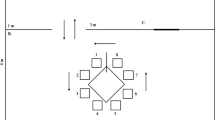
Abstract
The odor span task (OST) is frequently used to assess memory capacity in rodents. Odor stimuli are presented in a large arena and choices of session-novel odors produce food reward. The procedure can be described as an incrementing non-matching-to-sample contingency because on each trial one new stimulus is presented along with one or more previously presented (non-reinforced) comparison odors. An automated version of this task has recently been developed in which odors are presented with an olfactometer in an operant chamber using a successive conditional discrimination procedure. The present study compared the acquisition of matching- vs. non-matching-to-sample versions of the task with six rats tested under each procedure. All six rats trained on the non-matching variation showed rapid acquisition of the discrimination with high rates of responding to odor stimuli when they were session-novel and low rates of responding to subsequent presentations of those odors. However, only three of the six rats trained on the matching variation met acquisition criteria, and two of the three that did acquire the task required extensive training to do so. These results support findings from the OST that rats can differentiate between stimuli that are session-novel and those previously encountered, but also that a matching contingency is more difficult to learn than a non-matching arrangement. These findings parallel differences observed between acquisition of simple matching- and non-matching-to-sample tasks, but accounts such as novelty preference or the oddity preference effect may not be sufficient to explain the present results.







Similar content being viewed by others
Availability of data and material (data transparency)
The raw data supporting the conclusions of this article will be made available by the authors, without undue reservation.
Code availability
Not applicable.
References
Antunes M, Biala G (2012) The novel object recognition memory: neurobiology, test procedure, and its modifications. Cogn Process 13:93–110
April LB, Bruce K, Galizio M (2013) The magic number 70 (plus or minus 20): variables determining performance in the rodent odor span task. Learn Motiv 44(3):143–158
Bratch A, Kann S, Cain JA, Wu JE, Rivera-Reyes N, Dalecki S, Crystal JD (2016) Working memory systems in the rat. Curr Biol 26(3):351–355
Bruce K, Dyer K, Mathews M, Nealley C, Phasukkan T, Prichard A, Galizio M (2018) Successive odor matching- and non-matching-to-sample in rats: a reversal design. Behavioral Processes 155:26–32
Cowie S, Davison M (2020) Generalizing from the past, choosing the future. Perspect Behav Sci 43(2):245–258
Cowie S, Davison M (2021) Pigeons prefer to invest early for future reinforcers. J Exp Anal Behav 115(3):650–666
De Falco E, An L, Sun N, Roebuck AJ, Greba Q, Lapish CC, Howland JG (2019) The rat medial prefrontal cortex exhibits flexible neural activity states during the performance of an odor span task. Eneuro 6(2):1–16
de Lima MNM, Laranja DC, Bromberg E, Roesler R, Schröder N (2005) Pre-or post-training administration of the NMDA receptor blocker MK-801 impairs object recognition memory in rats. Behav Brain Res 156(1):139–143
Dudchenko PA, Wood ER, Eichenbaum H (2000) Neurotoxic hippocampal lesions have no effect on odor span and little effect on odor recognition memory but produce significant impairments on spatial span, recognition, and alternation. J Neurosci 20:2964–2977
Ennaceur A (2010) One-trial object recognition in rats and mice: methodological and theoretical issues. Behav Brain Res 215:244–254
Galizio M, Deal M, Hawkey A, April B (2013) Working memory in the odor span task: effects of chlordiazepoxide, dizocilpine (MK801), morphine, and scopolamine. Psychopharmacology 225:397–406
Galizio M, Deal M, Mathews M, Panoz-Brown D, Prichard A, Bruce KE (2019) Effects of NMDA antagonist dizocilpine (MK-801) are modulated by the number of distractor stimuli in the rodent odor span task of working memory. Neurobiol Learn Mem 161:51–56
Galizio M, Mason MG, Bruce K (2020) Successive incrementing non-matching-to-samples in rats: an automated version of the odor span task. J Exp Anal Behav 114(2):248–265
Krichbaum S, Rogers B, Cox E, Waggoner LP, Katz JS (2020) Odor span task in dogs (Canis familiaris). Anim Cogn 23(3):571–580
Lazarowski L, Goodman A, Galizio M, Bruce K (2019) Effects of set size on identity and oddity abstract-concept learning in rats. Anim Cogn 22:733–742
MacQueen DA, Bullard L, Galizio M (2011) Effects of dizocilpine (MK801) on olfactory span in rats. Neurobiol Learn Mem 95:57–63
MacQueen DA, Dalrymple SR, Drobes DJ, Diamond DM (2016) Influence of pharmacological manipulations of NMDA and cholinergic receptors on working versus reference memory in a dual component odor span task. Learn Mem 23(6):270–277
Marks WN, Parker ME, Zabder NK, Greba Q, Snutch TP, Howland JG (2018) T-type calcium channels in the orbitofrontal cortex mediate sensory integration as measured using a spontaneous oddity task in rats. Learn Mem 25(7):317–324
Nigrosh BJ, Slotnick BM, Nevin JA (1975) Olfactory discrimination, reversal learning, and stimulus control in rats. J Comp Physiol Psychol 89(4):285–294
Rushforth SL, Steckler T, Shoaib M (2011) Nicotine improves working memory span capacity in rats following sub-chronic ketamine exposure. Neuropsychopharmacology 36(13):2774–2781
Scott GA, Liu MC, Tahir NB, Zabder NK, Song Y, Greba Q, Howland JG (2020) Roles of the medial prefrontal cortex, mediodorsal thalamus, and their combined circuit for performance of the odor span task in rats: analysis of memory capacity and foraging behavior. Learn Mem 27(2):67–77
Shahan TA (2017) Moving beyond reinforcement and response strength. Behav Anal 40(1):107–121
Slotnick BM, Kufera A, Silberberg AM (1991) Olfactory learning and odor memory in the rat. Physiol Behav 50(3):555–561
Spinetta MJ, Woodlee MT, Feinberg LM, Stroud C, Schallert K, Cormack LK, Schallert T (2008) Alcohol-induced retrograde memory impairment in rats: prevention by caffeine. Psychopharmacology 201(3):361–371
Thomas RK, Noble LM (1988) Visual and olfactory oddity learning in rats: what evidence is necessary to show conceptual behavior? Anim Learn Behav 16(2):157–163
White KG (2013) Remembering and forgetting. In: Madden GJ (ed) APA handbook of behavior analysis, vol. 1: methods and principles. American Psychological Association, pp 411–437
Wright AA, Delius JD (2005) Learning processes in matching and oddity: the oddity preference effect and sample reinforcement. J Exp Psychol Anim Behav Process 31(4):425–432
Acknowledgements
The authors thank Angela Bennett, Ashley Campbell, Genevieve Guidone, Chandler Hicks, Calista Holt, Jonathan Shaw and Shandy Smith for assistance in data collection. Funding was provided by grants from the UNC Wilmington CSURF program.
Funding
Not applicable.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no conflicts of interest or competing interests.
Ethical approval
This research complied with the current laws of the United States of America and was reviewed and approved by the Institutional Animal Care and Use Committee (IACUC) at the University of North Carolina Wilmington.
Consent for publication
The authors have all approved this version of the manuscript and give consent for publication.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Wagner, T.J., Bruce, K. & Galizio, M. Incrementing non-matching- but not matching-to-sample is rapidly learned in an automated version of the odor span task. Anim Cogn 25, 1259–1270 (2022). https://doi.org/10.1007/s10071-022-01608-6
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10071-022-01608-6