Abstract
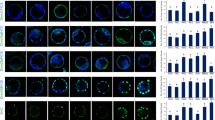
The aim of this study was to clarify the protective role of taurine in neuronal apoptosis and the role of the Wnt/PCP-Jnk pathway in mediating the preventive effects of taurine on neural tube defects (NTDs). HT-22 cells (a hippocampal neuron cell line) were divided into a control group, a glutamate-induced apoptosis group, and glutamate (4.0 mmol/L) plus low-dose taurine (L; 0.5 mmol/L) and high-dose taurine (H; 2.0 mmol/L) groups. The MTT assay was used to monitor cell proliferation and cell survival. Immunofluorescence and Western blot analyses were used to determine caspase 9 expression. Retinoic acid (RA) induced embryonic NTDs in Kunming mice, thus establishing an NTD model. Pregnant mice were divided into a control group, an RA (30 mg/kg body weight) group, and an RA (30 mg/kg body weight) plus taurine (free drinking of 2 g/L solution) group. Immunohistochemistry and Western blot analyses were used to detect the expression of Dvl, RhoA and phosphorylated (p)-Jnk/Jnk in the embryonic neural tubes. In HT-22 cells, the apoptosis rate was significantly higher and caspase 9 activation was also significantly increased in the glutamate-induced apoptosis group compared to the L and H taurine groups. In the NTD model, the expression levels of Dvl, RhoA, and p-Jnk were significantly higher in the RA group than in the control group, whereas they were significantly reduced in the RA + taurine group. This study suggests that taurine has positive effects on neuronal protection and NTD prevention. Moreover, the Wnt/PCP-Jnk-dependent pathway plays an important role in taurine-mediated prevention of NTDs.








Similar content being viewed by others
References
Ahmadian E, Babaei H, Mohajjel Nayebi A, Eftekhari A, Eghbal MA (2016) Venlafaxine-induced cytotoxicity towards isolated rat hepatocytes involves oxidative stress and mitochondrial/lysosomal dysfunction. Adv Pharm Bull 6:521–530. doi:10.15171/apb.2016.066
Blencowe H, Cousens S, Modell B, Lawn J (2010) Folic acid to reduce neonatal mortality from neural tube disorders. Int J Epidemiol 39(Suppl 1):i110–i121. doi:10.1093/ije/dyq028
Caddy J et al (2010) Epidermal wound repair is regulated by the planar cell polarity signaling pathway. Dev Cell 19:138–147. doi:10.1016/j.devcel.2010.06.008
Chen WQ et al (2001) Role of taurine in regulation of intracellular calcium level and neuroprotective function in cultured neurons. J Neurosci Res 66:612–619. doi:10.1002/jnr.10027
Dhaulakhandi DB, Rohilla S, Rattan KN (2010) Neural tube defects: review of experimental evidence on stem cell therapy and newer treatment options. Fetal Diagn Ther 28:72–78. doi:10.1159/000318201
Donato LJ, Noy N (2005) Suppression of mammary carcinoma growth by retinoic acid: proapoptotic genes are targets for retinoic acid receptor and cellular retinoic acid-binding protein II signaling. Can Res 65:8193–8199. doi:10.1158/0008-5472.CAN-05-1177
Engedal N, Auberger P, Blomhoff HK (2009) Retinoic acid regulates Fas-induced apoptosis in Jurkat T cells: reversal of mitogen-mediated repression of Fas DISC assembly. J Leukoc Biol 85:469–480. doi:10.1189/jlb.1107790
Glasco DM et al (2012) The mouse Wnt/PCP protein Vangl2 is necessary for migration of facial branchiomotor neurons, and functions independently of Dishevelled. Dev Biol 369:211–222. doi:10.1016/j.ydbio.2012.06.021
Heo SH, Kwak J, Jang KL (2015) All-trans retinoic acid induces p53-depenent apoptosis in human hepatocytes by activating p14 expression via promoter hypomethylation. Cancer Lett 362:139–148. doi:10.1016/j.canlet.2015.03.036
Jiang M, Zhu K, Grenet J, Lahti JM (2008) Retinoic acid induces caspase-8 transcription via phospho-CREB and increases apoptotic responses to death stimuli in neuroblastoma cells. Biochem Biophys Acta 1783:1055–1067. doi:10.1016/j.bbamcr.2008.02.007
Khalili Fard J, Hamzeiy H, Sattari M, Eghbal MA (2016) Protective roles of N-acetyl cysteine and/or taurine against sumatriptan-induced hepatotoxicity. Adv Pharm Bull 6:627–637. doi:10.15171/apb.2016.077
Kim GH, Han JK (2005) JNK and ROKalpha function in the noncanonical Wnt/RhoA signaling pathway to regulate Xenopus convergent extension movements. Dev Dyn 232:958–968. doi:10.1002/dvdy.20262
Kuan CY, Yang DD, Samanta Roy DR, Davis RJ, Rakic P, Flavell RA (1999) The Jnk1 and Jnk2 protein kinases are required for regional specific apoptosis during early brain development. Neuron 22:667–676
Kuida K et al (1998) Reduced apoptosis and cytochrome c-mediated caspase activation in mice lacking caspase 9. Cell 94:325–337
