Abstract
Purpose
One plausible mechanistic hypothesis is the potential contribution of inflammatory mechanisms to shortness of breath. This study was aimed to evaluate for associations between the occurrence of shortness of breath and perturbations in inflammatory pathways.
Methods
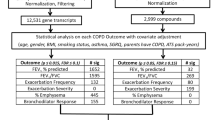
Patients with cancer reported the occurrence of shortness of breath six times over two cycles of chemotherapy. Latent class analysis was used to identify subgroups of patients with distinct shortness of breath occurrence profiles (i.e., none (70.5%), decreasing (8.2%), increasing (7.8%), high (13.5%)). Using an extreme phenotype approach, whole transcriptome differential gene expression and pathway impact analyses were performed to evaluate for perturbed signaling pathways associated with shortness of breath between the none and high classes. Two independent samples (RNA-sequencing (n = 293) and microarray (n = 295) methodologies) were evaluated. Fisher’s combined probability method was used to combine these results to obtain a global test of the null hypothesis. In addition, an unweighted knowledge network was created using the specific pathway maps to evaluate for interconnections among these pathways.
Results
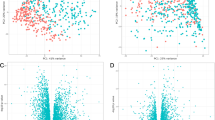
Twenty-nine Kyoto Encyclopedia of Genes and Genomes inflammatory signaling pathways were perturbed. The mitogen-activated protein kinase signaling pathway node had the highest closeness, betweenness, and degree scores. In addition, five common respiratory disease-related pathways, that may share mechanisms with cancer-related shortness of breath, were perturbed.
Conclusions
Findings provide preliminary support for the hypothesis that inflammation contribute to the occurrence of shortness of breath in patients with cancer. In addition, the mechanisms that underlie shortness of breath in oncology patients may be similar to other respiratory diseases.

Similar content being viewed by others
Data availability
Data will be provided to the publisher after they obtain a material transfer agreement from the University of California, San Francisco. Individuals who would like a copy of the survey can contact the corresponding author.
References
Hui D, Bohlke K, Bao T, Campbell TC, Coyne PJ, Currow DC, Gupta A, Leiser AL, Mori M, Nava S, Reinke LF, Roeland EJ, Seigel C, Walsh D, Campbell ML (2021) Management of dyspnea in advanced cancer: ASCO guideline. J Clin Oncol 39(12):1389–1411. https://doi.org/10.1200/JCO.20.03465
Undem BJ, Nassenstein C (2009) Airway nerves and dyspnea associated with inflammatory airway disease. Respir Physiol Neurobiol 167(1):36–44. https://doi.org/10.1016/j.resp.2008.11.012
Barnes PJ, Celli BR (2009) Systemic manifestations and comorbidities of COPD. Eur Respir J 33(5):1165. https://doi.org/10.1183/09031936.00128008
Bracci L, Schiavoni G, Sistigu A, Belardelli F (2014) Immune-based mechanisms of cytotoxic chemotherapy: implications for the design of novel and rationale-based combined treatments against cancer. Cell Death Differ 21(1):15–25. https://doi.org/10.1038/cdd.2013.67
Chen L, Deng H, Cui H, Fang J, Zuo Z, Deng J, Li Y, Wang X, Zhao L (2018) Inflammatory responses and inflammation-associated diseases in organs. Oncotarget 9(6):7204–7218. https://doi.org/10.18632/oncotarget.23208
Rivera MP, Detterbeck FC, Socinski MA, Moore DT, Edelman MJ, Jahan TM, Ansari RH, Luketich JD, Peng G, Monberg M, Obasaju CK, Gralla RJ (2009) Impact of preoperative chemotherapy on pulmonary function tests in resectable early-stage non-small cell lung cancer. Chest 135(6):1588–1595. https://doi.org/10.1378/chest.08-1430
