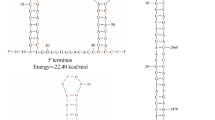
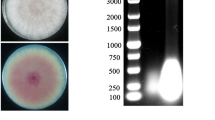
Abstract
Alternaria brassicicola is one of the causal agents of alternaria blackspot in rapeseed. In this study, a dsRNA segment was isolated and sequenced from the fungus. The complete nucleotide sequence of the dsRNA was 2506 bp in length and, using the fungal mitochondrial genetic code, was predicted to contain a single large open reading frame (ORF) in the positive strand. This ORF was predicted to encode a protein with 719 amino acids that contains characteristic conserved motifs of the RNA-dependent RNA polymerase (RdRp). BLAST analysis revealed that this protein had significant sequence similarity to the RdRp from viruses of the genus Mitovirus. These results indicated that the dsRNA segment represents the replicative form of a mitovirus, which is tentatively designated “Alternaria brassicicola mitovirus 1” (AbMV1) and is a new member of the genus Mitovirus in the family Narnaviridae.


Similar content being viewed by others
References
Ghabrial SA, Suzuki N (2009) Viruses of plant pathogenic fungi. Annu Rev Phytopathol 47:353–384
Ghabrial SA, Castón JR, Jiang D, Nibert ML, Suzuki N (2015) 50-plus years of fungal viruses. Virology 479:356–368
Xie J, Jiang D (2014) New insights into mycoviruses and exploration for the biological control of crop fungal diseases. Annu Rev Phytopathol 52:45–68
Nuss DL (1992) Biological control of chestnut blight: an example of virus-mediated attenuation of fungal pathogenesis. Microbiol Rev 56:561–576
Chiba S, Salaipeth L, Lin YH, Sasaki A, Kanematsu S, Suzuki N (2009) A novel bipartite double-stranded RNA mycovirus from the white root rot fungus Rosellinia necatrix: molecular and biological characterization, taxonomic considerations, and potential for biological control. J Virol 83:12801–12812
Yu X, Li B, Fu Y, Jiang D, Ghabrial SA, Li G, Yi X (2010) A geminivirus-related DNA mycovirus that confers hypovirulence to a plant pathogenic fungus. Proc Natl Acad Sci 107:8387–8392
Yu X, Li B, Fu Y, Xie J, Cheng J, Ghabrial SA, Jiang D (2013) Extracellular transmission of a DNA mycovirus and its use as a natural fungicide. Proc Natl Acad Sci 110:1452–1457
Liu S, Xie J, Cheng J, Li B, Chen T, Fu Y, Jiang D (2016) Fungal DNA virus infects a mycophagous insect and utilizes it as a transmission vector. Proc Natl Acad Sci 113:12803–12808
Liu L, Xie J, Cheng J, Fu Y, Li G, Yi X, Jiang D (2014) Fungal negative-stranded RNA virus that is related to bornaviruses and nyaviruses. Proc Natl Acad Sci 111:12205–12210
Zhang R, Liu S, Chiba S, Kondo H, Kanematsu S, Suzuki N (2014) A novel single-stranded RNA virus isolated from a phytopathogenic filamentous fungus, Rosellinia necatrix, with similarity to hypo-like viruses. Front Microbiol 5:360
Hillman BI, Cai G (2013) The family Narnaviridae: simplest of RNA viruses. Adv Virus Res 86:149–176
Wu M, Zhang L, Li G, Jiang D, Ghabrial SA (2010) Genome characterization of a debilitation-associated mitovirus infecting the phytopathogenic fungus Botrytis cinerea. Virology 406:117–126
Xie J, Ghabrial SA (2012) Molecular characterizations of two mitoviruses co-infecting a hyovirulent isolate of the plant pathogenic fungus Sclerotinia sclerotiorum. Virology 428:77–85
Woudenberg JHC, Groenewald JZ, Binder M, Crous PW (2013) Alternaria redefined. Stud Mycol 75:171–212
Aoki N, Moriyama H, Kodama M, Arie T, Teraoka T, Fukuhara T (2009) A novel mycovirus associated with four double-stranded RNAs affects host fungal growth in Alternaria alternata. Virus Res 140:179–187
Shepherd HS (1988) Viruslike particles in tentoxin-producing strains of Alternaria alternata. J Virol 62:3888–3891
Hayashi N, Tsuge T, Kobayashi H, Nishimura S (1988) The presence of doublestranded RNAs in Alternaria alternata Japanese pear pathotype and their participation in AK-toxin productivity. Annu Phytopathol Soc Jpn 54:250–252
Fuke K, Takeshita K, Aoki N, Fukuhara T, Egusa M, Kodama M, Moriyama H (2011) The presence of double-stranded RNAs in Alternaria alternata Japanese pear pathotype is associated with morphological changes. J Gen Virol 77:248–252
