Abstract
Purpose
The prompt initiation of a betalactam antibiotic in febrile neutropenic patients is considered standard of care, while the empiric use of vancomycin is recommended by guidelines in specific situations, with a low level of evidence. The objective of this study was to assess the utilization of vancomycin in the management of febrile neutropenia within four Brazilian medical centers that implemented more stringent criteria for its administration.
Methods
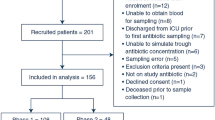
A comprehensive retrospective analysis was performed encompassing all instances of febrile neutropenia observed during the period from 2013 to 2019. The primary focus was to identify the reasons for initiating vancomycin therapy.
Results
A total of 536 consecutive episodes of febrile neutropenia were documented, involving 384 patients with a median age of 52 years (range 18–86). Chemotherapy preceded febrile neutropenia in 59.7% of cases, while 40.3% occurred after hematopoietic stem cell transplantation. The most prevalent underlying diseases were acute myeloid leukemia (26.5%) and non-Hodgkin's lymphoma (22%). According to international guidelines, vancomycin should have been initiated at the onset of fever in 145 episodes (27%); however, it was administered in only 27 cases (5.0%). Three episodes were associated with Staphylococcus aureus bacteremia, two of which were methicillin resistant. The 15-day and 30-day mortality rates were 5.0% and 9.9%, respectively.
Conclusions
The results of this study underscore the notably low utilization rate of vancomycin in cases of febrile neutropenia, despite clear indications outlined in established guidelines. These findings emphasize the importance of carefully implementing guideline recommendations, considering local epidemiological factors, especially when the strength of recommendation is weak.
Similar content being viewed by others
Data availability
The datasets generated during the current study are not publicly available but are available from the corresponding author upon reasonable request.
Code availability
Not applicable.
References
Klastersky J, Ameye L, Maertens J et al (2007) Bacteraemia in febrile neutropenic cancer patients. Int J Antimicrob Agents 30:51–59. https://doi.org/10.1016/j.ijantimicag.2007.06.012
Wisplinghoff H, Seifert H, Wenzel RP, Edmond MB (2003) Current trends in the epidemiology of nosocomial bloodstream infections in patients with hematological malignancies and solid neoplasms in hospitals in the United States. Clin Infect Dis 36:1103–1110. https://doi.org/10.1086/374339
Martinez-Nadal G, Puerta-Alcalde P, Gudiol C et al (2020) Inappropriate empirical antibiotic treatment in high-risk neutropenic patients with bacteremia in the era of multidrug resistance. Clin Infect Dis 70(6):1068–1074. https://doi.org/10.1093/cid/ciz319
Rosa RG, Goldani LZ (2014) Cohort study of the impact of time to antibiotic administration on mortality in patients with febrile neutropenia. Antimicrob Agents Chemother 58:3799–3803. https://doi.org/10.1128/aac.02561-14
Tofas P, Samarkos M, Piperaki ET et al (2017) Pseudomonas aeruginosa bacteraemia in patients with hematologic malignancies: Risk factors, treatment, and outcome. Diagn Microbiol Infect Dis 88:335–341. https://doi.org/10.1016/j.diagmicrobio.2017.05.003
Osih RB, McGregor JC, Rich SE et al (2007) Impact of empiric antibiotic therapy on outcomes in patients with Pseudomonas aeruginosa bacteremia. Antimicrob Agents Chemother 51:839–844. https://doi.org/10.1128/aac.00901-06
Garcia-Vidal C, Cardozo-Espinola C, Puerta-Alcalde P et al (2018) Risk factors for mortality in patients with acute leukemia and bloodstream infections in the era of multiresistance. PLoS ONE 13:e1-12. https://doi.org/10.1371/journal.pone.0199531
Averbuch D, Orasch C, Cordonnier C et al (2013) European guidelines for empirical antibacterial therapy for febrile neutropenic patients in the era of growing resistance: Summary of the 2011 4th European Conference on Infections in Leukemia. Haematologica 98(12):1826–1835. https://doi.org/10.3324/haematol.2013.091025
Freifeld AG, Bow EJ, Sepkowitz KA et al (2011) Clinical practice guideline for the use of antimicrobial agents in neutropenic patients with cancer: 2010 update by the Infectious Diseases Society of America. Clin Infect Dis 52(4):e56–e93. https://doi.org/10.1093/cid/cir073
Baden LR, Swaminathan S, Almyroudis NG et al. (2020). National Comprehensive Cancer Network. Prevention and Treatment of Cancer-Related Infections. https://www.nccn.org/professionals/physician_gls/pdf/infections.pdf. Accessed June 30, 2023
