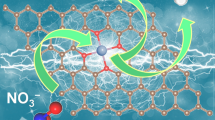
Abstract
Two biologically inspired tetranuclear nickel complexes [Ni4(L–H)4(CH3COO)3]·Cl (1) and [Ni4(L–H)4(CH3COO)4]·2CH3OH (2) (L = di(pyridin-2-yl)methanediol) have been synthesized and investigated by a combination of X-ray crystallography, PXRD, electrochemistry, in-situ UV–Vis spectroelectrochemistry and DLS. Both of the two complexes feature a core composed of four Ni(II) ions with the same peripheral ligation provided by the anionic di(pyridin-2-yl)methanediol and MeCOO− ligands. Whereas, complex 1 possesses one distorted cubane-like [Ni4(µ3-O)4] core, while 2 has one extended butterfly-like [Ni4(µ3-O)2] core. The homogeneous electrocatalytic reactivity of the two water-soluble complexes for water oxidation have been thoroughly studied, which demonstrates that both of them can efficiently electrocatalyze water oxidation with high stability under alkaline conditions, at relatively low over-potentials (η) of 420–790 mV for 1 and 390–780 mV for 2, both in the pH range of 7.67–12.32, with the high TOF of about 139 s−1 (1) and 69 s−1 (2) at pH = 12.32, respectively. By a series of comparative experiments for complexes 1 and 2, we proposed that their crystal geometries play an important role in their electrocatalytic reactivity for water oxidation. We verified that biomimetic cubane geometry could promote OER catalysis with two very similar compounds for the first time.
Graphic abstract
Compared with 2, the biomimetic cubane topology of 1 could promote OER catalysis by facilitating efficient charge delocalization and electron-transfer.









Similar content being viewed by others
References
Zhang W, Lai W, Cao R (2017) Energy-related small molecule activation reactions: oxygen reduction and hydrogen and oxygen evolution reactions catalyzed by porphyrin- and corrole-based systems. Chem Rev 117(4):3717–3797. https://doi.org/10.1021/acs.chemrev.6b00299
Okabe Y, Lee SK, Kondo M, Masaoka S (2017) Syntheses and CO2 reduction activities of π-expanded/extended iron porphyrin complexes. J Biol Inorg Chem 22(5):713–725. https://doi.org/10.1007/s00775-017-1438-3
Amanullah S, Dey A (2019) A bi-functional cobalt-porphyrinoid electrocatalyst: balance between overpotential and selectivity. J Biol Inorg Chem 24(4):437–442. https://doi.org/10.1007/s00775-019-01670-5
Izumi Y (2013) Recent advances in the photocatalytic conversion of carbon dioxide to fuels with water and/or hydrogen using solar energy and beyond. Coord Chem Rev 257(1):171–186. https://doi.org/10.1016/j.ccr.2012.04.018
Han Q, Ding Y (2018) Recent advances in the field of light-driven water oxidation catalyzed by transition-metal substituted polyoxometalates. Dalton Trans 47(25):8180–8188. https://doi.org/10.1039/C8DT01291A
Han Y, Wu Y, Lai W, Cao R (2015) Electrocatalytic water oxidation by a water-soluble nickel porphyrin complex at neutral pH with low overpotential. Inorg Chem 54(11):5604–5613. https://doi.org/10.1021/acs.inorgchem.5b00924
Lubitz W, Reijerse EJ, Messinger J (2008) Solar water-splitting into H2 and O2: design principles of photosystem II and hydrogenases. Energ & Environ Sci 1(1):15–31. https://doi.org/10.1039/B808792J
Loll B, Kern J, Saenger W, Zouni A, Biesiadka J (2005) Towards complete cofactor arrangement in the 3.0 Å resolution structure of photosystem II. Nature 438(7070):1040–1044. https://doi.org/10.1038/nature04224
Cox N, Retegan M, Neese F, Pantazis DA, Boussac A, Lubitz W (2014) Electronic structure of the oxygen-evolving complex in photosystem II prior to O–O bond formation. Science 345(6198):804. https://doi.org/10.1126/science.1254910