Ji Y, Li M, Chang M, Liu R, Qiu J, Wang K, Deng C, Shen Y, Zhu J, Wang W, Xu L, Sun H (2022) Inflammation: roles in skeletal muscle atrophy. Antioxidants (Basel) 11(9):1686. https://doi.org/10.3390/antiox11091686
Bruera E, Schmitz B, Pither J, Neumann CM, Hanson J (2000) The frequency and correlates of dyspnea in patients with advanced cancer. J Pain Symptom Manag 19(5):357–362. https://doi.org/10.1016/s0885-3924(00)00126-3
Gilliam LA, Moylan JS, Callahan LA, Sumandea MP, Reid MB (2011) Doxorubicin causes diaphragm weakness in murine models of cancer chemotherapy. Muscle Nerve 43(1):94–102. https://doi.org/10.1002/mus.21809
Currow DC, Quinn S, Ekstrom M, Kaasa S, Johnson MJ, Somogyi AA, Klepstad P (2015) Can variability in the effect of opioids on refractory breathlessness be explained by genetic factors? BMJ Open 5(5):e006818. https://doi.org/10.1136/bmjopen-2014-006818
Rausch SM, Clark MM, Patten C, Liu H, Felten S, Li Y, Sloan J, Yang P (2010) Relationship between cytokine gene single nucleotide polymorphisms and symptom burden and quality of life in lung cancer survivors. Cancer 116(17):4103–4113. https://doi.org/10.1002/cncr.25255
Su T, Sun H, Lu X, He C, Xiao L, He J, Yang Y, Tang Y (2020) Genetic polymorphisms and haplotypes of BRCA1 gene associated with quality of life and survival among patients with non-small-cell lung cancer. Qual Life Res 29(10):2631–2640
Shin J, Kober KM, Wong ML, Yates P, Cooper BA, Paul SM, Hammer M, Conley Y, Levine JD, Miaskowski C (2023) Distinct shortness of breath profiles in oncology outpatients undergoing chemotherapy. J Pain Symptom Manage 65(3):242–255. https://doi.org/10.1016/j.jpainsymman.2022.11.010
Koutrouli M, Karatzas E, Paez-Espino D, Pavlopoulos GA (2020) A guide to conquer the biological network era using graph theory. Front Bioeng Biotechnol 8:34. https://doi.org/10.3389/fbioe.2020.00034
Portenoy RK, Thaler HT, Kornblith AB, McCarthy Lepore J, Friedlander-Klar H, Kiyasu E, Sobel K, Coyle N, Kemeny N, Norton L, Scher H (1994) The memorial symptom assessment scale: an instrument for the evaluation of symptom prevalence, characteristics and distress. Eur J Cancer 30(9):1326–1336. https://doi.org/10.1016/0959-8049(94)90182-1
Team RC (2019) R: a language and environment for statistical computing. R Foundation for Statistical Computing, Vienna, Austria
Kober KM, Harris C, Conley YP, Dhruva A, Dokiparthi V, Hammer MJ, Levine JD, Oppegaard K, Paul S, Shin J, Sucher A, Wright F, Yuen B, Olshen AB, Miaskowski C (2022) Perturbations in common and distinct inflammatory pathways associated with morning and evening fatigue in outpatients receiving chemotherapy. Cancer Med 12(6):7369–7380. https://doi.org/10.1002/cam4.5435
Robinson MD, McCarthy DJ, Smyth GK (2010) edgeR: a Bioconductor package for differential expression analysis of digital gene expression data. Bioinformatics 26(1):139–140. https://doi.org/10.1093/bioinformatics/btp616
Smyth GK, Ritchie M, Thorne N, Wettenhall J (2005) LIMMA: linear models for microarray data. In: Bioinformatics and computational biology solutions using R and bioconductor. Statistics for Biology and Health
Leek JT, Storey JD (2007) Capturing heterogeneity in gene expression studies by surrogate variable analysis. PLoS Genet 3(9):1724–1735. https://doi.org/10.1371/journal.pgen.0030161
Draghici S, Khatri P, Tarca AL, Amin K, Done A, Voichita C, Georgescu C, Romero R (2007) A systems biology approach for pathway level analysis. Genome Res 17(10):1537–1545. https://doi.org/10.1101/gr.6202607
Aoki-Kinoshita KF, Kanehisa M (2007) Gene annotation and pathway mapping in KEGG. Methods Mol Biol 396:71–91. https://doi.org/10.1007/978-1-59745-515-2_6
