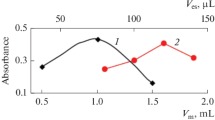
Abstract
A method is presented for the preconcentration of Cu(II) ions using a nanometer-sized TiO2 colloid, and for their determination in environmental water using graphite furnace atomic adsorption spectrometry (GFAAS) with slurry sampling. The adsorption efficiency is above 97% and is reached within short time at pH values between 5.5 and 6.0. Cu(II) was determined by GFAAS without elution. Conditions such as colloid dosage and effects of other ions were studied. The detection limit (3 σ) and the relative standard deviation are 290 ng L-1 and 9.8% (n = 6), respectively. This simple and rapid method was successfully applied to spiked environmental samples with recoveries ranging from 97% – 104%.

Similar content being viewed by others
References
Beppu MM, Arruda EJ, Vieira RS, Santos NN (2004) Adsorption of Cu(II) on Porous Chitosan Membranes Functionalized with Histidine. Journal of Membrane Science 240:227
Komjarova I, Blust R (2006) Comparison of liquid-liquid extraction, solid-phase extraction and co-precipitation preconcentration methods for the determination of cadmium, copper, nickel, lead and zinc in seawater. Analytica Chimica Acta 576:221
Brasil JL, Martins LC, Ev RR, Dupont J, Dias SLP, Sales JAA, Airoldi C, Lima ÉC (2005) Factorial design for optimization of flow-injection preconcentration procedure for copper(II) determination in natural waters, using 2-aminomethylpyridine grafted silica gel as adsorbent and spectrophotometric detection. International Journal of Environmental Analytical Chemistry 85(7):475
Lemos VA, Baliza PX, Yamaki RT, Rocha ME, Paula A, Aves O (2003) Synthesis and application of a functionalized resin in on-line system for copper preconcentration and determination in foods by flame atomic absorption spectrometry. Talanta 61:675
Roco MC (1999) Nanoparticles and nanotechnology research. Journal of Nanoparticle Research 1(1):1
Uehara M, Barbara B, Dieny B, Stamp PCE (1986) Staircase behaviour in the magnetization reversal of a chemically disordered magnet at low temperature. Physics letters. A. 114(1):23
Hada H, Tanemura H, Saikawa M (1982) Photoreduction of silver iron in a Titannium dioxide suspension. Bull Chem.Soc Jan. 55(7):2010
Vassileva E, Varimezova B, Hadjiivanov K (1996) Column solid-phase extraction of heavy metal ions on a high surface area CeO2 as a preconcentration method for trace determination. Anal. Chim. Acta 336:141
Li SX, Deng NS (2002) Separation and preconcentration of Se(IV)/Se(VI) species by selective absorption onto nanometer-sized titanium dioxide and determination by graphite furnace atomic absorption spectrometry. Anal. Bioanal. Chem. 374:1341
Li SX, Deng NS, Zheng FY, Huang YZ (2003) Spectrophotometric determination of tungsten(VI) enriched by nanometer-size titanium dioxide in water and sediment. Talanta 60:1097
Liang P, Shi TQ, Lu HB, Jiang ZC, Hu B (2003) Speciation of Cr(III) and Cr(VI) by nanometer titanium dioxide micro-column and inductively coupled plasma atomic emission spectrometry. Spectrochimica Acta Part B 58:1709
Liang P, Qin YC, Peng HuB, TY JZC (2001) Nanometer-size titanium dioxide microcolumn on-line preconcentration of trace metals and their determination by inductively coupled plasma atomic emission spectrometry in water. Analytica Chimica Acta 440:207
Liang P, Li Q, Liu R (2009) Determination of trace molybdenum in biological and water samples by graphite furnace atomic absorption spectrometry after separation and preconcentration on immobilized titanium dioxide nanoparticles. Microchimica Acta 164:119
Liu Y, Liang P, Guo L (2005) Nanometer titanium dioxide immobilized on silica gel as sorbent for preconcentration of metal ions prior to their determination by inductively coupled plasma atomic emission spectrometry. Talanta 68:25
Liu R, Liang P (2007) Determination of gold by nanometer titanium dioxide immobilized on silica gel packed microcolumn and flame atomic adsorption spectrometry in geological and water samples. Anal. Chim. Acta 604:114
Huang CZ, Jiang ZC, Hu B (2007) Mesoporous titanium dioxide as a novel solid-phase extraction material for flow injection micro-column preconcentration on-line coupled with ICP-OES determination of trace metals in environmental samples. Talanta 73:274
Zheng FY, Qian SH, Li SX, Huang XQ, Lin LX (2006) Speciation Analysis of Antimony by Preconcentration of Sb(III) and Sb(V) in Water Samples onto Nanometer-size Titanium Dioxide and Selective Determination by Flow Injection-Hydride Generation-Atomic Absorption Spectrometry. Analytical Sciences 22:1319
Qian SH, Luo Y, Mo SB, Weng Y (2006) Application of Nanometer-Size Titanium Dioxide in Extreme-Trace V(V) Analysis. Wuhan University Journal of Natural Science 11(2):437
Xiao YB, Ling J, Qian SH (2007) Preconcentration of Trace Arsenite and Arsenate with Titanium Dioxide Nanoparticles and Subsequent Determination by Silver Diethyldithiocarbamate Spectrophotometric Method. Water Environment Research 79(9):1015
Qian SH, Lin H, Li XQ, Xiao M, Deng HB, Xiang LJ (2007) Preconcentration and Separation of Trace Copper in Water Samples with Nanometer-size TiO2 Colloid and Determination by FAAS. Wuhan University Journal of Natural Science 12(2):349
Qian SH, Li XQ, Lin H, Xiao M, Deng HB, Xiang LJ (2006) Preconcentration of Ultra-trace Cadmium with Nanometer-size TiO2 Colloid and Determination by GFAAS with Slurry Sampling. Chinese Chemical Letters 17(7):933
Aline SL, Marco AZA (2009) Determination of tin and lead in sediment slurries by graphite furnace atomic absorption spectrometry. Microchimica Acta 164:445
Chen H, Hu W, Li SQ, Wang M (2008) Direct determination of Cd and Pb in gel forming konjac samples by enzymatic hydrolysis assisted slurry sampling graphite furnace atomic absorption spectrometry. Microchimica Acta 162:133
Acknowledgements
This research was financially supported by the National Natural Science Foundation of China (Grant No. 20877059).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Qian, S., Zhang, S., Huang, Z. et al. Preconcentration of ultra-trace copper in water samples with nanometer-size TiO2 colloid and determination by GFAAS with slurry sampling. Microchim Acta 166, 251–254 (2009). https://doi.org/10.1007/s00604-009-0191-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00604-009-0191-x