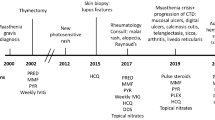
Abstract
Coexistent myasthenia gravis (MG) and primary Sjögren’s syndrome (pSS) is an absolute rarity. That is kind of a surprise as both entities seem to share the same corresponding immunologic mechanisms. We hereby report the case of a 41-year-old woman with coincident early-onset MG (EOMG) and pSS. Because EOMG was the leading clinical feature, she was primarily treated by innovative non-intubated uniportal subxiphoid video-assisted thoracoscopic surgery (VATS) thymectomy. As the association of EOMG and pSS is so unusual, we contextualize our findings with the relevant literature. Particular relevance is an anti-nuclear antibody screening throughout the clinical course of MG and—in reverse—a screening for MG variables when pSS patients complain either muscle fatigability or fatigable ptosis. As pSS patients do not develop any serious morbidity, supervising MG progress in patients with both diseases is of utmost importance. Apart from conscientious pSS diagnosis, prompt adjusting of EOMG progress is the essential aspect of targeted treatment. In this context, it is relevant that therapeutic decisions are made in a multidisciplinary approach. Due to its rarity, multicenter prospective studies of larger sample sizes are indispensably needed to obtain a better understanding of this unusual link.


Similar content being viewed by others
Data availability
The datasets generated and analyzed during the current study are available from the corresponding author on reasonable request. In principle, all data generated and analyzed during this study are included in this article.
Change history
11 August 2022
A Correction to this paper has been published: https://doi.org/10.1007/s10067-022-06328-3
References
Gilhus NE (2016) Myasthenia gravis. N Engl J Med 375:2570–2581
Nacu A, Andersen JB, Lisnic V, Owe JF, Gilhus NE (2015) Complicating autoimmune diseases in myasthenia gravis: a review. Autoimmunity 48:362–368
Li X, Zhao Y, Liao Q, Da Y (2020) Myasthenia gravis coexisting with primary Sjögren’s syndrome: report of three cases and literature review. Front Neurol 11:939
Evoli A, Caliandro P, Iorio R, Alboini PE, Damato V, LaTorre G et al (2015) Polyautoimmunity in patients with myasthenia gravis: a single-center experience. Autoimmunity 48:412–417
Mariette X, Criswell LA (2018) Primary sjögren’s syndrome. N Engl J Med 378:931–939
Tian Y, Yang H, Liu N, Li Y, Chen J (2021) Advances in pathogenesis of Sjögren’s syndrome. J Immunol Res 5928232
Zhou D, McNamara NA (2014) Macrophages: important players in primary Sjögren’s syndrome? Expert Rev Clin Immunol 10:513–520
Karabiyik A, Peck AB, Nguyen CQ (2013) The important role of T cells and receptor expression in Sjögren’s syndrome. Scand J Immunol 78:157–166
van Blokland SC, Wierenga-Wolf AF, van Helden-Meeuwsen CG, Drexhage HA, Hooijkaas H, van de Merwe JP et al (2000) Professional antigen presenting cells in minor salivary glands in Sjögren’s syndrome: potential contribution to the histopathological diagnosis? Lab Invest 80:1935–1941
Davidson A, Diamond B (2001) Autoimmune diseases. N Engl J Med 345:340–350
Sardu C, Cocco E, Mereu A, Massa R, Cuccu A, Marrosu MG et al (2012) Population based study of 12 autoimmune diseases in Sardinia, Italy: prevalence and comorbidity. PLoS ONE 7:e32487
Waterfield M, Anderson MS (2010) Clues to immune tolerance: the monogenic autoimmune syndromes. Ann N Y Acad Sci 1214:138–155
Bach JF (2012) The etiology of autoimmune diseases: the case of myasthenia gravis. Ann N Y Acad Sci 1274:33–39
Zhernakova A, Withoff S, Wijmenga C (2013) Clinical implications of shared genetics and pathogenesis in autoimmune diseases. Nat Rev Endocrinol 9:646–659
Avidan N, Le Panse R, Berrih-Aknin S, Miller A (2014) Genetic basis of myasthenia gravis—a comprehensive review. J Autoimmun 52:146–153
Lazarus MN, Isenberg DA (2005) Development of additional autoimmune diseases in a population of patients with primary Sjögren’s syndrome. Ann Rheum Dis 64:1062–1064
Rojas-Villarraga A, Amaya-Amaya J, Rodriguez-Rodriguez A, Mantilla RD, Anaya JM (2012) Introducing polyautoimmunity: secondary autoimmune diseases no longer exist. Autoimmune Dis 2012:254319
