Abstract
Introduction
Antibody against cyclic citrullinated protein (ACPA) is counted as one of the most important biomarkers in diagnosis, classification, and prognosis of rheumatoid arthritis (RA). We examined the evolution of ACPA during disease course and assess predictive value of time-weighted cumulative ACPA titer on radiographic progression in RA patients.
Method
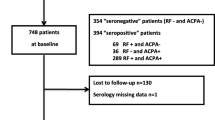
A group of 734 patients with RA was followed longitudinally over 2 years, with annual measurements of ACPA. The cumulative titers of ACPA were calculated using the trapezoidal rule and were divided into three categories: negative, low-to-moderate, and high. Radiographs of the hands were scored with the modified Sharp score (SHS). Multivariable logistic regression models were performed to identify independent predictors over follow-up for individual patients with different combinations of risk factors. The effect size was computed by Cohen’s d method.
Results
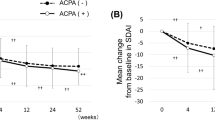
The patients with radiographic progression had a higher SHS at baseline; and smoking status, diabetes, RF positivity, and use of biologic DMARDs were independently associated with radiographic progression (all P < 0.05). As for ACPA, reversion happened more commonly in men and was associated with younger onset age and lower titer at baseline, but it had no direct relevance to radiographic outcome. In multivariable regression analysis, only high cumulative or baseline titer of ACPA had a predictive power for rapid radiographic progression (all P < 0.05), and cumulative ACPA titer was superior in terms of statistical significance (Cohen’s d, 0.637 versus 0.583).
Conclusions
High cumulative ACPA titer was independently associated with accelerated radiographic progression, especially with initiation of joint damage.


Similar content being viewed by others
References
Smolen JS, Aletaha D, Barton A, Burmester GR, Emery P, Firestein GS, Kavanaugh A, McInnes IB, Solomon DH, Strand V, Yamamoto K (2018) Rheumatoid arthritis. Nat Rev Dis Primers 4:18001. https://doi.org/10.1038/nrdp.2018.1
Neogi T, Aletaha D, Silman AJ, Naden RL, Felson DT, Aggarwal R, Bingham CO 3rd, Birnbaum NS, Burmester GR, Bykerk VP, Cohen MD, Combe B, Costenbader KH, Dougados M, Emery P, Ferraccioli G, Hazes JM, Hobbs K, Huizinga TW, Kavanaugh A, Kay J, Khanna D, Kvien TK, Laing T, Liao K, Mease P, Menard HA, Moreland LW, Nair R, Pincus T, Ringold S, Smolen JS, Stanislawska-Biernat E, Symmons D, Tak PP, Upchurch KS, Vencovsky J, Wolfe F, Hawker G (2010) The 2010 American College of Rheumatology/European League Against Rheumatism classification criteria for rheumatoid arthritis: Phase 2 methodological report. Arthritis Rheum 62:2582–2591. https://doi.org/10.1002/art.27580
Nielen MM, van Schaardenburg D, Reesink HW, van de Stadt RJ, van der Horst-Bruinsma IE, de Koning MH, Habibuw MR, Vandenbroucke JP, Dijkmans BA (2004) Specific autoantibodies precede the symptoms of rheumatoid arthritis: a study of serial measurements in blood donors. Arthritis Rheum 50:380–386. https://doi.org/10.1002/art.20018
van Venrooij WJ, van Beers JJ, Pruijn GJ (2011) Anti-CCP antibodies: the past, the present and the future. Nat Rev Rheumatol 7:391–398. https://doi.org/10.1038/nrrheum.2011.76
Schellekens GA, Visser H, de Jong BA, van den Hoogen FH, Hazes JM, Breedveld FC, van Venrooij WJ (2000) The diagnostic properties of rheumatoid arthritis antibodies recognizing a cyclic citrullinated peptide. Arthritis Rheum 43:155–163. https://doi.org/10.1002/1529-0131(200001)43:1<155::Aid-anr20>3.0.Co;2-3
Kroot EJ, de Jong BA, van Leeuwen MA, Swinkels H, van den Hoogen FH, van't Hof M, van de Putte LB, van Rijswijk MH, van Venrooij WJ, van Riel PL (2000) The prognostic value of anti-cyclic citrullinated peptide antibody in patients with recent-onset rheumatoid arthritis. Arthritis Rheum 43:1831–1835. https://doi.org/10.1002/1529-0131(200008)43:8<1831::Aid-anr19>3.0.Co;2-6
