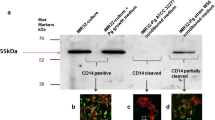
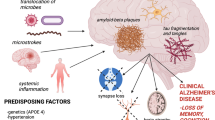
Abstract
Objectives
This study aimed to analyze the following PICO question: Are animals infected with Porphyromonas gingivalis (P. gingivalis) or bacterial lipopolysaccharide (Pg-LPS) more affected by neurodegeneration, similar to the pathogenesis generated by Alzheimer’s disease (AD), compared with non-infected animals?
Methods
Databases PubMed, Lilacs, SciELO, Science Direct, Scopus, Web of Science, and Cochrane were searched for pre-clinical in vivo studies in which mice were infected with P. gingivalis or received Pg-LPS, in order to assess the brain tissue and cognitive impairment. No limit for date or publication language was imposed and this study was registered at the International Prospective Register of Systematic Reviews (PROSPERO), with nine articles included. Syrcle’s protocol was used to evaluate bias in the selected studies.
Results
Nine articles were included. Infection by P. gingivalis or the administration of Pg-LPS increased the production of the inflammatory mediators, TNF-α (tumor necrosis factor-alpha), IL-6 (interleukin-6), and IL-1β (interleukin-1beta), augmented Aβ (amyloid beta) production, and activated the complement system, causing inflammation, brain tissue degeneration, and cognitive impairment, consistent with the damage in AD.
Conclusions
Infection by P. gingivalis and Pg-LPS administration appears to be in relation with the pathogenesis of AD by activating the complement cascade, increasing Aβ production and augmenting pro-inflammatory cytokine expression, causing age-dependent brain inflammation, neuroinflammation, and neurodegeneration.
Clinical relevance
Taking into account the importance of holistic treatment in the dental office, this study focuses on identifying highly prevalent oral diseases, such as periodontal disease, as risk factors for the aggravation of degenerative diseases in the elderly population.

Similar content being viewed by others
References
Nazir M, Al-Ansari A, Al-Khalifa K, Alhareky M, Gaffar B, Almas K (2020) Global prevalence of periodontal disease and lack of its surveillance. TheScientificWorldJournal 2020:2146160–2146168. https://doi.org/10.1155/2020/2146160
Thomas JT, Thomas T, Ahmed M, Kannan SK, Abdullah Z, Alghamdi SA, Joseph B (2020) Prevalence of periodontal disease among obese young adult population in Saudi Arabia-a cross-sectional study. Medicina (Kaunas, Lithuania) 56(4):197. https://doi.org/10.3390/medicina56040197
Fiorillo L, Cervino G, Laino L, D’Amico C, Mauceri R, Tozum TF, Gaeta M, Cicciù M (2019) Porphyromonas gingivalis, periodontal and systemic implications: a systematic review. Dentist J 7(4):114. https://doi.org/10.3390/dj7040114
Gao L, Kang M, Zhang MJ, Reza Sailani M, Kuraji R, Martinez A, Ye C, Kamarajan P, Le C, Zhan L, Rangé H, Ho SP, Kapila YL (2020) Polymicrobial periodontal disease triggers a wide radius of effect and unique virome. NPJ Biofilms Microbiomes 6(1):10. https://doi.org/10.1038/s41522-020-0120-7
Offenbacher S, Katz V, Fertik G, Collins J, Boyd D, Maynor G, McKaig R, Beck J (1996) Periodontal infection as a possible risk factor for preterm low birth weight. J Periodontol 67(10 Suppl):1103–1113. https://doi.org/10.1902/jop.1996.67.10s.1103
Ishida N, Ishihara Y, Ishida K, Tada H, Funaki-Kato Y, Hagiwara M, Ferdous T, Abdullah M, Mitani A, Michikawa M, Matsushita K (2017) Periodontitis induced by bacterial infection exacerbates features of Alzheimer’s disease in transgenic mice. NPJ Aging Mech Dis 3:15. https://doi.org/10.1038/s41514-017-0015-x
Ding Y, Ren J, Yu H, Yu W, Zhou Y (2018) Porphyromonas gingivalis, a periodontitis causing bacterium, induces memory impairment and age-dependent neuroinflammation in mice. Immun Ageing 15:1. https://doi.org/10.1186/s12979-017-0110-7
Poole S, Singhrao SK, Chukkapalli S, Rivera M, Velsko I, Kesavalu L, Crean S (2015) Active invasion of Porphyromonas gingivalis and infection-induced complement activation in ApoE-/- mice brains. J Alzheimers Dis 43(1):67–80. https://doi.org/10.3233/JAD-140315