Anaya JM, Restrepo-Jiménez P, Rodríguez Y, Rodríguez-Jiménez M, Acosta-Ampudia Y, Monsalve DM et al (2019) Sjögren’s syndrome and autoimmune thyroid disease: two sides of the same coin. Clin Rev Allergy Immunol 56:362–374
Duarte S, Santos E, Martins J, Martins Silva A, Lopes C, Gonçalves G et al (2017) Myasthenia gravis with systemic and neurological polyautoimmunity. J Neurol Sci 381:39–40
Garibaldi M, Fionda L, Vanoli F, Leonardi L, Loreti S, Bucci E et al (2020) Muscle involvement in myasthenia gravis: expanding the clinical spectrum of Myasthenia-Myositis association from a large cohort of patients. Autoimmun Rev 19:102498
Uchio N, Taira K, Ikenaga C, Kadoya M, Unuma A, Yoshida K et al (2018) Inflammatory myopathy with myasthenia gravis: thymoma association and polymyositis pathology. Neurol Neuroimmunol Neuroinflamm 6:e535
Berrih-Aknin S (2014) Myasthenia gravis: paradox versus paradigm in autoimmunity. J Autoimmun 52:1–28
Sanders DB, Wolfe GI, Benatar M, Evoli A, Gilhus NE, Illa I et al (2016) International consensus guidance for management of myasthenia gravis: executive summary. Neurology 87:419–425
Sanders DB, Wolfe GI, Narayanaswami P; MGFA Task Force on MG Treatment Guidance (2018) Developing treatment guidelines for myasthenia gravis. Ann N Y Acad Sci 1412:95–101
Ramos-Casals M, Brito-Zerón P, Bombardieri S, Bootsma H, De Vita S, Dörner T, et al; EULAR-Sjögren Syndrome Task Force Group (2020) EULAR recommendations for the management of Sjögren’s syndrome with topical and systemic therapies. Ann Rheum Dis 79:3-18
Wolfe GI, Kaminski HJ, Aban IB, Minisman G, Kuo HC, Marx A, et al; MGTX Study Group (2016) Randomized trial of thymectomy in myasthenia gravis. N Engl J Med 375:511-22
Litchman T, Roy B, Kumar A, Sharma A, Njike V, Nowak RJ (2020) Differential response to rituximab in anti-AChR and anti-MuSK positive myasthenia gravis patients: a single-center retrospective study. J Neurol Sci 411:116690
Mena-Vázquez N, Fernández-Nebro A, Pego-Reigosa JM, Galindo M, Melissa-Anzola A, Uriarte-Isacelay E et al (2020) Hydroxychloroquine is associated with a lower risk of polyautoimmunity: data from the RELESSER Registry. Rheumatology (Oxford) 59:2043–2051
Varan O, Kucuk H, Tufan A (2015) Myasthenia gravis due to hydroxychloroquine. Reumatismo 67:849
Jallouli M, Saadoun D, Eymard B, Leroux G, Haroche J, Le Thi HD et al (2012) The association of systemic lupus erythematosus and myasthenia gravis: a series of 17 cases, with a special focus on hydroxychloroquine use and a review of the literature. J Neurol 259:1290–1297
The American College of Rheumatology nomenclature and case definitions for neuropsychiatric lupus syndromes (1999) Arthritis Rheum 42:599–608
Fujieda Y (2020) Diversity of neuropsychiatric manifestations in systemic lupus erythematosus. Immunol Med 43:135–141
Author information
Authors and Affiliations
Contributions
All authors contributed in different degrees to the conception and design of the work, the acquisition, analysis, and interpretation of data. M. Hartert drafted the manuscript. B. Melcher created and provided the pathological images. M. Huertgen helped with finalizing the manuscript. All authors approved the final manuscript.
Corresponding author
Ethics declarations
Ethics approval
The authors are accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved. All procedures depicted in this case were in accordance with the ethical standards of the institutional and/or national research committee(s) and with the Helsinki Declaration (as revised in 2013). Written informed consent was obtained from the patient for publication of this case report and accompanying images.
Disclosures
None.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
The original online version of this article was revised: Table 2 data has been aligned correctly.
Rights and permissions
Springer Nature or its licensor holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Hartert, M., Melcher, B. & Huertgen, M. Association of early-onset myasthenia gravis and primary Sjögren’s syndrome: a case-based narrative review. Clin Rheumatol 41, 3237–3243 (2022). https://doi.org/10.1007/s10067-022-06294-w
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10067-022-06294-w