Abstract
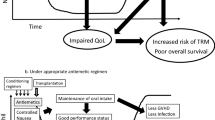
The goal of this work was to evaluate the efficacy of 5-HT3 receptor antagonist regimens in the transplant setting through a retrospective analysis of clinic patients and a review of the literature. A retrospective study was performed to evaluate the efficacy of an antiemetic regimen in 24 patients receiving high-dose chemotherapy with bone marrow transplantation. The antiemetic agent used in 96% of the patients was single-agent ondansetron 0.15 mg/kg as a continuous infusion every 12 h starting on the first day of chemotherapy. Efficacy or failure was defined by the need for administration of rescue antiemetics, as documented by nurses' notes and pharmacy records. Overall, 23 (96%) patients experienced antiemetic failure within the first 3 days. Rescue antiemetics yielded improved control of nausea and vomiting in 16 of 23 (70%) patients. These data demonstrate poor antiemetic efficacy of single-agent, continuous-infusion ondansetron in a small group of patients receiving high-dose chemotherapy with bone marrow transplantation. These data are considered in the context of a thorough literature review that shows the limitations of previously reported antiemetic studies in this setting, potential pharmacokinetic interactions, and also highlights the utilization of combination therapy, administered orally or intravenously, to improve efficacy and tolerability of antiemetic therapy. A number of recent investigations suggest that appropriate doses of 5-HT3 receptor antagonists with phenothiazine and corticosteroids can improve the safety and efficacy of antiemetic regimens in patients undergoing transplantation.
Similar content being viewed by others
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Perez, E., Tiemeier, T. & Solberg, L. Antiemetic therapy for high-dose chemotherapy with transplantation: report of a retrospective analysis of a 5-HT3 regimen and literature review. Support Care Cancer 7, 413–424 (1999). https://doi.org/10.1007/s005200050302
Issue Date:
DOI: https://doi.org/10.1007/s005200050302