Abstract
Introduction
The goal is to propose a material scientific hypothesis for the atomic arrangement of calcium phosphates during the mineralization of bones.
Materials and methods
It was reached by the analysis of bones of healthy and osteoporotic rats using analytical transmission electron microscopic methods.
Results
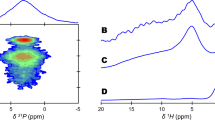
Electron diffraction patterns show hydroxyapatite (HAP) as dominant phase within the mineralized areas. In the electron energy loss spectrum, a double peak of the phosphorous L-edge seems to be a characteristic feature of the phosphorous binding in biological HAP. The hypothesis bases on periodic features on the collagen surface which agree with distances between oxygen atoms in the (200) plane of octacalcium phosphate (OCP). Bridge pillars for the HAP network consist of OCP coupled with a half unit cell on collagen by oxygen–hydrogen bridges. Possibly, the metastable OCP bridges are only a transient step, while the mineralization is starting. OCP and HAP couple by similar distances of calcium atoms in an interface close to the (100) planes of the OCP and the HAP network. To reach the perfect overlap of the equidistant Ca atoms, the HAP network has to be rotated by 22.5° around the a-axis, 11.5° around the c-axis of HAP, and 10.1° around an axis perpendicular to a and c.
Conclusions
A supercell based on this idea is able to explain the dominance of HAP in the electron diffraction patterns, the arrangement of the (002) lattice planes perpendicular to the collagen fiber axis, and sections of high-resolution TEM images.













Similar content being viewed by others
References
Dorozhkin SV, Epple M (2009) Die biologische und medizinische Bedeutung von Calciumphosphaten. Angew Chem 114:3260–3277
Wang Y, Azaïs T, Robin M, Vallée A, Catania C, Legriel P, Pehau-Arnaudet G, Babenneau F, Giraud-Guille MM, Nassif N (2012) The predominant role of collagen in the nucleation, growth, structure and orientation of bone apatite. Nat Mater 11:724–733. https://doi.org/10.1038/NMAT3362
Golub EE (2009) Role of matrix vesicles in biomineralization. Biochim Biophys Acta 1790:1592–1598
Bottini M, Mebarek S, Anderson KL, Strzelecka-Kiliszek A, Bozycki L, Sper-Simão AM, Bolean M, Ciancaglin P, Bandorowicz-Pikula J, Pikula S, Magne D, Volkmann N, Hanein D, Millán JL, Buchet R (2018) Matrix vesicles from chondrocytes and osteoblasts: their biogenesis, properties, functions and biomimetic models. Biochim Biophys Acta Gen Subj 1862:532–546
Cui L, Houston DA, Farquharson C, MacRae VE (2016) Characterisation of matrix vesicles in skeletal and soft tissue mineralization. Bone 87:147–158
Bleek K, Taubert A (2013) New developments in polymer-controlled, bioinspired calcium phosphate mineralization from aqueous solution. Acta Biomater 9:6283–6321. https://doi.org/10.1016/j.actbio.2012.12.027
Cölfen H, Mann S (2003) High-order organization by mesoscale self-assembly and transformation of hybrid nanostructures. Angew Chem Int Ed 42:2350–2365
Olszta MJ, Cheng X, Jee SS, Kumar R, Kim YY, Kaufmann MJ, Douglas EP, Gower LB (2007) Bone structure and formation: a new perspective. Mat Sci Eng. https://doi.org/10.1016/j.mser.2007.05.001
Weiner S, Wagner HD (1998) The material bone: structure mechanical function relations. Ann Rev Mater Sci 28:271–298
Reznikov N, Shahar R, Weiner S (2014) Bone hierarchical structure in three dimensions. Acta Biomater 10:3815–3826
Grandfield K, Vuong V, Schwarcz HP (2018) Ultrastructure of bone: hierarchical features from nanometer to micrometer scale revealed in focused ion beam sections in the TEM. Calcif Tissue Int 103:606–616. https://doi.org/10.1007/s00223-018-0454-9
