Abstract
Silver nanoparticles (AgNPs) are widely used in industrial and consumer products owing to its antimicrobial nature and multiple applications. Consequently, their release into the environment is becoming a big concern because of their negative impacts on living organisms. In this work, AgNPs were detected at a potential of + 0.70 V vs. Ag/AgCl reference electrode, characterized, and quantified in consumer products by particle collision coulometry (PCC). The electrochemical results were compared with those measured with electron microscopy and single-particle inductively coupled plasma mass spectrometry. The theoretical and practical peculiarities of the application of PCC technique in the characterization of AgNPs were studied. Reproducible size distributions of the AgNPs were measured in a range 10–100 nm diameters. A power allometric function model was found between the frequency of the AgNPs collisions onto the electrode surface and the number concentration of nanoparticles up to a silver concentration of 1010 L−1 (ca. 25 ng L−1 for 10 nm AgNPs). A linear relationship between the number of collisions and the number concentration of silver nanoparticles was observed up to 5 × 107 L−1. The PCC method was applied to the quantification and size determination of the AgNPs in three-silver containing consumer products (a natural antibiotic and two food supplements). The mean of the size distributions (of the order 10–20 nm diameters) agrees with those measured by electron microscopy.
Graphical abstract

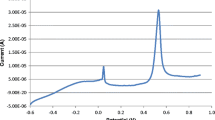
The areas of current spikes from the chronoamperogram allow the rapid calculation of size distributions of AgNPs that impact onto the working electrode.





Similar content being viewed by others
References
Haider A, Kang IK (2015) Preparation of silver nanoparticles and their industrial and biomedical applications: a comprehensive review. Adv Mater Sci Eng 2015:1–16. https://doi.org/10.1155/2015/165257
Zhang W, Xiao B, Fang T (2018) Chemical transformation of silver nanoparticles in aquatic environments: mechanism, morphology and toxicity. Chemosphere 191:324–334. https://doi.org/10.1016/j.chemosphere.2017.10.016
Marambio-Jones C, Hoek EMV (2010) A review of the antibacterial effects of silver nanomaterials and potential implications for human health and the environment. J Nanopart Res 12:1531–1551. https://doi.org/10.1007/s11051-010-9900-y
Bhatt I, Tripathi BNI (2011) Interaction of engineered nanoparticles with various components of the environment and possible strategies for their risk assessment. Chemosphere 82:308–317. https://doi.org/10.1016/j.chemosphere.2010.10.011
Stebounova LV, Guio E, Grassian VH (2011) Silver nanoparticles in simulated biological media: a study of aggregation, sedimentation, and dissolution. J Nanopart Res 13:233–244. https://doi.org/10.1007/s11051-010-0022-3
Laborda F, Bolea E, Cepriá G, Gómez MT, Jiménez MS, Pérez-Arantegui J, Castillo JR (2016) Detection, characterization and quantification of inorganic engineered nanomaterials: a review of techniques and methodological approaches for the analysis of complex samples. Anal Chim Acta 904:10–32. https://doi.org/10.1016/j.aca.2015.11.008
Calzolai L, Gilliland D, Rossi F (2012) Measuring nanoparticles size distribution in food and consumer products: a review. Food Addit Contam Part A Chem Anal Control Expo Risk Assess 29:1183–1193. https://doi.org/10.1080/19440049.2012.689777
Laborda F, Bolea E, Jiménez-Lamana J (2014) Single particle inductively coupled plasma mass spectrometry: a powerful tool for nanoanalysis. Anal Chem 86:2270–2278. https://doi.org/10.1021/ac402980q
Batchelor-McAuley C, Kätelhön E, Barnes EO, Compton RG, Laborda E, Molina A (2015) Recent advances in voltammetry. ChemistryOpen 4:224–260. https://doi.org/10.1002/open.201500042
Bettazi F, Palchetti I (2019) Nanotoxicity assessment: a challenging application for cutting edge electroanalytical tools. Anal Chim Acta 1072:61–74. https://doi.org/10.1016/j.aca.2019.04.035
