Abstract
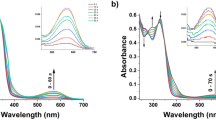
Orange-colored 7,8-dimethyl-2,3,12,13-tetramethoxy-(15H,17H)-tripyrrin-1,14-dione and its 2,3,7,8,12,13-hexaethyl analog were synthesized and converted to fluorescent derivatives by bridging a lactam nitrogen to the adjacent pyrrole nitrogen in a reaction with carbonyldiimidazole. The tripyrrindiones were also converted to the corresponding mono lactim methyl ethers by reaction with neat trimethylphosphite, and a corresponding difluoroboryl (BODIPY) derivative was made from the lactim by reaction with boron trifluoride etherate. X-ray structures of the tetramethoxytripyrrindione, the N,N′-carbonyl-bridged hexa-ethyltripyrrindione, and the lactim methyl ether of the hexa-ethyltripyrrindione were determined. The N,N′-bridged pigments exhibited very large Stokes’ shifts with fluorescence quantum yields ranging from 1.0 to 0.02.
Graphical Abstract





Similar content being viewed by others
References
Fischer H, Adler E (1931) Hoppe-Seyler’s Z physiol Chem 200:209
Fischer H, Reinecke H (1940) Hoppe-Seyler’s Z physiol Chem 265:9
von Dobeneck H, Schnierle F (1966) Tetrahedron Lett: 5327
von Dobeneck H, Messerschmitt T, Brunner E, Winderer U (1971) Liebig’s Ann Chem 751:40
Proust L (1800) Ann Chem 36:265
Simon A (1842) Anthropochemie (Berlin): 343
Pane N (1916) J Am Med Assoc 66:1433
Borrien V (1917) J Pharm et Chim 16:45
Weiss M (1930) Deutsch Arch Klin Med 166:331
Berüter J, Colombo J-P, Schlunegger UP (1975) Eur J Biochem 56:239
Yamaguchi T, Shioji I, Sugimoto A, Komoda Y, Nakajima H (1994) J Biochem 116:298
Falk H (1989) The chemistry of linear oligopyrroles and bile pigments. Springer, Vienna
Falk H, Müller N (1985) Org Magn Res 23:353
Roth SD, Shkindel T, Lightner DA (2007) Tetrahedron 63:11030
Falk H, Neufingerl F (1979) Monatsh Chem 110:987
Brower JO, Lightner DA (2002) J Org Chem 67:2713
Boiadjiev SE, Lightner DA (2008) Monatsh Chem 139:503
Loudet A, Burgess K (2007) Chem Rev 107:4891
Falk H, Leodolter A, Schade G (1978) Monatsh Chem 109:183
vos de Wael E, Pardoen JA, van Koeveringe KA, Lugtenburg J (1977) Recl Trav Chim Pays-Bas 96:306
Falk H, Gergely S, Grubmayr K, Hofer O (1977) Z Naturforsch 32b:299
Falk H, Grubmayr K, Neufingerl F (1979) Monatsh Chem 110:1127
Dey SK, Lightner DA (2007) Monatsh Chem 138:687
Fischer H, Hofelman H (1938) Liebig’s Ann Chem 533:216
Falk H, Flödl H (1985) Monatsh Chem 116:1177
Tardieux C, Bolze F, Gros CP, Guilard R (1998) Synthesis 267
Falk H, Hofer O, Lehner H (1974) Monatsh Chem 105:169
Datta S, Lightner DA (2008) Monatsh Chem 139:1519
Huggins MT, Lightner DA (2001) Monatsh Chem 132:203
Dey SK, Lightner DA (2008) Monatsh Chem 139:1377
Sheldrick GM (2003) SADABS, vers. 2.1. Bruker Analytical X-ray Systems, Madison, WI
SAINT (2003) vers 6.45. Bruker Analytical X-ray Systems, Madison, WI
Sheldrick GM (1997) SHELXT-L, vers. 6.14; Bruker Analytical X-ray Systems: Madison, WI
Acknowledgments
We thank the US National Institutes of Health (HD 17779) for generous support of this work, the National Science Foundation (CHE-0226402 and CHE-0521191) for providing funding for purchase of the X-ray diffractometer used in this work and for acquisition of a 400 MHz NMR spectrophotometer and upgrade of our 500 MHz NMR. We thank Dr. David Quilici and Ms. Rebekah Woolsey for the MALDI-TOF HRMS at the University of Nevada Proteomics Center (NIH P20 RR-016464 from the INBRE program of the National Center for Research.) We also thank Prof. T.W. Bell for use of the VPO apparatus.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Dey, S.K., Datta, S. & Lightner, D.A. Tripyrrindiones and a red-emitting fluorescent derivative. Monatsh Chem 140, 1171–1181 (2009). https://doi.org/10.1007/s00706-009-0168-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00706-009-0168-9