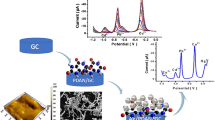
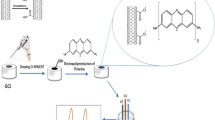
Abstract
A Schiff base ligand named bis(5-bromo-2-hydroxybenzaldehyde) 1,2-propanediimine has been used to fabricate a modified glassy carbon electrode for the selective voltammetric detection of the low-level of Cu(II). The electrochemical behavior of unmodified and modified electrodes was investigated using the cyclic voltammetry technique, while the optimization of the parameters affecting the electrode characteristics and its analytical performance were evaluated by employing the differential pulse anodic stripping voltammetry. Under the optimized conditions (pH 4, decomposition time 360 s, decomposition potential vs. SCE − 1.2 V), the peak current variation as a function of the copper(II) concentration in the range of 10–100 nM was linear, with a calculated detection limit of 4.2 nM. The evaluated selectivity, reproducibility, and repeatability of the developed modified electrode showed its satisfactory performance. The ability of the prepared electrode was evaluated by its application for the analysis of Cu(II) in a fish liver tissue sample. It was confirmed also that the proposed modifie delectrode can be successfuly used for the determination of Cd(II) and Pb(II).
Graphic abstract










Similar content being viewed by others
References
Mohammed AS, Kapri A, Goel R (2011) Heavy metal pollution: source, impact, and remedies. In: Khan MS, Zaidi A, Goel R, Musarrat J (eds) Biomanagement of metal-contaminated soils. Environmental Pollution, vol 20. Springer, Dordrecht
Alloway BJ (2013) Sources of heavy metals and metalloids in soils. In: Alloway B (ed) Heavy metals in soils. Environmental pollution, vol 22. Springer, Dordrecht, p 11
Carolin CF, Kumar PS, Saravanan A, Joshiba GJ, Naushad M (2017) J Environ Chem Eng 5:2782
Lu Y, Liang X, Niyungeko C, Zhou J, Xu J, Tian G (2018) Talanta 178:324
Scheiber IF, Mercer JF, Dringen R (2014) Prog Neurobiol 116:33
Tishchenko K, Beloglazkina E, Mazhuga A, Zyk N (2016) Rev J Chem 6:49
Noël L, Chekri R, Millour S, Vastel C, Kadar A, Sirot V, Leblanc J-C, Guérin T (2012) Food Chem 132:1502
Cherfi A, Abdoun S, Gaci O (2014) Food Chem Toxicol 70:48
Adrees M, Ali S, Rizwan M, Ibrahim M, Abbas F, Farid M, Zia-ur-Rehman M, Irshad MK, Bharwana SA (2015) Environ Sci Pollut Res 22:8148
Vasile D, Gaina G, Petcu LC, Coprean D, Tofan L, Dinischiotu A (2019) Bull Environ Contam Toxicol 102:39
Bilal M, Kazi TG, Afridi HI, Arain MB, Baig JA, Khan M, Khan N (2016) J Ind Eng Chem 40:137
Baytak S, Kasumov VT (2017) Anal Lett 50:105
Yigit M, Celikkol B, Yilmaz S, Bulut M, Ozalp B, Dwyer RL, Maita M, Kizilkaya B, Yigit Ü, Ergün S (2018) Hum Ecol Risk Assess 24:465
Wu W, Jia M, Wang Z, Zhang W, Zhang Q, Liu G, Zhang Z, Li P (2019) Microchim Acta 186:1
Dias FDS, de Neto SC, Pires LDN, Lemos VA (2020) Anal Methods 12:865
Mirabi A, Rad AS, Khodadad H (2015) J Magn Magn Mater 389:130
Cui C, Peng H, Zhang Y, Nan K, He M, Chen B, Hu B (2015) J Anal At Spectrom 30:1386
Topuz B, Adanur ŞM, Yalcuk A (2017) Turk J Chem 41:619
Asghari A, Ghazaghi M, Rajabi M, Behzad M, Ghaedi M (2014) J Serb Chem Soc 79:63
Teixeira LS, Rocha RB, Sobrinho EV, Guimarães PR, Pontes LA, Teixeira JS (2007) Talanta 72:1073
Akanji SP, Ama OM, Ray SS, Osifo PO (2020) Metal oxide nanomaterials for electrochemical detection of heavy metals in water. In: Ray SS (ed) Ama OM. Nanostructured metal-oxide electrode materials for water purification. Engineering Materials. Springer, Cham
Liu Z-G, Huang X-J (2014) TrAC Trends Anal Chem 60:25
Ariño C, Serrano N, Díaz-Cruz JM, Esteban M (2017) Anal Chim Acta 990:11
Chen G, Wang X, Wang L (2020) Int J Electrochem Sci 15:4252
Baig N, Sajid M, Saleh TA (2019) TrAC Trends Anal Chem 111:47
Oztekin Y, Tok M, Nalvuran H, Kiyak S, Gover T, Yazicigil Z, Ramanaviciene A, Ramanavicius A (2010) Electrochim Acta 56:387
Zou L, Li Y, Zhao W, Zhang S, Ye B (2012) J Solid State Electrochem 16:505
Škrivanj SB, Stanković D, Khan M, Nikolić AS, Vulić PJ, Manojlović DD (2015) Anal Bioanal Electrochem 7:230
Xing H, Xu J, Zhu X, Duan X, Lu L, Wang W, Zhang Y, Yang T (2016) J Electroanal Chem 760:52
Zhang T, Jin H, Fang Y, Guan J, Ma S, Pan Y, Zhang M, Zhu H, Liu X, Du M (2019) Mater Chem Phys 225:433
Ansari RM, Bhat BR (2017) J Chem Sci 129:1483
Afkhami A, Soltani-Felehgari F, Madrakian T, Ghaedi H, Rezaeivala M (2013) Anal Chim Acta 771:21
Nourifard F, Payehghadr M, Kalhor M, Nejadali A (2015) Electroanalysis 27:2479
Izadkhah V, Farmany A, Mortazavi S (2015) J Ind Eng Chem 21:994
Afkhami A, Soltani Shahrivar M, Ghaedi H, Madrakian T (2016) Electroanalysis 28:296
Fathi S, Yaftian M (2009) J Hazard Mater 164:133
Shams E (2000) Anal Lett 33:465
Bassie T, Siraj K, Tesema TE (2013) Adv Sci Eng Med 5:275
Li J, Zhang L, Wei G, Zhang Y, Zeng Y (2015) Biosens Bioelectron 69:316
Di Masi S, Garcia Cruz A, Canfarotta F, Cowen T, Marote P, Malitesta C, Piletsky SA (2019) Chem Nano Mat 5:754
Muralikrishna S, Sureshkumar K, Varley TS, Nagaraju DH, Ramakrishnappa T (2014) Anal Methods 6:8698
Monroy M, Maceda-Veiga A, de Sostoa A (2014) Sci Total Environ 487:233
Acknowledgements
The authors appreciate the Vice-presidency of Research of the University of Zanjan for the financial support of this study.
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Tahaei, R., Shayani-Jam, H. & Yaftian, M.R. Voltammetric determination of trace copper(II), cadmium(II), and lead(II) using a Schiff base modified glassy carbon working electrode. Monatsh Chem 152, 51–59 (2021). https://doi.org/10.1007/s00706-020-02730-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00706-020-02730-2