Abstract
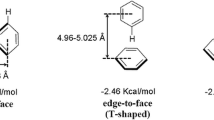
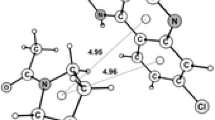
In this study we have described the non-canonical interactions between the porphyrin ring and the protein part of porphyrin-containing proteins to better understand their stabilizing role. The analysis reported in this study shows that the predominant type of non-canonical interactions at porphyrins are CH···O and CH···N interactions, with a small percentage of CH···π and non-canonical interactions involving sulfur atoms. The majority of non-canonical interactions are formed from side-chains of charged and polar amino acids, whereas backbone groups are not frequently involved. The main-chain non-canonical interactions might be slightly more linear than the side-chain interactions, and they have somewhat shorter median distances. The analysis, performed in this study, shows that about 44% of the total interactions in the dataset are involved in the formation of multiple (furcated) non-canonical interactions. The high number of porphyrin–water interactions show importance of the inclusion of solvent in protein–ligand interaction studies. Furthermore, in the present study we have observed that stabilization centers are composed predominantly from nonpolar amino acid residues. Amino acids deployed in the environment of porphyrin rings are deposited in helices and coils. The results from this study might be used for structure-based porphyrin protein prediction and as scaffolds for future porphyrin-containing protein design.





Similar content being viewed by others
References
Accelrys I (2011) Discovery studio visualizer 3.0, San Diego, CA 92121, USA, 10188 Telesis Court, Suite 100
Anand S, Anbarasu A, Sethumadhavan R (2008) Influence of C-H…pi hydrogen bonds in interleukins. In Silico Biol 8:261–273
Armstrong KM, Fairman R, Baldwin RL (1993) The (i, i + 4) Phe-His interaction studied in an alanine-based alpha-helix. J Mol Biol 230:284–291
Babu M, Kumar SS, Balaram P (2002) A C–H triplebond O hydrogen bond stabilized polypeptide chain reversal motif at the C terminus of helices in proteins. J Mol Biol 322:871–880
Bahatyrova S, Frese RN, Siebert CA, Olsen JD, Van Der Werf KO, Van GR, Niederman RA, Bullough PA, Otto C, Hunter CN (2004) The native architecture of a photosynthetic membrane. Nature 430:1058–1062
Banci L, Bertini I, Cavallaro G, Luchinat C (2002) Chemical shift-based constraints for solution structure determination of paramagnetic low-spin heme proteins with bis-His and His-CN axial ligands: the cases of oxidized cytochrome b(5) and Met80Ala cyano-cytochrome c. J Biol Inorg Chem 7:416–426
Bartlett GJ, Porter CT, Borkakoti N, Thornton JM (2002) Analysis of catalytic residues in enzyme active sites. J Mol Biol 324:105–121
Ben-Naim A, Ting KL, Jernigan RL (1990) Solvent effect on binding thermodynamics of biopolymers. Biopolymers 29:901–919
Brandl M, Weiss MS, Jabs A, Suhnel J, Hilgenfeld R (2001) C–H···pi-interactions in proteins. J Mol Biol 307:357–377
Chakkaravarthi S, Babu MM, Gromiha MM, Jayaraman G, Sethumadhavan R (2006) Exploring the environmental preference of weak interactions in (alpha/beta)8 barrel proteins. Proteins 65:75–86
Desiraju GR, Steiner T (1999) The weak hydrogen bond in structural chemistry and Biology. Oxford University Press, Oxford
Dosztanyi Z, Fiser A, Simon I (1997) Stabilization centers in proteins: identification, characterization and predictions. J Mol Biol 272:597–612
Dosztanyi Z, Magyar C, Tusnady G, Simon I (2003) SCide: identification of stabilization centers in proteins. Bioinformatics 19:899–900
Fabian M, Skultety L, Jancura D, Palmer G (2004a) Implications of ligand binding studies for the catalytic mechanism of cytochrome c oxidase. Biochim Biophys Acta 1655:298–305