Zhang C, Chen C, Dong H, Shen J-R, Dau H, Zhao J (2015) A synthetic Mn<sub>4</sub>Ca-cluster mimicking the oxygen-evolving center of photosynthesis. Science 348(6235):690. https://doi.org/10.1126/science.aaa6550
Vinyard DJ, Brudvig GW (2017) Progress toward a molecular mechanism of water oxidation in photosystem II. Annu Rev Phys Chem 68(1):101–116. https://doi.org/10.1146/annurev-physchem-052516-044820
Suen N-T, Hung S-F, Quan Q, Zhang N, Xu Y-J, Chen HM (2017) Electrocatalysis for the oxygen evolution reaction: recent development and future perspectives. Chem Soc Rev 46(2):337–365. https://doi.org/10.1039/C6CS00328A
Blakemore JD, Crabtree RH, Brudvig GW (2015) Molecular catalysts for water oxidation. Chem Rev 115(23):12974–13005. https://doi.org/10.1021/acs.chemrev.5b00122
Matheu R, Garrido-Barros P, Gil-Sepulcre M, Ertem MZ, Sala X, Gimbert-Suriñach C, Llobet A (2019) The development of molecular water oxidation catalysts. Nat Rev Chem 3(5):331–341. https://doi.org/10.1038/s41570-019-0096-0
Bonchio M, Syrgiannis Z, Burian M, Marino N, Pizzolato E, Dirian K, Rigodanza F, Volpato GA, La Ganga G, Demitri N, Berardi S, Amenitsch H, Guldi DM, Caramori S, Bignozzi CA, Sartorel A, Prato M (2019) Hierarchical organization of perylene bisimides and polyoxometalates for photo-assisted water oxidation. Nat Chem 11(2):146–153. https://doi.org/10.1038/s41557-018-0172-y
Lauinger SM, Piercy BD, Li W, Yin Q, Collins-Wildman DL, Glass EN, Losego MD, Wang D, Geletii YV, Hill CL (2017) Stabilization of polyoxometalate water oxidation catalysts on hematite by atomic layer deposition. ACS Appl Mater Interface 9(40):35048–35056. https://doi.org/10.1021/acsami.7b12168
Han Z, Horak KT, Lee HB, Agapie T (2017) Tetranuclear manganese models of the OEC displaying hydrogen bonding interactions: application to electrocatalytic water oxidation to hydrogen peroxide. J Am Chem Soc 139(27):9108–9111. https://doi.org/10.1021/jacs.7b03044
Liao R-Z, Siegbahn PEM (2015) Mechanism for OO bond formation in a biomimetic tetranuclear manganese cluster—a density functional theory study. J Photochem Photobio B 152:162–172. https://doi.org/10.1016/j.jphotobiol.2014.12.005
Atanu D, Virendra K, Shubhadeep P, Anku G, Sumit B, Tharangattu N, Vadapalli C (2019) A Tetranuclear Cobalt (II) phosphate possessing a D4R core: an efficient water oxidation catalyst. https://doi.org/10.1039/D0DT00010H
Hodel FH, Luber S (2016) What influences the water oxidation activity of a bioinspired molecular CoII4O4 Cubane? an in-depth exploration of catalytic pathways. ACS Catal 6(3):1505–1517. https://doi.org/10.1021/acscatal.5b02507
Gao W-S, Wang J-M, Shi N-N, Chen C-N, Fan Y-H, Wang M (2019) Electrocatalytic water oxidation studies of a tetranuclear Cu(ii) complex with cubane-like core Cu4(μ3-O)4. New J Chem 43(11):4640–4647. https://doi.org/10.1039/C8NJ06263C
Li T-T, Zheng Y-Q (2016) Electrocatalytic water oxidation using a chair-like tetranuclear copper(ii) complex in a neutral aqueous solution. Dalton Trans 45(32):12685–12690. https://doi.org/10.1039/C6DT01891B
Evangelisti F, Moré R, Hodel F, Luber S, Patzke GR (2015) 3d–4f CoII3Ln(OR)4 cubanes as bio-inspired water oxidation catalysts. J Am Chem Soc 137(34):11076–11084. https://doi.org/10.1021/jacs.5b05831
Lan T-X, Gao W-S, Chen C-N, Wang H-S, Wang M, Fan Y-H (2018) Two tetranuclear 3d–4f heterometal complexes Mn2Ln2 (Ln = Dy, Gd): synthesis, structure, magnetism, and electrocatalytic reactivity for water oxidation. New J Chem 42(8):5798–5805. https://doi.org/10.1039/C8NJ00149A
Daniel Q, Duan L, Timmer BJJ, Chen H, Luo X, Ambre R, Wang Y, Zhang B, Zhang P, Wang L, Li F, Sun J, Ahlquist M, Sun L (2018) Water oxidation initiated by in situ dimerization of the molecular Ru(pdc) catalyst. ACS Catal 8(5):4375–4382. https://doi.org/10.1021/acscatal.7b03768
