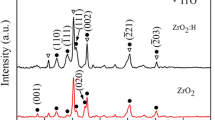
Abstract
Zn-Ti-O composite thin film prepared on FTO by sol-gel technique is discovered presenting electrochromic behavior in electrolytes with Li+ ion (LiClO4) and K+ (KCl) as well. The observed EC colored/bleached switching is electrolyte dependent which is blue-green/transparent in LiClO4 and gray-blue/transparent in KCl, respectively. Accordantly, the respective most optical modulation (ΔT) between colored and bleached states is ~ 32% (710 nm) in LiClO4 and ~ 37% (600 nm) in KCl. The finding of appreciable steady EC durability with appealing visual optical contrast (ΔT) in conventionally fabricated Zn-Ti-O thin film using with bigger/heavier K+ electrolyte helps expanding the applicable components in EC device.





Similar content being viewed by others
References
Mishra S, Pandey H, Yogi P, Saxena SK, Roy S, Sagdeo PR, Kumar R (2017) Interfacial redox centers as origin of color switching in organic electrochromic device. Opt Mater 66:65–71
Zhang S, Sun G, He Y, Fu R, Gu Y, Chen S (2017) Preparation, characterization, and electrochromic properties of nanocellulose-based polyaniline nanocomposite films. ACS Appl Mater Interfaces 9(19):16426–16434
Falahatgar SS, Ghodsi FE, Tepehan FZ, Tepehan GG, Turhan İ, Pishdadian S (2015) Electrochromic performance of sol-gel derived amorphous MnO2-ZnO nanogranular composite thin films. J Non-Cryst Solids 427:1–9
Fu X, Jia C, Wan Z, Weng X, Xie J, Deng L (2014) Hybrid electrochromic film based on polyaniline and TiO2 nanorods array. Org Electron 15(11):2702–2709
Zhang W, Ju W, Wu X, Wang Y, Wang Q, Zhou H, Wang S, Hu C (2016) Structure, stability and electrochromic properties of polyaniline film covalently bonded to indium tin oxide substrate. Appl Surf Sci 367:542–551
Sorar I, Pehlivan E, Niklasson GA, Granqvist CG (2013) Electrochromism of DC magnetron sputtered TiO2 thin films: role of deposition parameters. Sol Energy Mater Sol Cells 115:172–180
Sun XW, Wang JX (2008) Fast switching electrochromic display using a viologen-modified ZnO nanowire array electrode. Nano Lett 8(7):1884–1889
Wen RT, Granqvist CG, Niklasson GA (2015) Eliminating degradation and uncovering ion-trapping dynamics in electrochromic WO3 thin films. Nat Mater 4:996–1001
Coskun OD, Demirel S, Atak G (2015) The effects of heat treatment on optical, structural, electrochromic and bonding properties of Nb2O5 thin films. J Alloys Compd 648:994–1004
Reyes-Gil KR, Stephens ZD, Stavila V, Robinson DB (2015) Composite WO3 / TiO2 nanostructures for high electrochromic activity. ACS Appl Mater Interfaces 7(4):2202–2213
Bo G, Wang X, Wang K, Gao R, Dong B, Cao L, Ge S (2017) Preparation and electrochromic performance of NiO/TiO2 nanorod composite film. J Alloys Compd 728:878–886
Barawi M, Trizio LD, Giannuzzi R, Veramonti G, Manna L, Monca M (2017) Dual band electrochromic devices based on Nb-doped TiO2 nanocrystalline electrodes. ACS Nano 11(4):3576–3584
Bi Z, Zhang S, Xu X, Hu X, Li X, Gao X (2015) A novel nanocomposite of WO3 modified Al-doped ZnO nanowires with enhanced electrochromic performance. Mater Lett 160:186–189
Bi Z, Li X, Chen Y, Xu X, Zhang S, Zhu Q (2017) Bi-functional flexible electrodes based on tungsten trioxide/zinc oxide nanocomposites for electrochromic and energy storage applications. Electrochim Acta 227:61–68
Liu X, Zhou A, Dou Y, Pan T, Shao M, Han J, Wei M (2015) Ultrafast switching of an electrochromic device based on layered double hydroxide/Prussian blue multilayered films. Nanoscale 7(40):17088–17095
Kitao M, Oshima Y, Urabe K (1997) Preparation and Electrochromism of RF-sputtered TiO2 films. Jpn J Appl Phys 36:4423–4426
Patel KJ, Panchal CJ, Desai MS, Mehta PK (2010) An investigation of the insertion of the cations H+, Na+, K+ on the electrochromic properties of the thermally evaporated WO3 thin films grown at different substrate temperatures. Mater Chem Phys 124(1):884–890
Yang MH, Chen TT, Wang YS, Chiu HT, Lee CY (2011) Electrochromism of rutile nanowires, vertically aligned along the [001] direction, due to alkali metal ion intercalation. J Mater Chem 21(46):18738–18743
Velevska J, Ristova M (2002) Electrochromic properties of NiOx prepared by low vacuum evaporation. Sol Energy Mater Sol Cells 73(2):131–139
Hanzu I, Hornebecq V, Djenizian T, Knauth P (2013) In situ study of electrochromic properties of self-assembled TiO2 nanotubes. CR Chim 16(1):96–102
Sivakumar R, Manisankar P, Jayachandran M, Sanjeeviraja C (2006) Intercalation studies on electron beam evaporated MoO3 films for electrochemical devices. Sol Energy Mater Sol Cells 90(15):2438–2448
Yoshimura K, Miki T, Tanemura S (1997) TiO2 electrochromic thin films by reactive direct current magnetron sputtering. J Vac Sci Technol A 15(5):2673–2676
Eren E, Alver C, Karaca GY, Uygun E, Oksuz AU (2018) Enhanced electrochromic performance of WO3 hybrids using polymer plasma hybridization process. Synth Met 235:115–124
Sahu DR, Wu TJ, Wang SC, Huang JL (2017) Electrochromic behavior of NiO film prepared by e-beam evaporation. J Sci Adv Mater Devices 2(2):225–232
Vijayalakshmi R, Jayachandran M, Trivedi DC, Sanjeeviraja C (2004) Study of the potassium ion insertion of the electrodeposited electrochromic tungsten trioxide thin films. Ionics 10(1–2):151–154
Lee KD (1997) Deposition of WO3 thin films by the sol-gel method. Thin Solid Films 302(1-2):84–88
Sivakumar R, Shanthakumari K, Thayumanavan A, Jayachandran M, Sanjeeviraja C (2007) Coloration and bleaching mechanism of tungsten oxide thin films in different electrolytes. Surf Eng 23(5):373–379
Acknowledgements
The authors are grateful for XRD characterization work assisted by Laboratory for Microstructures of Shanghai University.
Funding
This work was supported by the Shandong Jinjing Technology Co., Ltd. and Shanghai Committee of Science and Technology under Grant No. 15JC1400303.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Shi, J., Guo, R. & Yu, S. Electrochromic performance of Zn-Ti-O composite thin film with electrolyte dependence. J Solid State Electrochem 22, 3815–3820 (2018). https://doi.org/10.1007/s10008-018-4090-0
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10008-018-4090-0