Kourkoutis LF, Plitzko JM, Baumeister W (2012) Electron microscopy of biological materials at the nanometer scale. Annu Rev Mater Res 42:33–58
Thomas J, Gemming T (2014) Analytical transmission electron microscopy—an introduction for operators. Springer, Dordrecht, Heidelberg, New York, London (ISBN 978-94-017-8600-3)
Reznikov N, Shahar R, Weiner S (2014) Three-dimensional structure of human lamellar bone: the presence of two different materials and new insights into the hierarchical organization. Bone 50:93–104
Nalla RK, Porter AE, Daraio C, Minor AM, Radmilovic V, Stach EA, Tomsia AP, Ritchie RO (2005) Ultrastructural examination of dentin using focused ion-beam cross-sectioning and transmission electron microscopy. Micron 36:672–680
Heinemann S, Coradin T, Worch H, Wiesmann HP, Hanke T (2011) Possibilities and limitations of preparing silica/collagen/hydroxyapatite composite xerogels as load-bearing biomaterials. Compos Sci Technol 71:1873–1880
Rößler S, Heinemann C, Kruppke B, Wagner AS, Wenisch S, Wiesmann HP, Hanke T (2018) Manipulation of osteoclastogenesis: bioactive multiphasic silica/collagen composites and their effects of surface and degradation products. Mater Sci Eng C 93:265–276
Schwarcz HP (2015) The ultrastructure of bone as revealed in electron microscopy of ion-milled sections. Semin Cell Dev Biol 46:44–50
Kern C, Quade M, Ray S, Thomas J, Schumacher M, Gemming T, Gelinsky M, Alt V, Rohnke M (2018) Investigation of strontium transport and strontium quantification in cortical rat bone by time-of-flight secondary ion mass spectrometry. J R Soc Interface 16:20180638. https://doi.org/10.1098/rsif.2018.0638
Kay MI, Young RA, Posner AS (1964) Crystal structure of hydroxyapatite. Nature 204:1050–1052 (cited by FIZ Karlsruhe, ICSD code 22059)
Brown PW, Martin RI (1999) An analysis of hydroxyapatite surface layer formation. J Phys Chem B 103:1671–1675
Brown WE (1962) Octacalcium phosphate and hydroxyapatite. Nature 196:1048–1050 (cited by FIZ Karlsruhe, ICSD code 27050)
Henning S (2006) Morphologie und Mikromechanik von Knochen und neuartigen, partiell resorbierbaren Knochenzementen. Diss Halle: urn:nbn:de:gbv:3-000012394. https://doi.org/10.25673/2739
McNally EA, Schwarcz HP, Botton GA, Arsenault AL (2012) A model for the ultrastructure of bone based on electron microscopy of ion-milled sections. PLoS ONE 7:e29258
Rubin MA, Jasiuk I, Taylor J, Rubin J, Ganey T, Apkarian RP (2003) TEM analysis of the nanostructure of normal and osteoporotic human trabecular bone. Bone 33:270–282
Rohnke M, Pfitzenreuter S, Mogwitz B, Henß A, Thomas J, Bieberstein D, Gemming T, Otto SK, Ray S, Schumacher M, Gelinsky M, Alt V (2017) Strontium release from Sr2+-loaded bone cements and dispersion in healthy and osteoporotic rat bone. J Control Release 262:159–169
Dorozhkin SV (2011) Calcium orthophosphates—occurrence, properties, biomineralization, pathological calcification and biomimetic applications. Biomatter 1:121–164
EELS.info: EELS Atlas: Calcium http://www.eels.info/atlas/calcium. Accessed 23 Oct 2018
Bearden JA, Burr AF (1967) Reevaluation of X-ray atomic energy levels. Rev Mod Phys 39:125–142
Schreiber TP, Wims AM (1981) A quantitative X-ray microanalysis thin film method using K-, L- and M-lines. Ultramicroscopy 6:323–334
Boonrungsiman S, Gentleman E, Carzaniga R, Evans ND, McComb DW, Porter AE, Stevens MM (2012) The role of intracellular calcium phosphate in osteoblast-mediated bone apatite formation. PNAS 109:14170–14175
EELS.info: EELS Atlas: Phosphorus. http://www.eels.info/atlas/phosphorus Accessed 11 Nov 2018
Kruse J, Leinweber P, Eckhardt KU, Godlinski F, Hu Y (2009) Phosphorus L2,3-edge XANES: overview of reference compounds. J Synchrotron Rad 16:247–259
