Abstract
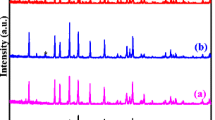
In the present study, Li x AlZr[PO4]3 (x = 1.8, 2.0, 2.2) has been prepared by the Pechini-type polymerizable precursor method, and structural and electrical analyses have been performed. Formation of the compound has been confirmed from X-ray diffraction pattern. Vibrational analysis has been performed using Fourier transform infrared spectroscopy. Ion transport mechanism has been analyzed using the AC impedance technique. Electrical characterization reveals that the maximum conductivity (4.4 × 10−5S cm−1) has been observed for the sample Li2.2AlZr[PO4]3 at 673K.



Similar content being viewed by others
References
Forsyth M, Wong S, Narin K, Best AS (1999) Solid State Ion 124:213
Miguel París A, Ana Martínez-Juárez , José Rojov M, Jesús Sanz (1996) J Phys Chem 8:5355
Aono H, Sugimoto E, Sadaoka Y (1993) J Electrochem Soc 140:1827
Miguel París A, Jesús Sanz A (2000) Phys Rev B 62(2):810
Aono H, Sugimoto E, Sadaoka Y (1989) J Electrochem Soc 136:590
Forsyth M, Wong S, Nairn KM, Best AS (1999) Solid State Ion 124:213
Tranter GE, Holmes JL, Lindon JC (eds) (2000) In: Encyclopedia of spectroscopy and spectrometry. vol. 2. Academic, New York, pp 1030
Ravikumar RVSSN, Chandrasekhar AV, Reddy BJ, Reddy YP, Ikeda K (2002) Cryst Res Technol 37(10):1127
Weiwei Huang, Roger Frech (1996) Solid State Ion 86–88:395
Bernard Boukamp A (1986) Solid State Ion 18–19:136
Ryoji Kanno A, Takayuki Hata A, Yoji Kawamoto A, Michihiko Irie A (2000) Solid State Ion 130:97
Miguel París A, Ana Martínez-Juárez, José Rojo M (1996) J Phys Chem 8:5355
Author information
Authors and Affiliations
Corresponding author
Additional information
Contribution to ICMAT 2007, Symposium K: Nanostructured and bulk materials for electrochemical power sources, July 1–6, 2007, Singapore.
Rights and permissions
About this article
Cite this article
Savitha, T., Selvasekarapandian, S. & Ramya, C.S. Structural and electrical conductivity studies of Li x AlZr[PO4]3 (x = 1.8, 2.0, 2.2), solid electrolyte for lithium-rechargeable batteries. J Solid State Electrochem 12, 857–860 (2008). https://doi.org/10.1007/s10008-008-0520-8
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10008-008-0520-8