Liu Y, Wu Z, Nakanishi Y, Ni J, Hayashi Y, Takayama F, Zhou Y, Kadowaki T, Nakanishi H (2017) Infection of microglia with Porphyromonas gingivalis promotes cell migration and an inflammatory response through the gingipain-mediated activation of protease-activated receptor-2 in mice. Sci Rep 7(1):11759. https://doi.org/10.1038/s41598-018-27508-9
Wu Z, Ni J, Liu Y, Teeling JL, Takayama F, Collcutt A, Ibbett P, Nakanishi H (2017) Cathepsin B plays a critical role in inducing Alzheimer’s diseaselike phenotypes following chronic systemic exposure to lipopolysaccharide from Porphyromonas gingivalis in mice. Brain Behav Immun 65:350–361. https://doi.org/10.1016/j.bbi.2017.06.002
Ilievski V, Zuchowska PK, Green SJ, Toth PT, Ragozzino ME, Le K, O’Brien-Simpson HNM, Reynolds EC, Watanabe K (2018) Chronic oral application of periodontal pathogen results in brain inflammation, neurodegeneration and amyloid beta production in wild type mice. PLoS One 13(10):e0204941. https://doi.org/10.1371/journal.pone.0204941
Dominy SS, Lynch C, Ermini F, Benedyk M, Marczyk A, Konradi A, Nguyen M, Haditsch U, Raha D, Griffin C, Holsinger LJ, Arastu-Kapur S, Kaba S, Lee A, Ryder MI, Potempa B, Mydel P, Hellvard A, Adamowicz K, Hasturk H, Walker GD, Reynolds EC, Faull RLM, Curtis MA, Dragunow M, Potempa J (2019) Porphyromonas gingivalis in Alzheimer’s disease brains: evidence for disease causation and treatment with small-molecule inhibitors. Sci Adv 5(1):eaau3333. https://doi.org/10.1126/sciadv.aau3333
Hayashi K, Hasegawa Y, Takemoto Y, Cao C, Takeya H, Komohara Y, Mukasa A, Kim-Mitsuyama S (2019) Continuous intracerebroventricular injection of Porphyromonas gingivalis lipopolysaccharide induces systemic organ dysfunction in a mouse model of Alzheimer’s disease. Exp Gerontol 120:1–5. https://doi.org/10.1016/j.exger.2019.02.007
Nie R, Wu Z, Ni J, Zeng F, Yu W, Zhang Y, Kadowaki T, Kashiwazaki H, Teeling JL, Zhou Y (2019) Porphyromonas gingivalis infection induces amyloid-β accumulation in monocytes/macrophages. J Alzheimer’s Dis 72(2):479–494. https://doi.org/10.3233/JAD-190298
Sochocka M, Zwolińska K, Leszek J (2017) The infectious etiology of Alzheimer’s Disease. Curr Neuropharmacol 15(7):996–1009. https://doi.org/10.2174/1570159X15666170313122937
Association A (2014) Alzheimer’s disease facts and figures. Alzheimers Dement 10(2):e47–e92. https://doi.org/10.1016/j.jalz.2014.02.001
Dioguardi M, Crincoli V, Laino L, Alovisi M, Sovereto D, Mastrangelo F, Russo LL, Muzio LL (2020) The role of periodontitis and periodontal bacteria in the onset and progression of Alzheimer’s disease: a systematic review. J Clin Med 9(2):495. https://doi.org/10.3390/jcm9020495
Nadim R, Tang J, Dilmohamed A, Yuan S, Wu C, Bakre AT, Partridge M, Ni J, Copeland JR, Anstey KJ, Chen R (2020) Influence of periodontal disease on risk of dementia: a systematic literature review and a meta-analysis. Eur J Epidemiol. https://doi.org/10.1007/s10654-020-00648-x
Ming Y, Hsu SW, Yen YY, Lan SJ (2019) Association of oral health-related quality of life and Alzheimer disease: a systematic review. J Prosthet Dent S0022-3913(19):30545–30541. https://doi.org/10.1016/j.prosdent.2019.08.015
Moher D, Shamseer L, Clarke M, Ghersi D, Liberati A, Petticrew M, Shekelle P, Stewart LA, PRISMA-P Group (2015) Preferred reporting items for systematic review and meta-analysis protocols (PRISMA-P) 2015 statement. Syst Rev 4(1):1. https://doi.org/10.1186/2046-4053-4-1
Hooijmans CR, Rovers MM, de Vries RB, Leenaars M, Ritskes-Hoitinga M, Langendam MW (2014) SYRCLE’s risk of bias tool for animal studies. BMC Med Res Methodol 14:43. https://doi.org/10.1186/1471-2288-14-43
Matsushita K, Yamada-Furukawa M, Kurosawa M, Shikama Y (2020) Periodontal disease and periodontal disease-related bacteria involved in the pathogenesis of Alzheimer’s disease. J Inflamm Res 13:275–283. https://doi.org/10.2147/JIR.S255309
Ryder MI (2020) Porphyromonas gingivalis and Alzheimer’s disease: recent findings and potential therapies. J Periodontol 91. https://doi.org/10.1002/JPER.20-0104
Sheppard O, Coleman MP, Durrant CS (2019) Lipopolysaccharide-induced neuroinflammation induces presynaptic disruption through a direct action on brain tissue involving microglia-derived interleukin 1 beta. J Neuroinflammation 16(1):106. https://doi.org/10.1186/s12974-019-1490-8
