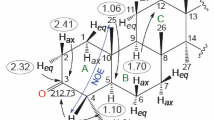
Abstract
The TES ether of the C6-hydroxy derivative of naturally occurring epi-jasmonic acid (epi-JA) was designed as epimerization-free equivalent of epi-JA. The TES ether was synthesized from (1R,4S)-4-hydroxycyclopent-2-enyl acetate in 13 steps. The acid part of the ether was activated with ClCO2Bui and subjected to condensation with l-amino acid at room temperature for 48 h. The TES group in the condensation product was removed in HCO2H (0°C, 30 min) and the resulting hydroxyl group was oxidized with Jones reagent (acetone, 0°C, 30 min) to furnish the amino acid conjugate of epi-JA. The amino acids examined are l-isoleucine, l-leucine, l-alanine, l-valine, and d-allo-isoleucine, which afforded the conjugates in 48–68% yields with 89–96% diastereomeric purity over the trans isomers. Similarly, the possible three stereoisomers of epi-JA were condensed with l-isoleucine successfully, producing the corresponding stereoisomers in good yields.






Similar content being viewed by others
References
Acharya HP, Kobayashi Y (2006) Highly efficient total synthesis of Δ12-PGJ2, 15-deoxy-Δ12,14-PGJ2, and their analogues. Tetrahedron 62:3329–3343
Ainai T, Matsuumi M, Kobayashi Y (2003) Efficient total synthesis of 12-oxo-PDA and OPC-8:0. J Org Chem 68:7825–7832
Asamitsu Y, Nakamura Y, Ueda M, Kuwahara S, Kiyota H (2006) Synthesis and odor description of both enantiomers of methyl 4,5-didehydrojasmonate, a component of jasmin absolute. Chem Biodivers 3:654–659
Chini A, Fonseca S, Fernández G, Adie B, Chico JM, Lorenzo O, García-Casado G, López-Vidriero I, Lozano FM, Ponce MR, Micol JL, Solano R (2007) The JAZ family of repressors is the missing link in jasmonate signalling. Nature 448:666–671
Chung HS, Cooke TF, DePew CL, Patel LC, Ogawa N, Kobayashi Y, Howe GA (2010) Alternative splicing expands the repertoire of dominant JAZ repressors of jasmonate signaling. Plant J 63:613–622
Creelman RA, Mullet JE (1997) Biosynthesis and action of jasmonates in plants. Annu Rev Plant Physiol Plant Mol Biol 48:355–381
Fonseca S, Chini A, Hamberg M, Adie B, Porzel A, Kramell R, Miersch O, Wasternack C, Solano R (2009) (+)-7-iso-Jasmonoyl-l-isoleucine is the endogenous bioactive jasmonate. Nature Chem Biol 5:344–350
Kiyota H, Higashi E, Koike T, Oritani T (2001) Lipase-catalyzed preparation of both enantiomers of methyl jasmonate. Tetrahedron Asymmetry 12:1035–1038
Kramell R, Schmidt J, Schneider G, Sembdner G, Schreiber K (1988) Synthesis of N-(jasmonoyl)amino acid conjugates. Tetrahedron 44:5791–5807
Kramell R, Miersch O, Hause B, Ortel B, Parthier B, Wasternack C (1997a) Amino acid conjugates of jasmonic acid induce jasmonate-responsive gene expression in barley (Hordeum vulgare L.) leaves. FEBS Lett 414:197–202
Kramell R, Schneider G, Miersch O (1997b) Chiral separation of amide conjugates of jasmonic acid by liquid chromatography. Chromatographia 45:104–108
Miersch O, Brückner B, Schmidt J, Sembdner G (1992) Cyclopentane fatty acids from Gibberella Fujikuroi. Phytochemistry 31:3835–3837
Miersch O, Bohlmann H, Wasternack C (1999) Jasmonates and related compounds from Fusarium oxysporum. Phytochemistry 50:517–523
Nonaka H, Ogawa N, Maeda N, Wang YG, Kobayashi Y (2010) Stereoselective synthesis of epi-jasmonic acid, tuberonic acid, and 12-oxo-PDA. Org Biomol Chem 8:5212–5223
Ogawa N, Kobayashi Y (2008) Strategy for synthesis of the isoleucine conjugate of epi-jasmonic acid. Tetrahedron Lett 49:7124–7127
Schmidt J, Kramell R, Brückner C, Schneider G, Sembdner G, Schreiber K, Stach J, Jensen E (1990) Gas chromatographic/mass spectrometric and tandem mass spectrometric investigations of synthetic amino acid conjugates of jasmonic acid and endogenously occurring related compounds from Vicia faba L. Biomed Environ Mass Spectrom 19:327–338
Seto H, Fujioka S, Fujisawa H, Goto K, Nojiri H, Yamane H, Yoshida S (1996) Preparation of (±)-2-(2,3–2H2)jasmonic acid and its methyl ester, methyl (±)-2-(2,3-2H2)jasmonate. Biosci Biotechnol Biochem 60:1709–1711
Seto H, Nomura E, Fujioka S, Koshino H, Suenaga T, Yoshida S (1999) Easy preparation of methyl 7-epi-jasmonate and four stereoisomers of methyl cucurbate, and assessment of the stereogenic effect of jasmonate on phytohormonal activities. Biosci Biotechnol Biochem 63:361–367
Sheard LB, Tan X, Mao H, Withers J, Ben-Nissan G, Hinds TR, Kobayashi Y, Hsu FF, Sharon M, Browse J, He SY, Rizo J, Howe GA, Zheng N (2010) Jasmonate perception by inositol-phosphate-potentiated COI1–JAZ co-receptor. Nature 468:400–405
Soloshonok VA (2002) Highly diastereoselective Michael addition reactions between nucleophilic glycine equivalents and β-substituted-α,β-unsaturated carboxylic acid derivatives; a general approach to the stereochemically defined and sterically χ-constrained α-amino acids. Curr Org Chem 6:341–364
Staswick PE, Tiryaki I (2004) The oxylipin signal jasmonic acid is activated by an enzyme that conjugates it to isoleucine in arabidopsis. Plant Cell 16:2117–2127
Tamogami S, Rakwal R, Agrawal GK (2008) Interplant communication: airborne methyl jasmonate is essentially converted into JA and JA-Ile activating jasmonate signaling pathway and VOCs emission. Biochem Biophys Res Commun 376:723–727
Thines B, Katsir L, Melotto M, Niu Y, Mandaokar A, Liu G, Nomura K, He SY, Howe GA, Browse J (2007) JAZ repressor proteins are targets of the SCFCOI1 complex during jasmonate signalling. Nature 448:661–665
Walter A, Mazars C, Maitrejean M, Hopke J, Ranjeva R, Boland W, Mithöfer A (2007) Structural requirements of jasmonates and synthetic analogues as inducers of Ca2+ signals in the nucleus and the cytosol of plant cells. Angew Chem Int Ed 46:4783–4785
Xie DX, Feys BF, James S, Nieto-Rostro M, Turner JG (1998) COI1: an arabidopsis gene required for jasmonate-regulated defense and fertility. Science 280:1091–1094
Acknowledgments
Racemic methyl jasmonate was kindly provided by Zeon Co. Ltd., Japan. This work was supported by a Grant-in-Aid for Scientific Research from the Ministry of Education, Science, Sports, and Culture, Japan.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Ogawa, N., Kobayashi, Y. Synthesis of the amino acid conjugates of epi-jasmonic acid. Amino Acids 42, 1955–1966 (2012). https://doi.org/10.1007/s00726-011-0925-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00726-011-0925-z