Fabian M, Jancura D, Palmer G (2004b) Two sites of interaction of anions with cytochrome a in oxidized bovine cytochrome c oxidase. J Biol Chem 279:16170–16177
Fabiola GF, Krishnaswamy S, Nagarajan V, Pattabhi V (1997) C–H···O hydrogen bonds in beta-sheets. Acta Crystallogr D Biol Crystallogr 53:316–320
Galstyan AS, Zarić SD, Knapp EW (2005) Computational studies on imidazole heme conformations. J Biol Inorg Chem 10:343–354
Griep S, Hobohm U (2010) PDBselect 1992–2009 and PDBfilter-select. Nucleic Acids Res 38:D318–D319
Halgren T (1996) Merck molecular force field. I. Basis, form, scope, parameterization, and performance of MMFF94. J Comp Chem 17:490–519
Hu J, Barbour LJ, Gokel GW (2002) Probing alkali metal-pi interactions with the side chain residue of tryptophan. Proc Natl Acad Sci USA 99:5121–5126
Huang SS, Koder RL, Lewis M, Wand AJ, Dutton PL (2004) The HP-1 maquette: from an apoprotein structure to a structured hemoprotein designed to promote redox-coupled proton exchange. Proc Natl Acad Sci USA 101:5536–5541
Iakovleva O, Reiner M, Rau H, Haehnel W (2002) Mössbauer and EPR study of a cytochrome b model. Phys Chem Chem Phys 4:660
Jeffrey GA, Saenger W (1991) Hydrogen bonding in biological structures. Springer-Verlag, Berlin
Kabsch W, Sander C (1983) Dictionary of protein secondary structure: pattern recognition of hydrogen-bonded and geometrical features. Biopolymers 22:2577–2637
Karrasch S, Bullough PA, Ghosh R (1995) The 8.5 A projection map of the light-harvesting complex I from Rhodospirillum rubrum reveals a ring composed of 16 subunits. EMBO J 14:631–638
Koder RL, Dutton PL (2006) Intelligent design: the de novo engineering of proteins with specified functions. Dalton Trans, 3045–3051
Kryger G, Silman I, Sussman JL (1999) Structure of acetylcholinesterase complexed with E2020 (Aricept): implications for the design of new anti-Alzheimer drugs. Structure 7:297–307
Lehmann A, Saven JG (2008) Computational design of four-helix bundle proteins that bind nonbiological cofactors. Biotechnol Prog 24:74–79
Liu D, Williamson DA, Kennedy ML, Williams TD, Morton MM, Benson DR (1999) Aromatic side chain-porphyrin interactions in designed hemoproteins. J Am Chem Soc 121:11798–11812
Magrane M. and the UniProt consortium (2011) UniProt Knowledgebase: a hub of integrated protein data. Database, 2011: bar009
Magyar C, Gromiha MM, Pujadas G, Tusnady GE, Simon I (2005) SRide: a server for identifying stabilizing residues in proteins. Nucleic Acids Res 33:W303–W305
McDermott G, Prince SM, Freer AA, Hawthornthwaite-Lawless AM, Papiz MZ, Cogdell RJ, Isaacs NW (1995) Crystal structure of an integral membrane light-harvesting complex from photosynthetic bacteria. Nature 374:517–521
McPhail AT, Sim GA (1965) Hydroxyl-benzene hydrogen bonding: an X-ray study. Chem Commun, 124–126
Medaković VB, Milčić MK, Bogdanović GA, Zarić SD (2004) C–H···pi interactions in the metal-porphyrin complexes with chelate ring as the H acceptor. J Inorg Biochem 98:1867–1873
Novoa JJ, Mota F (1997) Substituent effects in intermolecular C(sp3)-H cdots, three dots, centered O(sp3) contacts: how strong can a C(sp3)-H cdots, three dots, centered O(sp3) hydrogen bond be? Chem Phys Lett 266:23–30
Olkhova E, Padan E, Michel H (2007) The influence of protonation states on the dynamics of the NhaA antiporter from Escherichia coli. Biophys J 92:3784–3791
Panigrahi SK (2008) Strong and weak hydrogen bonds in protein-ligand complexes of kinases: a comparative study. Amino Acids 34:617–633
Panigrahi SK, Desiraju GR (2007) Strong and weak hydrogen bonds in the protein-ligand interface. Proteins 67:128–141