Wess TJ (2008) Collagen fibrillar structure and hierarchies, chapter 3. In: Fratzl P (ed) Collagen structure and mechanics. Springer, New York (ISBN 978-0-387-73905-2)
Shoulders MD, Raines RT (2009) Collagen structure and stability. Annu Rev Biochem 78:929–958
Stadelmann P (1987) EMS—a software package for electron diffraction analysis and HREM image simulation in materials science. Ultramicroscopy 21:131–146 (used Java-EMS Version 6.6201U2011)
Su X, Sun K, Cui FZ, Landis WJ (2003) Organization of apatite crystals in human woven bone. Bone 32:150–162
Göbel C, Simon P, Buder J, Tlatlik H, Kniep R (2004) Phase formation and morphology of calcium phosphate-gelatine-composites grown by double diffusion technique: the influence of fluoride. J Mater Chem 14:2225–2230
Kruppke B, Heinemann C, Keroué A, Thomas J, Rößler S, Wiesmann HP, Gemming T, Worch H, Hanke T (2017) Calcite and hydroxyapatite gelatin composites as bone substitution material made by double migration technique. Cryst Growth Des 17:738–745
Rühl RR (2011) Zur Calciumphosphatpräzipitation mit Phosphoserin, Fetuin, Osteocalcin, Kollagen und in Vesikeln. Diss TU Dresden. https://nbn-resolving.org/urn:nbn:de:bsz:14-qucosa-78417
Brown WE, Smith JP, Lehr JP, Frazier AW (1962) Octacalcium phosphate and hydroxyapatite. Nature 196:1048–1055
Fernandez ME, Zorrilla-Cangas C, Garcia-Garcia R, Ascencio JA, Reyes-Gasga J (2003) New model for the hydroxyapatite-octacalcium phosphate interface. Acta Cryst B59:175–181
Rich A, Crick FHC (1961) The molecular structure of collagen. J Mol Biol 3:483–506
Rey C, Combes C, Drouet C, Glimcher MJ (2009) Bone mineral: update on chemical composition and structure. Osteoporos Int 20:1013–1021
Weiner S (2006) Transient precursor strategy in mineral formation of bone. Bone 39:431–433
Lotsari A, Rajasekharan AK, Halvarsson M, Andersson M (2018) Transformation of amorphous calcium phosphate to bone-like apatite. Nat Commun 9:4170. https://doi.org/10.1038/s41467-018-06570-x
Ducy P, Desbois C, Boyce B, Pinerot G, Story B, Dunstan C, Smith E, Bonadio J, Goldstein S, Gundberg C, Bradley A, Karsenty G (1996) Increased bone formation in osteocalcin-deficient mice. Nature 382:448–452
Timpel M (2012) Einfluss von strontium auf die mikrostruktur von aluminium-silizium legierungen. Diss Berl. https://doi.org/10.5442/d0026(ISSN 1868-5781)
Christoffersen J, Christoffersen MR, Kolthoff N, Bärenholdt O (1997) Effect of strontium ions on growth and dissolution of hydroxyapatite and on bone mineral detection. Bone 20:47–54
Tovani CB, Gloter A, Azaïs T, Selmane M, Ramos AP, Nassif N (2019) Formation of stable strontium-rich amorphous calcium phosphate: possible effects on bone mineral. Acta Biomater 92:315–324
Acknowledgements
The authors would like to thank Christine Kern from the Justus Liebig-University Giessen for provision of the rat bones and Sina Rößler (TU Dresden) for provision of silicate samples as well as Dina Bieberstein from the Leibniz-Institute for Solid State and Materials Research (IFW) Dresden for the careful electron-microscopic preparation of the cross-sections. This work was supported by the Deutsche Forschungsgemeinschaft (DFG, Collaborative Research Centre Transregio 79—subprojects Z2, M3, and M5).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
All authors have no conflicts of interest.
Ethical approval
All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
About this article
Cite this article
Thomas, J., Worch, H., Kruppke, B. et al. Contribution to understand the biomineralization of bones. J Bone Miner Metab 38, 456–468 (2020). https://doi.org/10.1007/s00774-020-01083-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00774-020-01083-4