Beydoun MA, Beydoun HA, Weiss J, Hossain S, El-Hajj ZW, Zonderman AB (2020) Helicobacter pylori, periodontal pathogens, and their interactive association with incident all-cause and Alzheimer’s disease dementia in a large national survey. Mol Psychiatry. https://doi.org/10.1038/s41380-020-0736-2
Panzarella V, Mauceri R, Baschi R, Maniscalco L, Campisi G, Monastero R (2020) Oral Health status in subjects with amnestic mild cognitive impairment and Alzheimer’s disease: data from the Zabút Aging Project. J Alzheimers Dis:1–11. https://doi.org/10.3233/JAD-200385
Siddaway AP, Wood AM, Hedges LV (2019) How to do a systematic review: a best practice guide for conducting and reporting narrative reviews, meta-analyses, and meta-syntheses. Annu Rev Psychol 70:747–770. https://doi.org/10.1146/annurev-psych-010418-102803
Miguel-Álvarez M, Santos-Lozano A, Sanchis-Gomar F, Fiuza-Luces C, Pareja-Galeano H, Garatachea N, Lucia A (2015) Non-steroidal anti-inflammatory drugs as a treatment for Alzheimer’s disease: a systematic review and meta-analysis of treatment effect. Drugs Aging 32(2):139–147. https://doi.org/10.1007/s40266-015-0239-z
Funding
This work was supported by the Post-graduate Program in Dental Sciences, Federal University of Rio Grande do Norte (Brazil), and also by the Coordination for the Improvement of Higher-Level Personnel (CAPES/Brazil).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Ethical approval
This work follows the entire protocol prepared by PRISMA for the elaboration of systematic reviews. Since the study was secondary data and did not involve humans, it did not require submission to an ethics committee. Nevertheless, all authors declare that they followed a well elaborated protocol to minimize conflicts or possible biases in the elaboration of the manuscript.
Informed consent
No informed consent was required for this type of study.
Conflict of interest
All the authors involved in the elaboration of this manuscript declare there is no conflict of interest, be it ethical, moral or financial, with any of the information or magazines cited in the elaboration of this manuscript. Moan Jéfter Fernandes Costa declares that it knows all the steps taken, has actively participated in the production of the manuscript, and has no conflict of interest. Isabela Dantas Torres de Araújo declares that she knows all the steps taken, has actively participated in the production of the manuscript, and has no conflict of interest. Luana da Rocha Alves declares that she is aware of all the steps taken, has actively participated in the production of the manuscript, and has no conflict of interest. Romerito Lins da Silva declares that he knows all the steps taken, has actively participated in the production of the manuscript, and has no conflict of interest. Patricia dos Santos Calderon declares that she knows all the steps taken, has actively participated in the production of the manuscript, and has no conflict of interest. Boniek Castillo Dutra Borges declares that he knows all the steps taken, has actively participated in the production of the manuscript, and has no conflict of interest. Ana Rafaela Luz de Aquino Martins declares that she knows all the steps taken, has actively participated in the production of the manuscript, and has no conflict of interest. Bruno Cesar de Vasconcelos Gurgel declares that he knows all the steps taken, has actively participated in the production of the manuscript, and has no conflict of interest. Ruthineia Diogenes Alves Uchoa Lins declares that she knows all the steps taken, has actively participated in the production of the manuscript, and has no conflict of interest.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Costa, M.J.F., de Araújo, I.D.T., da Rocha Alves, L. et al. Relationship of Porphyromonas gingivalis and Alzheimer’s disease: a systematic review of pre-clinical studies. Clin Oral Invest 25, 797–806 (2021). https://doi.org/10.1007/s00784-020-03764-w
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00784-020-03764-w