura M, Weingrill P, Plapler PG, Radominski S, Tourinho T, Szejnfeld VL, Andrada NC (2012) Diretrizes para prevenção e tratamento da osteoporose induzida por glicocorticoide. Rev Bras Reumatol 52:569–593
Gudbjornsson B, Juliusson UI, Gudbjornsson FV (2002) Prevalence of long-term steroid treatment and the frequency of decision making to prevent steroid induced osteoporosis in daily clinical practice. Ann Rheum Dis 61:32–26
Mitra R (2011) Adverse effects of corticosteroids on bone metabolism: a review. Am J Phy Med Rehabil 3:466–471
van Staa TP, Leufkens HGM, Cooper C (2002) The epidemiology of corticosteroid-induced osteoporosis: a meta-analysis. Osteoporos Int 13:777–787
Holick FH (2007) Vitamin D deficiency. N Engl J Med 357:266–281
Bischoff-Ferrari HA, Dietrich T, Orav EJ, Dawson-Hughes B (2004) Positive association between 25-hydroxy vitamin D levels and bone mineral density: a population-based study of younger and older adults. Am J Med 116:634–639
D’Amelio P, Grimaldi A, Bernabei P (2006) Immune system and bone metabolism: does thymectomy influence postmenopausal bone loss in humans? Bone 39:658–665
Ahmed HH, Morcos NYS, Eskander EF, Seoudi DMS, Shalby AB (2012) Potential role of leptin against glucocorticoid-induced secondary osteoporosis in adult female rats. Eur Rev Med Pharmacol Sci 16:1446–1452
Shiguang L, Gupta QA, Darryl L (2007) Emerging role of fibroblast growth factor 23 in a bone–kidney axis regulating systemic phosphate homeostasis and extracellular matrix mineralization. Curr Opin Nephrol Hypertens 16:329–335
Henneicke H, Gasparini SJ, Brennan-Speranza TC, Zhou H, Seibel MJ (2014) Glucocorticoids and bone: local effects and systemic implications. Trends Endocrinol Metabol 25:197–211
Yao Z, Li P, Zhang Q, Schwarz EM, Keng P, Arbini A et al (2006) Tumor necrosis factor-alpha increases circulating osteoclast precursor numbers by promoting their proliferation and differentiation in the bone marrow through up-regulation of c-Fms expression. J Biol Chem 281:11846–11855
Franchimont N, Wertz S, Malaise M (2005) Interleukin-6: an osteotropic factor influencing bone formation? Bone 37:601–606
Ishimi Y, Miyaura C, Jin CH, Akatsu T, Abe E, Nakamura Y et al (1990) IL-6 is produced by osteoblasts and induces bone resorption. J Immunol 145:3297–3303
Carr AS, Cardwell CR, McCarron PO, McConville K (2010) A systematic review of population based epidemiological studies in myasthenia gravis. BMC Neurol 10:1–9
Acknowledgements
This study was funded by Brazilian government funding agencies Conselho Nacional de Desenvolvimento Científico e Tecnológico (CNPq), Coordenação de Aperfeiçoamento de Pessoal de Nível Superior (CAPES), and Fundação de Amparo à Pesquisa do Estado de Minas Gerais (FAPEMIG), Brazil.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
All subjects provided written informed consent before admission to the study. The Research Ethics Committee of the Universidade Federal de Minas Gerais, Brazil, approved this study (protocol number: CAAE-19045413.0.0000.5149).
Conflicts of interest
The authors declare that they have no conflict of interest.
Rights and permissions
About this article
Cite this article
Braz, N.F.T., Rocha, N.P., Vieira, É.L.M. et al. Negative impact of high cumulative glucocorticoid dose on bone metabolism of patients with myasthenia gravis. Neurol Sci 38, 1405–1413 (2017). https://doi.org/10.1007/s10072-017-2964-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10072-017-2964-z