Abstract
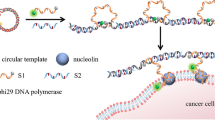
Tumor-associated antigen (TAA)–based diagnosis has gained prominence for early tumor screening, treatment monitoring, prognostic assessment, and minimal residual disease detection. However, limitations such as low sensitivity and difficulty in extracting non-specific binding membrane proteins still exist in traditional detection methods. Upconversion luminescence (UCL) exhibits unique physical and chemical properties under wavelength near-infrared light excitation. Rolling circle amplification (RCA) is an efficient DNA amplification technique with amplification factors as high as 105. Therefore, the above two excellent techniques can be employed for highly accurate imaging analysis of tumor cells. Herein, we developed a novel nanoplatform for TAA-specific cell imaging based on UCL and RCA technology. An aptamer–primer complex selectively binds to Mucin 1 (MUC1), one of TAA on cell surface, to trigger RCA reaction, generating a large number of repetitive sequences. These sequences provide lots of binding sites for complementary signal probes, producing UCL from lanthanide-doped upconversion nanoparticles (UCNPs) after releasing quencher group. The experimental results demonstrate the specific attachment of upconversion nanomaterials to cancer cells which express a high level of MUC1, indicating the potential of UCNPs and RCA in tumor imaging.
Graphical Abstract








Similar content being viewed by others
Data availability
The authors confirm that the data supporting the findings of this study are available within the article (and/or its supplementary materials).
References
Amri C, Shukla AK, Lee JH (2021) Recent advancements in nanoparticle-based optical biosensors for circulating cancer biomarkers. Materials (Basel) 240:1541
Laraib U, Sargazi S, Rahdar A, Khatami M, Pandey S (2022) Nanotechnology-based approaches for effective detection of tumor markers: a comprehensive state-of-the-art review. Int J Biol Macromol 195:356–383
Almaguel FA, Sanchez TW, Ortiz-Hernandez GL, Casiano CA (2021) Alpha-enolase: emerging tumor-associated antigen, cancer biomarker, and oncotherapeutic target. Front Genet 11:2020
Li JT, Qin B, Huang MY, Ma Y, Li DS, Li WY, Guo ZP (2021) Tumor-associated antigens (TAAs) for the serological diagnosis of osteosarcoma. Front Immunol 12:2021
Wang QT, Nie Y, Sun SN, Lin T, Han RJ, Jiang J, Li Z, Li JQ, Xiao YP, Fan YY, Yuan XH, Zhang H, Zhao BB, Zeng M, Li SY, Liao HX, Zhang J, He YW (2020) Tumor-associated antigen-based personalized dendritic cell vaccine in solid tumor patients. Cancer Immunol Immunother 69:1375–1387
Lu LG, Jiang J, Zhan MX, Zhang H, Wang QT, Sun SN, Guo XK, Yin H, Wei YD, Li SY, Liu JO, Li Y, He YW (2021) Targeting tumor-associated antigens in hepatocellular carcinoma for immunotherapy: past pitfalls and future strategies. Hepatology 73:821–832
Rashidijahanabad Z, Huang XF (2020) Recent advances in tumor associated carbohydrate antigen based chimeric antigen receptor T cells and bispecific antibodies for anti-cancer immunotherapy. Semin Immunol 47:101390
Feng C, Chen T, Mao D, Zhang F, Tian B, Zhu X (2020) Construction of a ternary complexes-based DNA logic nanomachine for highly accurate imaging analysis of cancer cells. ACS Sensors 10:3116–3123
Liu YH, Liu X, Liu HX, Wang JZ, Zhang YW, Zhao WB, Zhou JH (2022) DNA-gated N-CDs@SiO2 nanoparticles-based biosensor for MUC1 detection. Chemistryselect 7(4):e202104309
Liu X, Mao D, Deng G, Song Y, Zhang F, Yang S, Li G, Liu F, Cao W, Zhu X (2020) Nondestructive analysis of tumor-associated membrane protein MUC1 in living cells based on dual-terminal amplification of a DNA ternary complex. Theranostics 10:4410–4421
Maleki F, Rezazadeh F, Varmira K (2021) MUC1-targeted radiopharmaceuticals in cancer imaging and therapy. Mol Pharm 18:1842–1861
Wang S, Li W, Yuan D, Song J, Fang J (2016) Quantitative detection of the tumor-associated antigen large external antigen in colorectal cancer tissues and cells using quantum dot probe. Int J Nanomedicine 11:235–247
Sheng L, Cai L, Liu J, Zhang S, Xu J, Zhang X, Chen H (2017) Imaging specific newly synthesized proteins within cells by fluorescence resonance energy transfer†. Chem Sci 8:748