Matheu R, Ertem MZ, Gimbert-Suriñach C, Sala X, Llobet A (2019) Seven coordinated molecular ruthenium-water oxidation catalysts: a coordination chemistry journey. Chem Rev 119(6):3453–3471. https://doi.org/10.1021/acs.chemrev.8b00537
Liu X, Maegawa Y, Goto Y, Hara K, Inagaki S (2016) Heterogeneous catalysis for water oxidation by an iridium complex immobilized on bipyridine-periodic Mesoporous organosilica. Angew Chem Int Edit 55(28):7943–7947. https://doi.org/10.1002/anie.201601453
Azadi G, Zand Z, Mousazade Y, Bagheri R, Cui J, Song Z, Bikas R, Wozniak K, Allakhverdiev SI, Najafpour MM (2019) A tetranuclear nickel(II) complex for water oxidation: meeting new challenges. Int J Hydrog Energy 44(5):2857–2867. https://doi.org/10.1016/j.ijhydene.2018.12.059
Han A, Chen H, Sun Z, Xu J, Du P (2015) High catalytic activity for water oxidation based on nanostructured nickel phosphide precursors. Chem Commun 51(58):11626–11629. https://doi.org/10.1039/C5CC02626A
Jia H, Yao Y, Zhao J, Gao Y, Luo Z, Du P (2018) A novel two-dimensional nickel phthalocyanine-based metal–organic framework for highly efficient water oxidation catalysis. J Mater Chem A 6(3):1188–1195. https://doi.org/10.1039/C7TA07978H
Guan J, Duan Z, Zhang F, Kelly SD, Si R, Dupuis M, Huang Q, Chen JQ, Tang C, Li C (2018) Water oxidation on a mononuclear manganese heterogeneous catalyst. Nat Catal 1(11):870–877. https://doi.org/10.1038/s41929-018-0158-6
Kottrup KG, D’Agostini S, van Langevelde PH, Siegler MA, Hetterscheid DGH (2018) Catalytic activity of an iron-based water oxidation catalyst: substrate effects of graphitic electrodes. ACS Catal 8(2):1052–1061. https://doi.org/10.1021/acscatal.7b03284
Okamura M, Kondo M, Kuga R, Kurashige Y, Yanai T, Hayami S, Praneeth VKK, Yoshida M, Yoneda K, Kawata S, Masaoka S (2016) A pentanuclear iron catalyst designed for water oxidation. Nature 530(7591):465–468. https://doi.org/10.1038/nature16529
Najafpour MM, Feizi H (2018) Water oxidation catalyzed by two cobalt complexes: new challenges and questions. Catal Sci Technol 8(7):1840–1848. https://doi.org/10.1039/C7CY02602A
Xu J-H, Guo L-Y, Su H-F, Gao X, Wu X-F, Wang W-G, Tung C-H, Sun D (2017) Heptanuclear CoII5CoIII2 cluster as efficient water oxidation catalyst. Inorg Chem 56(3):1591–1598. https://doi.org/10.1021/acs.inorgchem.6b02698
Su X-J, Gao M, Jiao L, Liao R-Z, Siegbahn PEM, Cheng J-P, Zhang M-T (2015) Electrocatalytic water oxidation by a dinuclear copper complex in a neutral aqueous solution. Angew Chem Intern Edit 54(16):4909–4914. https://doi.org/10.1002/anie.201411625
Wang J-M, Liu Y-R, Mao X-Y, Shi N-N, Zhang X, Wang H-S, Fan Y-H, Wang M (2019) Two trinuclear CuII complexes: effect of phosphonate ligand on the magnetic property and electrocatalytic reactivity for water oxidation. Chem Asian J 14(15):2685–2693. https://doi.org/10.1002/asia.201900531
Zhou W, Zheng S, Schultz JW, Rath NP, Mirica LM (2016) Aromatic cyanoalkylation through double C-H activation mediated by Ni(III). J Am Chem Soc 138(18):5777–5780. https://doi.org/10.1021/jacs.6b02405
Wang J-W, Zhang X-Q, Huang H-H, Lu T-B (2016) A Nickel(II) complex as a homogeneous electrocatalyst for water oxidation at neutral pH: dual role of HPO42− in catalysis. ChemCatChem 8(20):3287–3293. https://doi.org/10.1002/cctc.201600796
Asraf MA, Younus HA, Yusubov M, Verpoort F (2015) Earth-abundant metal complexes as catalysts for water oxidation; is it homogeneous or heterogeneous? Catal Sci & Techn 5(11):4901–4925. https://doi.org/10.1039/C5CY01251A
