Abstract
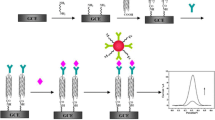
Micro-sized glassy carbon microspheres (GCMs, typically 3 μm in diameter) instead of nano-sized gold nanoparticles (AuNPs, typically 20 nm in diameter) were for the first time used as signal markers for the quantitative detection of antigen such as prostate-specific antigen (PSA). After being treated with concentrated HNO3, GCMs bear carboxyl groups at their surfaces, which enables antibodies to be conjugated with GCMs to yield new type of micro-sized material-based colorimetric probes used for immunochromatographic test strips (ICTSs). The captured black GCMs (with strong and wide-band light absorption) on the T-line of ICTS were used both for qualitative and quantitative determination of PSA. In the case of quantitative determination, a lab-assembled optical strip reader system was used to measure the reflected LED light intensity at 550 nm. The sensing performances of the developed GCM-based ICTSs, such as sensitivity, selectivity, reproducibility, stability, and applicability, were investigated in detail. The developed GCM-based ICTSs can have much higher (3 times) detection sensitivity than AuNP-based ICTSs, showing promising applications in sensitive immunoassay.
Graphical abstract







Similar content being viewed by others
References
Hanahan D, Weinberg RA (2011) Hallmarks of cancer: the next generation. Cell 144(5):646–674
Sturgeon C (2002) Practice guidelines for tumor marker use in the clinic. Clin Chem 48(8):1151–1159
Faraggi D, Kramar A (2000) Methodological issues associated with tumor marker development: biostatistical aspects. Urol Oncol 5(5):211–213
Shiomi K, Hagiwara Y, Sonoue K, Segawa T, Miyashita K, Maeda M, Izumi H, Masuda K, Hirabayashi M, Moroboshi T, Yoshiyama T, Ishida A, Natori Y, Inoue A, Kobayashi M, Sakao Y, Miyamoto H, Takahashi K, Hino O (2008) Sensitive and specific new enzyme-linked immunosorbent assay for N-ERC/mesothelin increases its potential as a useful serum tumor marker for mesothelioma. Clin Cancer Res 14(5):1431–1437
Gordon SG, Cross BA (1990) An enzyme-linked immunosorbent assay for cancer procoagulant and its potential as a new tumor marker. Cancer Res 50(19):6229–6234
Waldmann TA, Mclntire KR (1974) The use of a radioimmunoassay for alpha-fetoprotein in the diagnosis of malignancy. Cancer 34:1510–1515
Ferroni P, Szpak C, Greiner JW, Simpson JF, Guadagni F, Johnston WW, Colcher D (1990) Ca 72-4 radioimmunoassay in the diagnosis of malignant effusions. Comparison of various tumor markers. Int J Cancer 46(3):445–451
Chen Z, Wu Y, Chen M, Hou JY, Ren ZQ, Sun D, Liu TC (2013) A novel homogeneous time-resolved fluoroimmunoassay for carcinoembryonic antigen based on water-soluble quantum dots. J Fluoresc 23(4):649–657
He Y, Li Y, Hun X (2010) Polymer nanoparticles as fluorescent labels in a fluoroimmunoassay for human chorionic gonadotropin. Microchim Acta 171(3-4):393–398
Zong C, Wu J, Wang C, Ju H, Yan F (2012) Chemiluminescence imaging immunoassay of multiple tumor markers for cancer screening. Anal Chem 84(5):2410–2415
Fu Z, Liu H, Ju H (2006)Flow-through multianalyte chemiluminescent immunosensing system with designed substrate zone-resolved technique for sequential detection of tumor markers. Anal Chem 78(19):6999–7005
Liu Z, Rong Q, Ma Z, Han H (2015)One-step synthesis of redox-activepolymer/AU nanocomposites for electrochemical immunoassay of multiplexed tumor markers. Biosens Bioelectron 65:307–313
Wilson MS (2005) Electrochemical immunosensors for the simultaneous detection of two tumor markers. Anal Chem 77(5):1496–1502
Bahadır EB, Sezgintürk MK (2016) Lateral flow assays: principles, designs and labels. TRAC-Trend Anal Chem 82:286–306
Huang X, Aguilar ZP, Xu H, Lai W, Xiong Y (2016)Membrane-based lateral flow immunochromatographic strip with nanoparticles as reporters for detection: a review. Biosens Bioelectron 75:166–180
Quesada-González D, Merkoci A (2015)Nanoparticle-based lateral flow biosensors. Biosens Bioelectron 73:47–63
Sun J, Lu Y, He L, Pang J, Yang F, Liu Y (2020) Colorimetric sensor array based on gold nanoparticles: design principles and recent advances. eTrAC Trend Anal Chem 122:115754
Khlebtsov BN, Tumskiy RS, Burov AM, Pylaev TE, Khlebtsov NG (2019) Quantifying the numbers of gold nanoparticles in the test zone of lateral flow immunoassay strips. ACS Appl Nano Mater 2:5020–5028
Wang R, Huang Y, Chen Y, Chi Y (2020) Electrochemiluminescence from the graphene- and fullerene-like nanostructures of glassy carbon microspheres and its application in immunoassay. ACS Appl Bio Mater 3:6358–6367
Zhou J, Zhuang J, Miro M, Gao Z, Chen G, Tang D (2012) Carbon nanospheres-promoted electrochemical immunoassay coupled with hollow platinum nanolabels for sensitivity enhancement. Biosens Bioelectron 35:394–400
Lin S, Zheng D, Li A, Chi Y (2019) Black oxidized 3,3′,5,5′-tetramethylbenzidine nanowires (oxTMB NWs) for enhancing Pt nanoparticle-based strip immunosensing. Anal Bioanal Chem 411:4063–4071
Zhou C, Chen Y, Shang P, Chi Y (2016) Strong electrochemiluminescent interactions between carbon nitride nanosheet–reduced graphene oxide nanohybrids and folic acid, and ultrasensitive sensing for folic acid. Analyst 141:3379–3388
Funding
This study was financially supported by the National Natural Science Foundation of China (22074018) and the Program for Scientific and Technological Innovation Leading Talents in Fujian Province.
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare that they have no competing interests.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
ESM 1
(DOCX 771 kb)
Rights and permissions
About this article
Cite this article
Lin, S., Zhong, J., Chi, Y. et al. Colorimetric immunosensor based on glassy carbon microspheres test strips for the detection of prostate-specific antigen. Microchim Acta 188, 366 (2021). https://doi.org/10.1007/s00604-021-04907-w
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s00604-021-04907-w