Montassier E, Batard E, Gastinne T, Potel G, de La Cochetière MF (2013) Recent changes in bacteremia in patients with cancer: A systematic review of epidemiology and antibiotic resistance. Eur J Clin Microbiol Infect Dis 32:841–850. https://doi.org/10.1007/s10096-013-1819-7
Chumbita M, Puerta-Alcalde P, Gudiol C et al (2022) Impact of empirical antibiotic regimens on mortality in neutropenic patients with bloodstream infection presenting with septic shock. Antimicrob Agents Chemother 66(2):e0174421. https://doi.org/10.1128/AAC.01744-21
Guarana M, Nucci M, Nouer SA (2019) Shock and early death in hematologic patients with febrile neutropenia. Antimicrob Agents Chemother 63:e01250-e1319. https://doi.org/10.1128/aac.01250-19
Ramphal R, Bolger M, Oblon DJ et al (1992) Vancomycin is not an essential component of the initial empiric treatment regimen for febrile neutropenic patients receiving ceftazidime: A randomized prospective study. Antimicrob Agents Chemother 36:1062–1067. https://doi.org/10.1128/aac.36.5.1062
Beyar-Katz O, Dickstein Y, Borok S, Vidal L, Leibovici L, Paul M (2017) Empirical antibiotics targeting gram-positive bacteria for the treatment of febrile neutropenic patients with cancer. Cochrane Database Syst Rev 6:CD003914. https://doi.org/10.1002/14651858.CD003914.pub4
Cometta A, Kern WV, De Bock R et al (2003) Vancomycin versus placebo for treating persistent fever in patients with neutropenic cancer receiving piperacillin-tazobactam monotherapy. Clin Infect Dis 37:382–389. https://doi.org/10.1086/376637
Erjavec Z, de Vries-Hospers HG, Laseur M, Halie RM, Daenen S (2000) A prospective, randomized, double-blinded, placebo-controlled trial of empirical teicoplanin in febrile neutropenia with persistent fever after imipenem monotherapy. J Antimicrob Chemother 45(6):843–849. https://doi.org/10.1093/jac/45.6.843
Vicente M, Al-Nahedh M, Parsad S et al (2017) Impact of a clinical pathway on appropriate empiric vancomycin use in cancer patients with febrile neutropenia. J Oncol Pharm Pract 23:575–581. https://doi.org/10.1177/1078155216668672
Verlinden A, Mikulska M, Knelange NS et al (2020) Current antimicrobial practice in febrile neutropenia across Europe and Asia: The EBMT Infectious Disease Working Party survey. Bone Marrow Transplant 55:1588–1594. https://doi.org/10.1038/s41409-020-0811-y
National Healthcare safety Network (2023). Patient Safety Component Manual. https://www.cdc.gov/nhsn/pdfs/pscmanual/pcsmanual_current.pdf. Accessed June 30, 2023
Libuit J, Whitman A, Wolfe R, Washington CS (2014) Empiric vancomycin use in febrile neutropenic oncology patients. Open Forum Infectious Diseases, 10 1(1):ofu006. https://doi.org/10.1093/ofid/ofu006
Chastain DB, Wheeler S, Franco-Paredes C et al (2018) Evaluating guideline adherence regarding empirical vancomycin use in patients with neutropenic fever. Int J Infect Dis 69:88–93. https://doi.org/10.1016/j.ijid.2018.02.016
Tenover FC, Biddle JW, Lancaster MV (2001) Increasing resistance to vancomycin and other glycopeptides in Staphylococcus aureus. Emerg Infect Dis 7(2):327–332
Finch RG, Eliopoulos GM (2005) Safety and efficacy of glycopeptide antibiotics. J Antimicrob Chemother 55:5–13. https://doi.org/10.3201/eid0702.010237
Bruniera FR, Ferreira FM, Saviolli LR et al (2015) The use of vancomycin with its therapeutic and adverse effects: A review. Eur Rev Med Pharmacol Sci 19:694–700
Elbarbry F (2018) Vancomycin dosing and monitoring: Critical evaluation of the current practice. Eur J Drug Metab Pharmacokinet 43:259–268. https://doi.org/10.1007/s13318-017-0456-4
Zhang S, Shanley R, Weisdorf DJ, Rashidi A (2020) Vancomycin use and cytomegalovirus reactivation after allogeneic hematopoietic cell transplantation. Blood Adv 4:2640–2643. https://doi.org/10.1182/bloodadvances.2020001984
Peled JU, Gomes ALC, Devlin SM et al (2020) Microbiota as predictor of mortality in allogeneic hematopoietic-cell transplantation. N Engl J Med 382(9):822–834. https://doi.org/10.1056/NEJMoa1900623
van Lier YF, Vos J, Blom B, Hazenberg MD (2023) Allogeneic hematopoietic cell transplantation, the microbiome, and graft-versus-host disease. Gut Microbes 15(1):2178805. https://doi.org/10.1080/19490976.2023.2178805
Weber D, Jenq RR, Peled JU et al (2017) Microbiota disruption induced by early use of broad-spectrum antibiotics is an independent risk factor of outcome after allogeneic stem cell transplantation. Biol Blood Marrow Transplant 23(5):845–852. https://doi.org/10.1016/j.bbmt.2017.02.006
Rashidi A, Gao F, Fredricks DN et al (2023) Analysis of antibiotic exposure and development of acute graft-vs-host disease following allogeneic hematopoietic cell transplantation. JAMA Netw Open 6(6):e2317188. https://doi.org/10.1001/jamanetworkopen.2023.17188