Cheng W, Compton RG (2014) Electrochemical detection of nanoparticles by ‘nano-impact’ methods. TrAC Trends Anal Chem 58:79–89. https://doi.org/10.1016/j.trac.2014.01.008
Stuart EJE, Zhou YG, Rees NV, Compton RG (2012) Determining unknown concentrations of nanoparticles: the particle-impact electrochemistry of nickel and silver. RSC Adv 2:6879–6884. https://doi.org/10.1039/C2RA20628E
Rees NV (2014) Electrochemical insight from nanoparticle collisions with electrodes: a mini-review. Electrochem Commun 43:83–86. https://doi.org/10.1016/j.elecom.2014.03.018
Zhou YG, Rees NV, Compton RG (2011) The electrochemical detection and characterization of silver nanoparticles in aqueous solution. Angew Chem 50:4219–4221. https://doi.org/10.1002/anie.201100885
Stuart EJE, Rees NV, Cullen JT, Compton RG (2013) Direct electrochemical detection and sizing of silver nanoparticles in seawater media. Nanoscale 5:174–177. https://doi.org/10.1039/C2NR33146B
Boika A, Bard AJ (2015) Time of first arrival in electrochemical collision experiments as a measure of ultra low concentrations of analytes in solution. Anal Chem 87:4341–4346. https://doi.org/10.1021/acs.analchem.5b00037
Sokolov SV, Bartlett TR, Fair P, Fletcher S, Compton RG (2016) Femtomolar detection of silver nanoparticles by flow-enhanced direct-impact voltammetry at a microelectrode array. Anal Chem 88:8908–8912. https://doi.org/10.1021/acs.analchem.6b02670
Rees NV, Zhou YG, Compton RG (2011) The aggregation of silver nanoparticles in aqueous solution investigated via anodic particle coulometry. ChemPhysChem 12:1645–1647. https://doi.org/10.1002/cphc.201100207
Hernández D, Cepriá G, Laborda F, Castillo JR (2019) Detection and determination of released ions in the presence of nanoparticles : selectivity or strategy? Electroanalysis 31:405–410. https://doi.org/10.1002/elan.201800597
Ngamchuea K, Clark ROD, Sokolov SV, Young NP, Batchelor-McAuley C, Compton RG (2017) Single oxidative collision events of silver nanoparticles: understanding the rate-determining chemistry. Chem Eur J 23:16085–16096. https://doi.org/10.1002/chem.201703591
Little CA, Xie R, Batchelor-McAuley C, Käthelön E, Li X, Young NP, Compton RG (2018) A quantitative methodology for the study of particle-electrode impacts. Phys Chem Chem Phys 20:13537–13546. https://doi.org/10.1039/C8CP01561A
Toh HS, Jurkschat K, Compton RG (2015) The influence of the capping agent on the oxidation of silver nanoparticles: nano-impacts versus stripping voltammetry. Chem Eur J 21:2998–3004. https://doi.org/10.1002/chem.201406278
Eloul S, Kätelhön E, Batchelor-McAuley C, Tschulik K, Compton RG (2015) Diffusional impacts of nanoparticles on microdisc and microwire electrodes: the limit of detection and first passage statistics. J Electroanal Chem 755:136–142. https://doi.org/10.1016/j.jelechem.2015.07.042
Abad-Álvaro I, Peña-Vázquez E, Bolea E, Bermejo-Barrera P, Castillo JR, Laborda F (2016) Evaluation of number concentration quantification by single-particle inductively coupled plasma mass spectrometry: microsecond vs. millisecond dwell times. Anal Bioanal Chem 408:5089–5097. https://doi.org/10.1007/s00216-016-9515-y
Funding
This work has been supported by the Ministry of Science, Innovation, and Universities and the European Regional Development Fund project RTI2018-096111-B-I00 (MICINN/FEDER) and by the Government of Aragon (E29_17R), and co-financed with FEDER 2014-2020. The work has been also 65% cofinanced by the European Regional Development Fund (ERDF) through the Interreg VA Spain-France-Andorra programme (POCTEFA 2014-2020), Project OUTBIOTICS EFA183/16.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
In memoriam of Prof. Juan R. Castillo
Supplementary Information
ESM 1
(DOCX 959 kb).
Rights and permissions
About this article
Cite this article
Hernández, D., Vidal, J.C., Laborda, F. et al. Detection, size characterization and quantification of silver nanoparticles in consumer products by particle collision coulometry. Microchim Acta 188, 12 (2021). https://doi.org/10.1007/s00604-020-04662-4
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s00604-020-04662-4