Hammond TM, Andrewski MD, Roossinck MJ, Keller NP (2008) Aspergillus mycoviruses are targets and suppressors of RNA silencing. Eukaryot Cell 7:350–357
Lin Y, Zhang H, Zhao C, Liu S, Guo L (2015) The complete genome sequence of a novel mycovirus from Alternaria longipes strain HN28. Arch Virol 160:577–580
Shang HH, Zhong J, Zhang RJ, Chen CY, Gao BD, Zhu HJ (2015) Genome sequence of a novel endornavirus from the phytopathogenic fungus Alternaria brassicicola. Arch Virol 160:1827–1830
Zhong J, Shang HH, Zhu CX, Zhu JZ, Zhu HJ, Hu Y, Gao BD (2016) Characterization of a novel single-stranded RNA virus, closely related to fusariviruses, infecting the plant pathogenic fungus Alternaria brassicicola. Virus Res 217:1–7
Morris TJ, Dodds JA (1979) Isolation and analysis of double stranded RNA from virus-infected plant and fungal tissue. Phytopathology 69:854–858
Zhong J, Lei XH, Zhu JZ, Song G, Zhang YD, Chen Y, Gao BD (2014) Detection and sequence analysis of two novel co-infecting double-strand RNA mycoviruses in Ustilaginoidea virens. Arch Virol 159:3063–307019
Altschul SF, Madden TL, Schäffer AA, Zhang J, Zhang Z, Miller W, Lipman DJ (1997) Gapped BLAST and PSI-BLAST: a new generation of protein database search programs. Nucleic Acids Res 25:3389–3402
Thompson JD, Gibson TJ, Plewniak F, Jeanmougin F, Higgins DG (1997) The CLUSTAL_X windows interface: flexible strategies for multiple sequence alignment aided by quality analysis tools. Nucleic Acids Res 25:4876–4882
Tamura K, Stecher G, Peterson D, Filipski A, Kumar S (2013) MEGA6: molecular evolutionary genetics analysis version 6.0. Mol Biol Evol 30:2725–2729
Komatsu K, Katayama Y, Omatsu T, Mizutani T, Fukuhara T, Kodama M, Moriyama H (2016) Genome sequence of a novel mitovirus identified in the phytopathogenic fungus Alternaria arborescens. Arch Virol 161:2627–2631
Marzano SYL, Domier LL (2016) Reprint of “Novel mycoviruses discovered from metatranscriptomics survey of soybean phyllosphere phytobiomes”. Virus Res 219:11–21
Xu Z, Wu S, Liu L, Cheng J, Fu Y, Jiang D, Xie J (2015) A mitovirus related to plant mitochondrial gene confers hypovirulence on the phytopathogenic fungus Sclerotinia sclerotiorum. Virus Res 197:127–136
King AM, Lefkowitz E, Adams MJ, Carstens EB (eds) (2011) Virus taxonomy: ninth report of the International Committee on Taxonomy of Viruses. Elsevier, Amsterdam
Hillman BI, Esteban R (2011) Narnaviridae. In: King AMQ, Adams MJ, Carstens EB, Lefkowitz EJ (eds) Virus taxonomy: classification and nomenclature of viruses: ninth report of the International Committee on Taxonomy of Viruses, Elsevier, San Diego, pp 1055–1060
Martínez-Álvarez P, Vainio EJ, Botella L, Hantula J, Diez JJ (2014) Three mitovirus strains infecting a single isolate of Fusarium circinatum are the first putative members of the family Narnaviridae detected in a fungus of the genus Fusarium. Arch Virol 159:2153–2155
Deng F, Boland GJ (2004) A satellite RNA of Ophiostoma novo-ulmi mitovirus 3a in hypovirulent isolates of Sclerotinia homoeocarpa. Phytopathology 94:917–923
Hong Y, Dover SL, Cole TE, Brasier CM, Buck KW (1999) Multiple Mitochondrial Viruses in an Isolate of the Dutch Elm Disease Fungus Ophiostoma novo-ulmi. Virology 258:118–127
Acknowledgements
This study is supported by Excellent Doctoral Dissertation Cultivating Fund of Hunan Agricultural University (YB2015002).
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare that they have no conflicts of interest.
Ethical approval
This article does not contain any studies with human participants or animals that were performed by any of the authors.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Chen, Y., Shang, H.H., Yang, H.Q. et al. A mitovirus isolated from the phytopathogenic fungus Alternaria brassicicola . Arch Virol 162, 2869–2874 (2017). https://doi.org/10.1007/s00705-017-3371-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00705-017-3371-8