Syversen SW, Gaarder PI, Goll GL, Odegard S, Haavardsholm EA, Mowinckel P, van der Heijde D, Landewe R, Kvien TK (2008) High anti-cyclic citrullinated peptide levels and an algorithm of four variables predict radiographic progression in patients with rheumatoid arthritis: results from a 10-year longitudinal study. Ann Rheum Dis 67:212–217. https://doi.org/10.1136/ard.2006.068247
Hetland ML, Stengaard-Pedersen K, Junker P, Ostergaard M, Ejbjerg BJ, Jacobsen S, Lottenburger T, Hansen I, Tarp U, Andersen LS, Svendsen A, Pedersen JK, Lauridsen UB, Ellingsen T, Lindegaard H, Podenphant J, Vestergaard A, Jurik AG, Horslev-Petersen K (2010) Radiographic progression and remission rates in early rheumatoid arthritis - MRI bone oedema and anti-CCP predicted radiographic progression in the 5-year extension of the double-blind randomised CIMESTRA trial. Ann Rheum Dis 69:1789–1795. https://doi.org/10.1136/ard.2009.125534
Hecht C, Englbrecht M, Rech J, Schmidt S, Araujo E, Engelke K, Finzel S, Schett G (2015) Additive effect of anti-citrullinated protein antibodies and rheumatoid factor on bone erosions in patients with RA. Ann Rheum Dis 74:2151–2156. https://doi.org/10.1136/annrheumdis-2014-205428
Jilani AA, Mackworth-Young CG (2015) The role of citrullinated protein antibodies in predicting erosive disease in rheumatoid arthritis: a systematic literature review and meta-analysis. Int J Rheumatol 2015:728610–728618. https://doi.org/10.1155/2015/728610
Berglin E, Johansson T, Sundin U, Jidell E, Wadell G, Hallmans G, Rantapaa-Dahlqvist S (2006) Radiological outcome in rheumatoid arthritis is predicted by presence of antibodies against cyclic citrullinated peptide before and at disease onset, and by IgA-RF at disease onset. Ann Rheum Dis 65:453–458. https://doi.org/10.1136/ard.2005.041376
Kuhn KA, Kulik L, Tomooka B, Braschler KJ, Arend WP, Robinson WH, Holers VM (2006) Antibodies against citrullinated proteins enhance tissue injury in experimental autoimmune arthritis. J Clin Invest 116:961–973. https://doi.org/10.1172/jci25422
Ge C, Tong D, Liang B, Lonnblom E, Schneider N, Hagert C, Viljanen J, Ayoglu B, Stawikowska R, Nilsson P, Fields GB, Skogh T, Kastbom A, Kihlberg J, Burkhardt H, Dobritzsch D, Holmdahl R (2017) Anti-citrullinated protein antibodies cause arthritis by cross-reactivity to joint cartilage. JCI Insight 2. https://doi.org/10.1172/jci.insight.93688
England BR, Thiele GM, Mikuls TR (2017) Anticitrullinated protein antibodies: origin and role in the pathogenesis of rheumatoid arthritis. Curr Opin Rheumatol 29:57–64. https://doi.org/10.1097/bor.0000000000000356
Chatzidionysiou K, Lie E, Nasonov E, Lukina G, Hetland ML, Tarp U, Gabay C, van Riel PL, Nordstrom DC, Gomez-Reino J, Pavelka K, Tomsic M, Kvien TK, van Vollenhoven RF (2011) Highest clinical effectiveness of rituximab in autoantibody-positive patients with rheumatoid arthritis and in those for whom no more than one previous TNF antagonist has failed: pooled data from 10 European registries. Ann Rheum Dis 70:1575–1580. https://doi.org/10.1136/ard.2010.148759
Alessandri C, Bombardieri M, Papa N, Cinquini M, Magrini L, Tincani A, Valesini G (2004) Decrease of anti-cyclic citrullinated peptide antibodies and rheumatoid factor following anti-TNFalpha therapy (infliximab) in rheumatoid arthritis is associated with clinical improvement. Ann Rheum Dis 63:1218–1221. https://doi.org/10.1136/ard.2003.014647
Atzeni F, Sarzi-Puttini P, Dell’ Acqua D, de Portu S, Cecchini G, Cruini C, Carrabba M, Meroni PL (2006) Adalimumab clinical efficacy is associated with rheumatoid factor and anti-cyclic citrullinated peptide antibody titer reduction: a one-year prospective study. Arthritis Res Ther 8:R3. https://doi.org/10.1186/ar1851
Bos WH, Bartelds GM, Wolbink GJ, de Koning MH, van de Stadt RJ, van Schaardenburg D, Dijkmans BA, Nurmohamed MT (2008) Differential response of the rheumatoid factor and anticitrullinated protein antibodies during adalimumab treatment in patients with rheumatoid arthritis. J Rheumatol 35:1972–1977