Fisher RA (1925) Statistical methods for research workers. Oliver and Boyd, Edinburgh
Dunn OJ (1961) Multiple comparisons among means. J Am Stat Assoc 56(293):52–64. https://doi.org/10.1080/01621459.1961.10482090
Kanehisa M, Furumichi M, Tanabe M, Sato Y, Morishima K (2017) KEGG: new perspectives on genomes, pathways, diseases and drugs. Nucleic Acids Res 45(D1):D353–Dd61. https://doi.org/10.1093/nar/gkw1092
Shannon P, Markiel A, Ozier O, Baliga NS, Wang JT, Ramage D, Amin N, Schwikowski B, Ideker T (2003) Cytoscape: a software environment for integrated models of biomolecular interaction networks. Genome Res 13(11):2498–2504
Newman M (2018) Chapter 7 - Measures and metrics. Networks: Oxford University Press, pp 158–217
Cooper GM (2000) In: The cell: a molecular approach [Internet]. Sinauer Associates, Sunderland (MA) Available from: https://www.ncbi.nlm.nih.gov/books/NBK9839/
Newton R, Holden NS (2006) New aspects of p38 mitogen activated protein kinase (MAPK) biology in lung inflammation. Drug Discov Today Dis Mech 3(1):53–61. https://doi.org/10.1016/j.ddmec.2006.02.007
Halu A, Liu S, Baek SH, Hobbs BD, Hunninghake GM, Cho MH, Silverman EK, Sharma A (2019) Exploring the cross-phenotype network region of disease modules reveals concordant and discordant pathways between chronic obstructive pulmonary disease and idiopathic pulmonary fibrosis. Hum Mol Genet 28(14):2352–2364. https://doi.org/10.1093/hmg/ddz069
Liu J, Wang F, Luo F (2023) The role of JAK/STAT pathway in fibrotic diseases: molecular and cellular mechanisms. Biomolecules 13(1):119. https://doi.org/10.3390/biom13010119
Hwang JA, Kim D, Chun S-M, Bae S, Song JS, Kim MY, Koo HJ, Song JW, Kim WS, Lee JC, Kim HR, Choi C-M, Jang SJ (2018) Genomic profiles of lung cancer associated with idiopathic pulmonary fibrosis. J Pathol 244(1):25–35. https://doi.org/10.1002/path.4978
Wang J, Hu K, Cai X, Yang B, He Q, Wang J, Weng Q (2022) Targeting PI3K/AKT signaling for treatment of idiopathic pulmonary fibrosis. Acta Pharm Sin B 12(1):18–32. https://doi.org/10.1016/j.apsb.2021.07.023
Schuliga M (2015) NF-kappaB signaling in chronic inflammatory airway disease. Biomolecules 5(3):1266–1283. https://doi.org/10.3390/biom5031266
He M, Zhang Y, Xie F, Dou X, Han M, Zhang H (2018) Role of PI3K/Akt/NF-κB and GSK-3β pathways in the rat model of cardiopulmonary bypass-related lung injury. Biomed Pharmacother 106:747–754. https://doi.org/10.1016/j.biopha.2018.06.125
Kim ME, Kim DH, Lee JS (2022) FoxO transcription factors: applicability as a novel immune cell regulators and therapeutic targets in oxidative stress-related diseases. Int J Mol Sci 23(19):11877. https://doi.org/10.3390/ijms231911877
Al-Tamari HM, Dabral S, Schmall A, Sarvari P, Ruppert C, Paik J, DePinho RA, Grimminger F, Eickelberg O, Guenther A, Seeger W, Savai R, Pullamsetti SS (2018) FoxO3 an important player in fibrogenesis and therapeutic target for idiopathic pulmonary fibrosis. EMBO Mol Med 10(2):276–293. https://doi.org/10.15252/emmm.201606261
Culley FJ (2009) Natural killer cells in infection and inflammation of the lung. Immunology 128(2):151–163. https://doi.org/10.1111/j.1365-2567.2009.03167.x
Cahilog Z, Zhao H, Wu L, Alam A, Eguchi S, Weng H, Ma D (2020) The role of neutrophil NETosis in organ injury: novel inflammatory cell death mechanisms. Inflammation 43(6):2021–2032. https://doi.org/10.1007/s10753-020-01294-x
Balamayooran T, Balamayooran G, Jeyaseelan S (2010) Review: Toll-like receptors and NOD-like receptors in pulmonary antibacterial immunity. Innate Immun 16(3):201–210. https://doi.org/10.1177/1753425910366058