Papiz MZ, Prince SM, Howard T, Cogdell RJ, Isaacs NW (2003) The structure and thermal motion of the B800–850 LH2 complex from Rps.acidophila at 2.0A resolution and 100K: new structural features and functionally relevant motions. J Mol Biol 326:1523–1538
Parkinson G, Gunasekera A, Vojtechovsky J, Zhang X, Kunkel TA, Berman H, Ebright RH (1996) Aromatic hydrogen bond in sequence-specific protein DNA recognition. Nat Struct Biol 3:837–841
Ramanavicius A, Ramanaviciene A (2009) Hemoproteins in design of biofuel cells. Fuel Cells 9:25–36
Robertson DE, Farid RS, Moser CC, Urbauer JL, Mulholland SE, Pidikiti R, Lear JD, Wand AJ, DeGrado WF, Dutton PL (1994) Design and synthesis of multi-haem proteins. Nature 368:425–432
Roszak AW, Howard TD, Southall J, Gardiner AT, Law CJ, Isaacs NW, Cogdell RJ (2003) Crystal structure of the RC-LH1 core complex from Rhodopseudomonas palustris. Science 302:1969–1972
Rothemund P (1935) Formation of porphyrins from pyrrole and aldehydes. J Am Chem Soc 57:2010–2011
Rothemund P (1936) A new porphyrin synthesis. The synthesis of porphin. J Am Chem Soc 58:625–627
Sarkhel S, Desiraju GR (2004) N–H···O, O–H···O, and C–H···O hydrogen bonds in protein-ligand complexes: strong and weak interactions in molecular recognition. Proteins 54:247–259
Senes A, Ubarretxena-Belandia I, Engelman DM (2001) The Calpha—H···O hydrogen bond: a determinant of stability and specificity in transmembrane helix interactions. Proc Natl Acad Sci USA 98:9056–9061
Steiner T (2002) The hydrogen bond in the solid state. Angew Chem Int Ed 41:48–76
Steiner T, Koellner G (2001) Hydrogen bonds with pi-acceptors in proteins: frequencies and role in stabilizing local 3D structures. J Mol Biol 305:535–557
Stojanović SĐ, Zarić SD (2009) Hydrogen bonds and hydrophobic interactions of porphyrins in porphyrin-containing proteins. Open Struct Biol J 3:34–41
Stojanović SĐ, Medaković VB, Predović G, Beljanski M, Zarić SD (2007) XH/pi interactions with the pi system of porphyrin ring in porphyrin-containing proteins. J Biol Inorg Chem 12:1063–1071
Stojanović SĐ, Isenović ER, Zarić BL (2011) Contribution of non-canonical interactions to the stability of Sm/LSm oligomeric assemblies. Mol Inf 30:430–442
Teyra J, Pisabarro MT (2007) Characterization of interfacial solvent in protein complexes and contribution of wet spots to the interface description. Proteins 67:1087–1095
Tiwari A, Panigrahi SK (2007) HBAT: a complete package for analysing strong and weak hydrogen bonds in macromolecular crystal structures. In Silico Biol 7:651–661
Ueno T, Yokoi N, Unno M, Matsui T, Tokita Y, Yamada M, Ikeda-Saito M, Nakajima H, Watanabe Y (2006) Design of metal cofactors activated by a protein–protein electron transfer system. Proc Natl Acad Sci USA 103:9416–9421
Walker FA (2004) Models of the bis-histidine-ligated electron-transferring cytochromes. Comparative geometric and electronic structure of low-spin ferro- and ferrihemes. Chem Rev 104:589–615
Zarić SD, Popović DM, Knapp EW (2001) Factors determining the orientation of axially coordinated imidazoles in heme proteins. Biochemistry 40:7914–7928
Zou H, Strzalka J, Xu T, Tronin A, Blasie JK (2007) Three-dimensional structure and dynamics of a de novo designed, amphiphilic, metallo-porphyrin-binding protein maquette at soft interfaces by molecular dynamics simulations. J Phys Chem B 111:1823–1833
Acknowledgments
This work was supported by the grants No. 172001 and 173033 (to E.R.I.) from the Ministry of Education and Science of the Republic of Serbia.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Stojanović, S.Đ., Isenović, E.R. & Zarić, B.L. Non-canonical interactions of porphyrins in porphyrin-containing proteins. Amino Acids 43, 1535–1546 (2012). https://doi.org/10.1007/s00726-012-1228-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00726-012-1228-8