Weber D, Hiergeist A, Weber M, Dettmer K, Wolff D, Hahn J, Herr W, Gessner A, Holler E (2019) Detrimental effect of broad-spectrum antibiotics on intestinal microbiome diversity in patients after allogeneic stem cell transplantation: Lack of commensal sparing antibiotics. Clin Infect Dis 68(5):830–839. https://doi.org/10.1093/cid/ciy711
Franklin S, Aitken SL, Shi Y, Sahasrabhojane PV, Robinson S, Peterson CB, Daver N, Ajami NA, Kontoyiannis DP, Shelburne SA, Galloway-Peña J (2022) Oral and stool microbiome coalescence and its association with antibiotic exposure in acute leukemia patients. Front Cell Infect Microbiol 12:848580. https://doi.org/10.3389/fcimb.2022.848580
World Health Organization (2021). Antimicrobial resistance. https://www.who.int/news-room/fact-sheets/detail/antimicrobial-resistance. Accessed June 30, 2023
Schimpff S, Satterlee W, Young VM, Serpick A (1971) Empiric therapy with carbenicillin and gentamicin for febrile patients with cancer and granulocytopenia. N Engl J Med 284(19):1061–1065. https://doi.org/10.1056/NEJM197105132841904
Tam CS, O’Reilly M, Andresen D, Lingaratnam S, Kelly A, Burbury K, Turnidge J, Slavin MA, Worth LJ, Dawson L, Thursky KA, Australian Consensus Guidelines Steering Committee (2011) Use of empiric antimicrobial therapy in neutropenic fever. Australian Consensus Guidelines 2011 Steering Committee. Internal Medicine Journal 41(1):90–101. https://doi.org/10.1111/j.1445-5994.2010.02340.x
Jeffres MN, Isakow W, Doherty JA, McKinnon PS, Ritchie DJ, Micek ST, Kollef MH (2006) Predictors of mortality for methicillin-resistant Staphylococcus aureus health-care-associated pneumonia: specific evaluation of vancomycin pharmacokinetic indices. Chest 130(4):947–955. https://doi.org/10.1378/chest.130.4.947
Schuster MG, Cleveland AA, Dubberke ER, Kauffman CA, Avery RK, Husain S, Paterson DL, Silveira FP, Chiller TM, Benedict K, Murphy K, Pappas PG (2017) Infections in Hematopoietic Cell Transplant Recipients: Results From the Organ Transplant Infection Project, a Multicenter, Prospective Cohort Study. Open Forum Infect Dis 22 4(2):050. https://doi.org/10.1093/ofid/ofx050
Aguilar-Guisado M, Jiménez-Jambrina M, Espigado I, Rovira M, Martino R, Oriol A, Borrell N, Ruiz I, Martín-Dávila P, de la Cámara R, Salavert M, de la Torre J, Cisneros JM (2011) Spanish Network for Research in Infectious Diseases. Pneumonia in allogeneic stem cell transplantation recipients: a multicenter prospective study. Clin Transplant 25(6):E629-38. https://doi.org/10.1111/j.1399-0012.2011.01495.x
Talagtag M, Patel TS, Scappaticci GB, Perissinotti AJ, Schepers AJ, Petty LA, Pettit KM, Burke PW, Bixby DL, Marini BL (2021) Utility of methicillin-resistant Staphylococcus aureus (MRSA) nasal screening in patients with acute myeloid leukemia (AML). Transpl Infect Dis 23(4):e13612. https://doi.org/10.1111/tid.13612
Kuderer NM, Dale DC, Crawford J, Cosler LE, Lyman GH (2006) Mortality, morbidity, and cost associated with febrile neutropenia in adult cancer patients. Cancer 106(10):2258–2266. https://doi.org/10.1002/cncr.21847
Lyman GH, Michels SL, Reynolds MW, Barron R, Tomic KS, Yu J (2010) Risk of mortality in patients with cancer who experience febrile neutropenia. Cancer 116(23):5555–5563. https://doi.org/10.1002/cncr.25332
Funding
The authors declare that no funds, grants, or other support were received during the preparation of this manuscript.
Author information
Authors and Affiliations
Contributions
All authors contributed to the study concept and design. Data collection was performed by JFR, ADP, MCNS, VCM, MG and SAN. Analysis was performed by JFR, MN and CAR. The first draft of the manuscript was written by JFR and MN. All authors contributed to and approved the final manuscript.
Corresponding author
Ethics declarations
Ethics approval
This is an observational study. The Ethics Committee of Hospital Universitário Clementino Fraga Filho approved the study, and a waiver of informed consent was granted by the Ethics Committee. The study was performed in accordance with the ethical standards as laid down in the 1964 Declaration of Helsinki and its later amendments.
Consent to participate
A waiver of informed consent was granted by the Ethics Committee.
Consent to publish
Not applicable.
Competing interests
The authors declare no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Ramos, J.F., Pereira, A.D., Seiwald, M.C.N. et al. Low utilization of vancomycin in febrile neutropenia: real-world evidence from 4 Brazilian centers. Support Care Cancer 31, 687 (2023). https://doi.org/10.1007/s00520-023-08152-0
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s00520-023-08152-0