Ouyang Q, Yang Y, Li S, Wang L, Li H, Chen Q (2021) Upconversion nanoparticles-based FRET system for sensitive detection of Staphylococcus aureus. Spectroscopy Acta A 255:1386–1425
Su Q, Feng W, Yang D, Li F (2017) Resonance energy transfer in upconversion nanoplatforms for selective biodetection. Acc Chem Res 50:32–40
Zhou J, Liu Q, Feng W, Sun Y, Li F (2015) Upconversion luminescent materials: advances and applications. Chem Rev 115:395–465
Zhu X, Su Q, Feng W, Li F (2017) Anti-Stokes shift luminescent materials for bio-applications. Chem Soc Rev 46:1025–1039
Liu Q, Zhang Y, Peng CS, Yang T, Joubert L-M, Chu S (2018) Single upconversion nanoparticle imaging at sub-10 W cm−2 irradiance. Nat Photonics 12(9):548–553
Qiu X, Zhu X, Su X, Xu M, Yuan W, Liu Q, Xue M, Liu Y, Feng W, Li F (2019) Near-infrared upconversion luminescence and bioimaging in vivo based on quantum dots. Adv Sci (Weinh) 6:1801834
Sun Y, Liu Q, Peng J, Feng W, Zhang Y, Yang P, Li F (2013) Radioisotope post-labeling upconversion nanophosphors for in vivo quantitative tracking. Biomaterials 34:2289–2295
Zhou J, Liu Z, Li F (2012) Upconversion nanophosphors for small-animal imaging. Chem Soc Rev 41:1323–1349
Zhou J, Zhu X, Chen M, Sun Y, Li F (2012) Water-stable NaLuF4-based upconversion nanophosphors with long-term validity for multimodal lymphatic imaging. Biomaterials 33:6201–6210
Peng J, Zhao L, Zhu X, Sun Y, Feng W, Gao Y, Wang L, Li F (2013) Hollow silica nanoparticles loaded with hydrophobic phthalocyanine for near-infrared photodynamic and photothermal combination therapy. Biomaterials 34:7905–7912
Zhao L, Peng J, Chen M, Liu Y, Yao L, Feng W, Li F (2014) Yolk-shell upconversion nanocomposites for LRET sensing of cysteine/homocysteine. ACS Appl Mater Interfaces 6:11190–11197
Feng C, Mao X, Shi H, Bo B, Chen X, Chen T, Zhu X, Li G (2017) Detection of microRNA: a point-of-care testing method based on a pH-responsive and highly efficient isothermal amplification. Anal Chem 89:6631–6636
Ali MM, Li F, Zhang Z, Zhang K, Kang DK, Ankrum JA, Le XC, Zhao W (2014) Rolling circle amplification: a versatile tool for chemical biology, materials science and medicine. Chem Soc Rev 43:3324–3341
Deng R, Tang L, Tian Q, Wang Y, Lin L, Li J (2014) Toehold-initiated rolling circle amplification for visualizing individual microRNAs in situ in single cells. Angew Chem Int Ed Engl 53:2389–2393
Huang R, He L, Li S, Liu H, Jin L, Chen Z, Zhao Y, Li Z, Deng Y, He N (2020) A simple fluorescence aptasensor for gastric cancer exosome detection based on branched rolling circle amplification. Nanoscale 12:2445–2451
Mohsen MG, Kool ET (2016) The discovery of rolling circle amplification and rolling circle transcription. Acc Chem Res 49:2540–2550
Yi G, Chow G (2006) Synthesis of hexagonal-phase NaYF4:Yb,Er and NaYF4:Yb,Tm nanocrystals with efficient up-conversion fluorescence. Adv Funct Mater 16:2324–2329
Xu L, Duan J, Chen J, Ding S, Cheng W (2021) Recent advances in rolling circle amplification-based biosensing strategies-a review. Anal Chim Acta 1148:238187
Shen B, Cheng S, Gu Y, Ni D, Gao Y, Su Q, Feng W, Li F (2017) Revisiting the optimized doping ratio in core/shell nanostructured upconversion particles. Nanoscale 9:1964–1971
Zhu X, Li J, Qiu X, Liu Y, Feng W, Li F (2018) Upconversion nanocomposite for programming combination cancer therapy by precise control of microscopic temperature. Nat Commun 9:2176
Cheng L, Yang K, Shao M, Lu X, Liu Z (2011) In vivo pharmacokinetics, long-term biodistribution and toxicology study of functionalized upconversion nanoparticles in mice. Nanomedicine 6:1327–1340
Funding
This work was supported by the National Natural Science Foundation of China (Grant Nos. 32000986, 82070518, 82102477, and 82370516).
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Ethics approval
This research did not involve human or animal samples.
Conflict of interest
The authors declare no competing interests.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
ESM 1
(DOCX 3571 kb)
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Zhan, Y., Mao, Y., Sun, P. et al. Tumor-associated antigen-specific cell imaging based on upconversion luminescence and nucleic acid rolling circle amplification. Microchim Acta 191, 248 (2024). https://doi.org/10.1007/s00604-024-06331-2
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s00604-024-06331-2