Liu Y-Y, Liang D, Lu L-Q, Xiao W-J (2019) Practical heterogeneous photoredox/nickel dual catalysis for C-N and C–O coupling reactions. Chem Commun 55(33):4853–4856. https://doi.org/10.1039/C9CC00987F
Feizi H, Bagheri R, Jagličić Z, Singh JP, Chae KH, Song Z, Najafpour MM (2019) A nickel(ii) complex under water-oxidation reaction: what is the true catalyst? Dalton Trans 48(2):547–557. https://doi.org/10.1039/C8DT03990A
Feizi H, Shiri F, Bagheri R, Singh JP, Chae KH, Song Z, Najafpour MM (2018) The application of a nickel(ii) Schiff base complex in water oxidation: the importance of nanosized materials. Catal Sci Technol 8(15):3954–3968. https://doi.org/10.1039/C8CY00582F
Zhang M, Zhang M-T, Hou C, Ke Z-F, Lu T-B (2014) Homogeneous electrocatalytic water oxidation at neutral ph by a robust macrocyclic Nickel(II) complex. Angew Chem Int Edit 53(48):13042–13048. https://doi.org/10.1002/anie.201406983
Bakir M, McKenzie JAM (1997) Electrochemical reactions of CO2 with fac-Re(dpk)(CO)3Cl (dpk = di-2-pyridyl ketone). J Electroanal Chem 425(1):61–66. https://doi.org/10.1016/S0022-0728(96)04940-6
Lin J, Liang X, Cao X, Wei N, Ding Y (2018) An octanuclear Cu(ii) cluster with a bio-inspired Cu4O4 cubic fragment for efficient photocatalytic water oxidation. Chem Commun 54(88):12515–12518. https://doi.org/10.1039/C8CC06362A
Serna Z, De la Pinta N, Urtiaga MK, Lezama L, Madariaga G, Clemente-Juan JM, Coronado E, Cortés R (2010) Defective dicubane-like tetranuclear Nickel(II) cyanate and azide nanoscale magnets. Inorg Chem 49(24):11541–11549. https://doi.org/10.1021/ic101970j
Tsohos A, Dionyssopoulou S, Raptopoulou CP, Terzis A, Bakalbassis EG, Perlepes SP (1999) The gem-Diol form of (py)2CO as a ligand in Cobalt(II) carboxylate clusters: a cubane complex and a novel nonanuclear species with a vertex-sharing double square pyramidal structure. Angew Chem Intern Edit 38(7):983–985. https://doi.org/10.1002/(SICI)1521-3773(19990401)38:7%3c983::AID-ANIE983%3e3.0.CO;2-K
Liu W, Thorp HH (1993) Bond valence sum analysis of metal-ligand bond lengths in metalloenzymes and model complexes 2 Refined distances and other enzymes. Inorg Chem 32(19):4102–4105. https://doi.org/10.1021/ic00071a023
Opalade AA, Gomez-Garcia CJ, Gerasimchuk N (2019) New route to polynuclear Ni(II) and Cu(II) complexes with bridging oxime groups that are inaccessible by conventional preparations. Cryst Growth Des 19(2):678–693. https://doi.org/10.1021/acs.cgd.8b01262
Vinayak R, Harinath A, Gómez-García CJ, Panda TK, Benmansour S, Nayek HP (2016) Solvent modulated assembly of two Ni(II) complexes: syntheses. Struct Magn Prop Chem Select 1(20):6532–6539. https://doi.org/10.1002/slct.201601385
Luo G-Y, Huang H-H, Wang J-W, Lu T-B (2016) Further investigation of a nickel-based homogeneous water oxidation catalyst with two cis labile sites. Chemsuschem 9(5):485–491. https://doi.org/10.1002/cssc.201501474
Shahadat HM, Younus HA, Ahmad N, Rahaman MA, Khattak ZAK, Zhuiykov S, Verpoort F (2019) Homogenous electrochemical water oxidation by a nickel(ii) complex based on a macrocyclic N-heterocyclic carbene/pyridine hybrid ligand. Catal Sci Technol 9(20):5651–5659. https://doi.org/10.1039/C9CY01485C
Wang J-W, Hou C, Huang H-H, Liu W-J, Ke Z-F, Lu T-B (2017) Further insight into the electrocatalytic water oxidation by macrocyclic nickel(ii) complexes: the influence of steric effect on catalytic activity. Catal Sci Technol 7(23):5585–5593. https://doi.org/10.1039/C7CY01527E
Zhang L-H, Yu F, Shi Y, Li F, Li H (2019) Base-enhanced electrochemical water oxidation by a nickel complex in neutral aqueous solution. Chem Commun 55(43):6122–6125. https://doi.org/10.1039/C9CC01865D