Li X, Weng H, Xu C, Reece EA, Yang P (2012) Oxidative stress-induced JNK1/2 activation triggers proapoptotic signaling and apoptosis that leads to diabetic embryopathy. Diabetes 61:2084–2092. doi:10.2337/db11-1624
Liu J, Qi J, Zhu J, Zhang L, Liang Y, Ning Q, Luo X (2008) Effects of retinoic acid on the expressions of Vangl1 and vangl2 in mouse fetuses. J Neurogenet 22:167–179. doi:10.1080/01677060802049605
Loebel DA, Tam PP (2012) Rho GTPases in endoderm development and differentiation. Small GTPases 3:40–44. doi:10.4161/sgtp.18820
Niizuma H et al (2006) Bcl-2 is a key regulator for the retinoic acid-induced apoptotic cell death in neuroblastoma. Oncogene 25:5046–5055. doi:10.1038/sj.onc.1209515
Owens DM, Keyse SM (2007) Differential regulation of MAP kinase signalling by dual-specificity protein phosphatases. Oncogene 26:3203–3213. doi:10.1038/sj.onc.1210412
Phadke S, Agarwal M (2012) Neural tube defects: a need for population-based prevention program. Indian J Hum Genet 18:145–147. doi:10.4103/0971-6866.100747
Roberts PJ, Der CJ (2007) Targeting the Raf-MEK-ERK mitogen-activated protein kinase cascade for the treatment of cancer. Oncogene 26:3291–3310. doi:10.1038/sj.onc.1210422
Saransaari P, Oja SS (2000) Taurine and neural cell damage. Amino Acids 19:509–526
Schaffer SW, Shimada-Takaura K, Jong CJ, Ito T, Takahashi K (2016) Impaired energy metabolism of the taurinedeficient heart. Amino Acids 48:549–558. doi:10.1007/s00726-015-2110-2
Shetewy A et al (2016) Mitochondrial defects associated with beta-alanine toxicity: relevance to hyper-beta-alaninemia. Mol Cell Biochem 416:11–22. doi:10.1007/s11010-016-2688-z
Soprano DR, Soprano KJ (1995) Retinoids as teratogens. Ann Rev Nutr 15:111–132. doi:10.1146/annurev.nu.15.070195.000551
Tada M, Kai M (2009) Noncanonical Wnt/PCP signaling during vertebrate gastrulation. Zebrafish 6:29–40. doi:10.1089/zeb.2008.0566
Torban E, Wang HJ, Groulx N, Gros P (2004) Independent mutations in mouse Vangl2 that cause neural tube defects in looptail mice impair interaction with members of the Dishevelled family. J Biol Chem 279:52703–52713. doi:10.1074/jbc.M408675200
Wansleeben C, Meijlink F (2011) The planar cell polarity pathway in vertebrate development. Dev Dyn 240:616–626. doi:10.1002/dvdy.22564
Wu CC, Bratton SB (2013) Regulation of the intrinsic apoptosis pathway by reactive oxygen species. Antioxid Redox Signal 19:546–558. doi:10.1089/ars.2012.4905
Wu G, Huang X, Hua Y, Mu D (2011) Roles of planar cell polarity pathways in the development of neural [correction of neutral] tube defects. J Biomed Sci 18:66. doi:10.1186/1423-0127-18-66
Ybot-Gonzalez P et al (2007) Convergent extension, planar-cell-polarity signalling and initiation of mouse neural tube closure. Development 134:789–799. doi:10.1242/dev.000380
Zhang H, Satyamoorthy K, Herlyn M, Rosdahl I (2003) All-trans retinoic acid (atRA) differentially induces apoptosis in matched primary and metastatic melanoma cells—a speculation on damage effect of atRA via mitochondrial dysfunction and cell cycle redistribution. Carcinogenesis 24:185–191
Zhang Y, Li D, Li H, Hou D, Hou J (2016) Taurine pretreatment prevents isoflurane-induced cognitive impairment by inhibiting ER stress-mediated activation of apoptosis pathways in the hippocampus in aged rats. Neurochem Res 41:2517–2525. doi:10.1007/s11064-016-1963-4
Zhang Z, Zhao L, Zhou Y, Lu X, Wang Z, Wang J, Li W (2017) Taurine ameliorated homocysteine-induced H9C2 cardiomyocyte apoptosis by modulating endoplasmic reticulum stress. Apoptosis Int J Program Cell Death 22:647–661. doi:10.1007/s10495-017-1351-9
Zhao Y et al (2010) Dishevelled-1 and dishevelled-3 affect cell invasion mainly through canonical and noncanonical Wnt pathway, respectively, and associate with poor prognosis in nonsmall cell lung cancer. Mol Carcinog 49:760–770. doi:10.1002/mc.20651
Zheng A, Mantymaa P, Saily M, Savolainen E, Vahakangas K, Koistinen P (2000) p53 pathway in apoptosis induced by all-trans-retinoic acid in acute myeloblastic leukaemia cells. Acta Haematol 103:135–143
Acknowledgements
This study was supported by the National Nature Science Foundation of China (81360183) and the Nature Science Foundation of NingXia.
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Ethics statement
All procedures performed in studies were in accordance with the ethical standards of the Ethics committee Ningxia Medical University.
Rights and permissions
About this article
Cite this article
Zhang, Q., Liu, Y., Wang, H. et al. The preventive effects of taurine on neural tube defects through the Wnt/PCP-Jnk-dependent pathway. Amino Acids 49, 1633–1640 (2017). https://doi.org/10.1007/s00726-017-2462-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00726-017-2462-x