Chen HA, Lin KC, Chen CH, Liao HT, Wang HP, Chang HN, Tsai CY, Chou CT (2006) The effect of etanercept on anti-cyclic citrullinated peptide antibodies and rheumatoid factor in patients with rheumatoid arthritis. Ann Rheum Dis 65:35–39. https://doi.org/10.1136/ard.2005.038851
Iannone F, Tampoia M, Giannini M, Lopalco G, Cantarini L, Villalta CD, Galeazzi M, Lapadula G (2016) Changes in anti-cyclic citrullinated peptide antibodies and rheumatoid factor isotypes serum levels in patients with rheumatoid arthritis following treatment with different biological drugs. Clin Exp Rheumatol 34:424–429
Bruns A, Nicaise-Roland P, Hayem G, Palazzo E, Dieude P, Grootenboer-Mignot S, Chollet-Martin S, Meyer O (2009) Prospective cohort study of effects of infliximab on rheumatoid factor, anti-cyclic citrullinated peptide antibodies and antinuclear antibodies in patients with long-standing rheumatoid arthritis. Joint Bone Spine 76:248–253. https://doi.org/10.1016/j.jbspin.2008.09.010
Mikuls TR, O'Dell JR, Stoner JA, Parrish LA, Arend WP, Norris JM, Holers VM (2004) Association of rheumatoid arthritis treatment response and disease duration with declines in serum levels of IgM rheumatoid factor and anti-cyclic citrullinated peptide antibody. Arthritis Rheum 50:3776–3782. https://doi.org/10.1002/art.20659
Guzian MC, Carrier N, Cossette P, de Brum-Fernandes AJ, Liang P, Menard HA, Boire G (2010) Outcomes in recent-onset inflammatory polyarthritis differ according to initial titers, persistence over time, and specificity of the autoantibodies. Arthritis Care Res 62:1624–1632. https://doi.org/10.1002/acr.20288
Kastbom A, Forslind K, Ernestam S, Geborek P, Karlsson JA, Petersson IF, Saevarsdottir S, Klareskog L, van Vollenhoven RF, Lundberg K (2016) Changes in the anticitrullinated peptide antibody response in relation to therapeutic outcome in early rheumatoid arthritis: results from the SWEFOT trial. Ann Rheum Dis 75:356–361. https://doi.org/10.1136/annrheumdis-2014-205698
Cambridge G, Leandro MJ, Edwards JC, Ehrenstein MR, Salden M, Bodman-Smith M, Webster AD (2003) Serologic changes following B lymphocyte depletion therapy for rheumatoid arthritis. Arthritis Rheum 48:2146–2154. https://doi.org/10.1002/art.11181
Bohler C, Radner H, Smolen JS, Aletaha D (2013) Serological changes in the course of traditional and biological disease modifying therapy of rheumatoid arthritis. Ann Rheum Dis 72:241–244. https://doi.org/10.1136/annrheumdis-2012-202297
Matthews JN, Altman DG, Campbell MJ, Royston P (1990) Analysis of serial measurements in medical research. BMJ 300:230–235
van der Heijde D (2000) How to read radiographs according to the Sharp/van der Heijde method. J Rheumatol 27:261–263
Lee DK (2016) Alternatives to P value: confidence interval and effect size. Korean J Anesthesiol 69:555–562. https://doi.org/10.4097/kjae.2016.69.6.555
Courvoisier N, Dougados M, Cantagrel A, Goupille P, Meyer O, Sibilia J, Daures JP, Combe B (2008) Prognostic factors of 10-year radiographic outcome in early rheumatoid arthritis: a prospective study. Arthritis Res Ther 10:R106. https://doi.org/10.1186/ar2498
Joo YB, Bang SY, Ryu JA, Lee S, Lee HS, Bae SC (2017) Predictors of severe radiographic progression in patients with early rheumatoid arthritis: a prospective observational cohort study. Int J Rheum Dis 20:1437–1446. https://doi.org/10.1111/1756-185x.13054
Rydell E, Forslind K, Nilsson JA, Jacobsson LTH, Turesson C (2018) Smoking, body mass index, disease activity, and the risk of rapid radiographic progression in patients with early rheumatoid arthritis. Arthritis Res Ther 20:82. https://doi.org/10.1186/s13075-018-1575-2
Odegard S, Landewe R, van der Heijde D, Kvien TK, Mowinckel P, Uhlig T (2006) Association of early radiographic damage with impaired physical function in rheumatoid arthritis: a ten-year, longitudinal observational study in 238 patients. Arthritis Rheum 54:68–75. https://doi.org/10.1002/art.21548