Llobet A (2014) Molecular water oxidation catalysis: a key topic for new sustainable energy conversion schemes. Wiley, Amsterdam
Zhang M-T, Chen Z, Kang P, Meyer TJ (2013) Electrocatalytic water oxidation with a copper(II) polypeptide complex. J Am Chem Soc 135(6):2048–2051. https://doi.org/10.1021/ja3097515
Kärkäs MD, Åkermark B (2016) Water oxidation using earth-abundant transition metal catalysts: opportunities and challenges. Dalton Trans 45(37):14421–14461. https://doi.org/10.1039/C6DT00809G
Nguyen AI, Ziegler MS, Oña-Burgos P, Sturzbecher-Hohne M, Kim W, Bellone DE, Tilley TD (2015) Mechanistic investigations of water oxidation by a molecular cobalt oxide analogue: evidence for a highly oxidized intermediate and exclusive terminal oxo participation. J Am Chem Soc 137(40):12865–12872. https://doi.org/10.1021/jacs.5b08396
Hong S, Lee Y-M, Cho K-B, Sundaravel K, Cho J, Kim MJ, Shin W, Nam W (2011) ligand topology effect on the reactivity of a mononuclear nonheme iron(IV)-oxo complex in oxygenation reactions. J Am Chem Soc 133(31):11876–11879. https://doi.org/10.1021/ja204008u
Wang B, Lee Y-M, Tcho W-Y, Tussupbayev S, Kim S-T, Kim Y, Seo MS, Cho K-B, Dede Y, Keegan BC, Ogura T, Kim SH, Ohta T, Baik M-H, Ray K, Shearer J, Nam W (2017) Synthesis and reactivity of a mononuclear non-haem cobalt(IV)-oxo complex. Nat Commun 8(1):14839. https://doi.org/10.1038/ncomms14839
Likhtenshtein G (2016) Electron Transfer in Biological Systems. Light Energy Conversion. In: Likhtenshtein G (ed) Electron Spin Interactions in Chemistry and Biology: Fundamentals, Methods, Reactions Mechanisms, Magnetic Phenomena, Structure Investigation. Springer International Publishing, Cham, pp 259–287. https://doi.org/10.1007/978-3-319-33927-6_11
Nocera DG (2012) The artificial leaf. Acc Chem Res 45(5):767–776. https://doi.org/10.1021/ar2003013
Smith PF, Kaplan C, Sheats JE, Robinson DM, McCool NS, Mezle N, Dismukes GC (2014) What determines catalyst functionality in molecular water oxidation? dependence on ligands and metal nuclearity in cobalt clusters. Inorg Chem 53(4):2113–2121. https://doi.org/10.1021/ic402720p
Zhang M, Frei H (2017) Water oxidation mechanisms of metal oxide catalysts by vibrational spectroscopy of transient intermediates. Annu Rev Phys Chem 68(1):209–231. https://doi.org/10.1146/annurev-physchem-052516-050655
Wang X, Li B, Wu Y-P, Tsamis A, Yu H-G, Liu S, Zhao J, Li Y-S, Li D-S (2020) Investigation on the component evolution of a tetranuclear nickel-cluster-based metal-organic framework in an electrochemical oxidation reaction. Inorg Chem 59(7):4764–4771. https://doi.org/10.1021/acs.inorgchem.0c00024
Sheldrick G (1996) SADABS, Program for area detector adsorption correction. Institute for Inorg. Chem., University of Göttingen, Germany 33
Sheldrick GM (2015) Crystal structure refinement with SHELXL. Acta Crystallogr Sect C Struct Chem 71(1):3–8
Acknowledgements
This work was supported by the National Natural Science Foundation of China (No. 21601171), Natural Science Foundation of Shandong Province (No. ZR2016BB08) and the Fundamental Research Funds for the Central Universities (Chinese Universities Scientific Fund, No. 201713028).
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare no conflict of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Wang, J., Meng, X., Xie, W. et al. Two biologically inspired tetranuclear nickel(II) catalysts: effect of the geometry of Ni4 core on electrocatalytic water oxidation. J Biol Inorg Chem 26, 205–216 (2021). https://doi.org/10.1007/s00775-020-01846-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00775-020-01846-4