Abstract
Aims
While there have been many outcome-focussed studies examining insulin pump therapy, only a few have looked at potential adverse events (AEs), with none examining the relationship between AEs and pump/infusion set type, ethnicity or socio-economic status. In addition, current data on the incidence and characteristics of pump-associated AEs are confined to one paediatric centre. We aimed to describe the incidence, characteristics and potential predictors of insulin pump-associated AEs in New Zealand adults and children with T1DM.
Methods
We approached adults and families of children with T1DM on insulin pumps in four main New Zealand centres. Participants completed a questionnaire examining pump-related issues they had experienced in the preceding 12 months.
Results
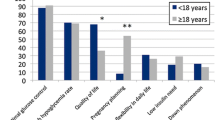
Response rate was 64 % with 174 of 270 eligible people participating in the study. 84 % of subjects reported one or more AEs, with an overall AE incidence of 3.42 per person/year (95 % CI 3.14, 3.73). An event serious enough to require a hospital presentation occurred in 9.8 %, all but one reporting high ketones or diabetic ketoacidosis (DKA). Set/site problems were the AE most commonly reported (by 53 % of respondents), followed by cutaneous complications (43 %) and pump malfunction (38 %). Few predictors of AEs (of any type) were found; however, a negative binomial regression model found that a longer duration of pumping (p = 0.018) and age <18 years (p = 0.043) were both associated with fewer AEs (all types combined).
Conclusions
Insulin pump-associated AEs are very common. However, few variables are predictive of them with no relationships seen with glycaemic control, socio-economic status, pump manufacturer or infusion set type. Based on these findings, AEs should be anticipated in both adults and children, with anticipatory patient education and training recommended for their successful and safe use.
Similar content being viewed by others
References
The Diabetes Control and Complications Trial Research Group (1993) The effect of intensive treatment of diabetes on the development and progression of long-term complications in insulin-dependent diabetes mellitus. N Engl J Med 329(14):977–986
Nathan DM, Cleary PA, Backlund JY, Genuth SM, Lachin JM, Orchard TJ et al (2005) Intensive diabetes treatment and cardiovascular disease in patients with type 1 diabetes. N England J Med 353(25):2643–2653
Craig ME, Twigg SM, Donaghue KC et al (2011) For the Australian type 1 diabetes guidelines expert advisory group. national evidence-based clinical care guidelines for type 1 diabetes in children, adolescents and adults. Australian Government Department of Health and Ageing, Australia
Misso Marie L, Egberts Kristine J, Page M, O’Connor D, Shaw J (2010) Continuous subcutaneous insulin infusion (CSII) versus multiple insulin injections for type 1 diabetes mellitus. cochrane database of systematic reviews (Internet).(1).http://onlinelibrary.wiley.com/doi/10.1002/14651858.CD005103.pub2/abstract, http://onlinelibrary.wiley.com/store/10.1002/14651858.CD005103.pub2/asset/CD005103.pdf?v=1&t=i259baf5&s=70d9e69fdda6c77de30f228eb9a794ce82608151
Pankowska E, Blazik M, Dziechciarz P, Szypowska A, Szajewska H (2009) Continuous subcutaneous insulin infusion vs. multiple daily injections in children with type 1 diabetes: a systematic review and meta-analysis of randomized control trials. Pediatr Diabetes 10(1):52–58
Bolli GB, Kerr D, Thomas R, Torlone E, Sola-Gazagnes A, Vitacolonna E et al (2009) Comparison of a multiple daily insulin injection regimen (basal once-daily glargine plus mealtime lispro) and continuous subcutaneous insulin infusion (lispro) in type 1 diabetes: a randomized open parallel multicenter study. Diabetes Care 32(7):1170–1176
Cooper MN, O’Connell SM, Davis EA, Jones TW (2013) A population-based study of risk factors for severe hypoglycaemia in a contemporary cohort of childhood-onset type 1 diabetes. Diabetologia 56(10):2164–2170
Wheeler BJ, Heels K, Donaghue KC, Reith DM, Ambler GR (2014) Insulin pump-associated adverse events in children and adolescents—a prospective study. Diabetes Tech Ther 16(9):558–562
Cope JU, Samuels-Reid JH, Morrison AE (2012) Pediatric use of insulin pump technology: a retrospective study of adverse events in children ages 1–12 years. J Diabetes Sci Tech 6(5):1053–1059
Guilhem I, Balkau B, Lecordier F, Malecot JM, Elbadii S, Leguerrier AM et al (2009) Insulin pump failures are still frequent: a prospective study over 6 years from 2001 to 2007. Diabetologia 52(12):2662–2664
Pickup JC, Yemane N, Brackenridge A, Pender S (2014) Nonmetabolic complications of continuous subcutaneous insulin infusion: a patient survey. Diabetes Tech Ther 16(3):145–149
Wheeler BJ, Donaghue KC, Heels K, Ambler GR (2014) Family perceptions of insulin pump adverse events in children and adolescents. Diabetes Tech Therapeutics 16(4):204–207
Ross PL, Milburn J, Reith DM, Wiltshire E, Wheeler BJ (2015) Clinical review: insulin pump-associated adverse events in adults and children. Acta Diabetol 52(6):1017–1024
Salmond C, Crampton P, Atkinson J (2007) NZDep2006 Index of deprivation. University of Otago, Wellington
Peduzzi P, Concato J, Kemper E, Holford TR, Feinstein AR (1996) A simulation study of the number of events per variable in logistic regression analysis. J Clin Epidemiol 49(12):1373–1379
Mecklenburg RS, Benson EA, Benson JW Jr, Fredlund PN, Guinn T, Metz RJ et al (1984) Acute complications associated with insulin infusion pump therapy. Report of experience with 161 patients. JAMA, J Am Med Assoc 252(23):3265–3269
Acknowledgments
The authors wish to thank all the individuals and families involved in the study, and all the staff from the collaborating diabetes centres for their time and assistance with this study. The Otago Medical Research Foundation and the University of Otago provided financial support for this project.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Ethical standard
Ethical approval for this study was granted by the University of Otago Human Ethics Committee (H14/089).
Human and animal rights
All procedures followed were in accordance with the ethical standards of the responsible committee on human experimentation (institutional and national) and with the Helsinki Declaration of 1975, as revised in 2008.
Informed consent
Informed consent was obtained from all patients for being included in the study.
Additional information
Managed by Massimo Porta.
Rights and permissions
About this article
Cite this article
Ross, P., Gray, A., Milburn, J. et al. Insulin pump-associated adverse events are common, but not associated with glycemic control, socio-economic status, or pump/infusion set type. Acta Diabetol 53, 991–998 (2016). https://doi.org/10.1007/s00592-016-0897-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00592-016-0897-2