Scott DL, Pugner K, Kaarela K, Doyle DV, Woolf A, Holmes J, Hieke K (2000) The links between joint damage and disability in rheumatoid arthritis. Rheumatology (Oxford) 39:122–132
Welsing PM, Landewe RB, van Riel PL, Boers M, van Gestel AM, van der Linden S, Swinkels HL, van der Heijde DM (2004) The relationship between disease activity and radiologic progression in patients with rheumatoid arthritis: a longitudinal analysis. Arthritis Rheum 50:2082–2093. https://doi.org/10.1002/art.20350
Tsuji H, Yano K, Furu M, Yamakawa N, Ikari K, Hashimoto M, Ito H, Fujii T, Yamamoto W, Ohmura K, Taniguchi A, Momohara S, Matsuda F, Allaart CF, Yamanaka H, Mimori T, Terao C (2017) Time-averaged disease activity fits better joint destruction in rheumatoid arthritis. Sci Rep 7:5856. https://doi.org/10.1038/s41598-017-05581-w
Aletaha D, Alasti F, Smolen JS (2013) Rheumatoid factor determines structural progression of rheumatoid arthritis dependent and independent of disease activity. Ann Rheum Dis 72:875–880. https://doi.org/10.1136/annrheumdis-2012-201517
Choi HK, Nguyen US, Niu J, Danaei G, Zhang Y (2014) Selection bias in rheumatic disease research. Nat Rev Rheumatol 10:403–412. https://doi.org/10.1038/nrrheum.2014.36
Ruscitti P, Ursini F, Cipriani P, Liakouli V, Carubbi F, Berardicurti O, De Sarro G, Giacomelli R (2017) Poor clinical response in rheumatoid arthritis is the main risk factor for diabetes development in the short-term: a 1-year, single-Centre, longitudinal study. PLoS One 12:e0181203. https://doi.org/10.1371/journal.pone.0181203
Jonsson MK, Hensvold AH, Hansson M, Aga AB, Sexton J, Mathsson-Alm L, Cornillet M, Serre G, Lillegraven S, Fevang BS, Catrina AI, Haavardsholm EA (2018) The role of anti-citrullinated protein antibody reactivities in an inception cohort of patients with rheumatoid arthritis receiving treat-to-target therapy. Arthritis Res Ther 20:146. https://doi.org/10.1186/s13075-018-1635-7
Cohen G, Gossec L, Dougados M, Cantagrel A, Goupille P, Daures JP, Rincheval N, Combe B (2007) Radiological damage in patients with rheumatoid arthritis on sustained remission. Ann Rheum Dis 66:358–363. https://doi.org/10.1136/ard.2006.057497
Koga T, Okada A, Fukuda T, Hidaka T, Ishii T, Ueki Y, Kodera T, Nakashima M, Takahashi Y, Honda S, Horai Y, Watanabe R, Okuno H, Aramaki T, Izumiyama T, Takai O, Miyashita T, Sato S, Kawashiri SY, Iwamoto N, Ichinose K, Tamai M, Origuchi T, Nakamura H, Aoyagi K, Eguchi K, Kawakami A (2017) Anti-citrullinated peptide antibodies are the strongest predictor of clinically relevant radiographic progression in rheumatoid arthritis patients achieving remission or low disease activity: a post hoc analysis of a nationwide cohort in Japan. PLoS One 12:e0175281. https://doi.org/10.1371/journal.pone.0175281
Ronnelid J, Wick MC, Lampa J, Lindblad S, Nordmark B, Klareskog L, van Vollenhoven RF (2005) Longitudinal analysis of citrullinated protein/peptide antibodies (anti-CP) during 5 year follow up in early rheumatoid arthritis: anti-CP status predicts worse disease activity and greater radiological progression. Ann Rheum Dis 64:1744–1749. https://doi.org/10.1136/ard.2004.033571
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
The study was carried out in accordance with the Helsinki Declaration and approved by the Institutional Review Board of St. Vincent’s Hospital, the Catholic University of Korea (No. VC18RESI0136)
Conflict of interest
The authors declare that there are no conflicts of interest.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Joo, Y.B., Park, YJ., Park, KS. et al. Association of cumulative anti-cyclic citrullinated protein antibodies with radiographic progression in patients with rheumatoid arthritis. Clin Rheumatol 38, 2423–2432 (2019). https://doi.org/10.1007/s10067-019-04554-w
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10067-019-04554-w