Tomankova T, Kriegova E, Liu M (2015) Chemokine receptors and their therapeutic opportunities in diseased lung: far beyond leukocyte trafficking. Am J Phys Lung Cell Mol Phys 308(7):L603–LL18. https://doi.org/10.1152/ajplung.00203.2014
Atamas SP, Chapoval SP, Keegan AD (2013) Cytokines in chronic respiratory diseases. F1000 Biol Rep 5:3. https://doi.org/10.3410/b5-3
Kuwano K (2007) Epithelial cell apoptosis and lung remodeling. Cell Mol Immunol 4(6):419–429
Zayet S, Kadiane-Oussou NJ, Lepiller Q, Zahra H, Royer PY, Toko L, Gendrin V, Klopfenstein T (2020) Clinical features of COVID-19 and influenza: a comparative study on Nord Franche-Comte cluster. Microbes Infect 22(9):481–488. https://doi.org/10.1016/j.micinf.2020.05.016
Acknowledgements
Anatol Sucher and Brian Yuen managed the storage and processing of the biospecimens. This study extends work previously published as part of Joosun Shin’s PhD dissertation.
Funding
This study was supported by grants from the National Cancer Institute (CA134900 and CA233774). Ms. Shin was supported by research grants from Oncology Nursing Foundation and International Society of Nurses in Genetics. Dr. Olshen is supported by grants UCSF NCI Cancer Center Support Grant (CA082103) and UCSF-CTSI (NCATS UL1 TR000004).
Author information
Authors and Affiliations
Contributions
Joosun Shin: Conceptualization; Investigation; Funding acquisition; Writing - original draft; Methodology; Visualization; Formal analysis; Validation; Writing - review & editing. Christine Miaskowski: Supervision; Writing - original draft; Writing - review & editing; Methodology; Conceptualization; Funding acquisition; Project administration; Resources; Investigation. Melisa L. Wong: Writing - review & editing; Supervision. Patsy Yates: Writing - review & editing; Supervision. Adam Olshen: Methodology; Writing - review & editing; Formal analysis. Roy Ritu: Formal analysis; Writing - review & editing. Vasuda Dokiparthi: Writing - review & editing; Formal analysis. Steven Paul: Writing - review & editing; Formal analysis. Bruce Cooper: Formal analysis; Writing - review & editing. Yvette Conley: Methodology; Writing - review & editing. Jon D. Levine: Writing - review & editing; Methodology. Marilyn J. Hammer: Writing - review & editing; Methodology. Kord Kober: Conceptualization; Funding acquisition; Writing - original draft; Writing - review & editing; Visualization; Validation; Methodology; Formal analysis; Data curation; Supervision; Software; Investigation; Project administration; Resources.
Corresponding author
Ethics declarations
Ethical approval
This study was approved by the Committee on Human Research at the University of California.
Consent to participate
This study was exempted from written informed consent.
Consent for publication
All of the authors read the final version of the paper and approved its submission for publication.
Competing interests
Dr. Wong reported conflicts of interest outside of the submitted work: An immediate family member is an employee of Genentech with stock ownership and Dr. Wong receives royalties from UpToDate. The remaining authors have no conflicts of interest to declare.
Disclaimer
The content is solely the responsibility of the authors and does not necessarily represent the official views of the National Institutes of Health.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Shin, J., Miaskowski, C., Wong, M.L. et al. Perturbations in inflammatory pathways are associated with shortness of breath profiles in oncology patients receiving chemotherapy. Support Care Cancer 32, 250 (2024). https://doi.org/10.1007/s00520-024-08446-x
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s00520-024-08446-x