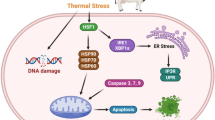
Abstract
Environmental temperature is one of the major factors to affect health and productivity of dairy cattle. Gene expression networks within the cells and tissues coordinate stress response, metabolism, and milk production in dairy cattle. Epigenetic DNA methylations were found to mediate the effect of environment by regulating gene expression patterns. In the present study, we compared three Indian native zebu cattle, Bos indicus (Sahiwal, Tharparkar, and Hariana) and one crossbred Bos indicus × Bos taurus (Vrindavani) for stress gene expression and differences in the DNA methylation patterns. The results indicated acute heat shock to cultured PBMC affected their proliferation, stress gene expression, and DNA methylation. Interestingly, expressions of HSP70, HSP90, and STIP1 were found more pronounced in zebu cattle than the crossbred cattle. However, no significant changes were observed in global DNA methylation due to acute heat shock, even though variations were observed in the expression patterns of DNA methyltransferases (DNMT1, DNMT3a) and demethylases (TET1, TET2, and TET3) genes. The treatment 5-AzaC (5-azacitidine) that inhibit DNA methylation in proliferating PBMC caused significant increase in heat shock-induced HSP70 and STIP1 expression indicating that hypomethylation facilitated stress gene expression. Further targeted analysis DNA methylation in the promoter regions revealed no significant differences for HSP70, HSP90, and STIP1. However, there was a significant hypomethylation for BDNF in both zebu and crossbred cattle. Similarly, NR3C1 promoter region showed hypomethylation alone in crossbred cattle. Overall, the results indicated that tropically adapted zebu cattle had comparatively higher expression of stress genes than the crossbred cattle. Furthermore, DNA methylation may play a role in regulating expression of certain genes involved in stress response pathways.






Similar content being viewed by others
Data availability
Data other than those presented in the manuscript will be made available upon request to the corresponding author.
References:
Abbas Z, Sammad A, Hu L, Fang H, Xu Q, Wang Y (2020) Glucose metabolism and dynamics of facilitative glucose transporters (GLUTs) under the influence of heat stress in dairy cattle. Metabolites 10(8):312. https://doi.org/10.3390/metabo10080312
Allen TE (1962) Responses of Zebu, Jersey, and Zebu X Jersey crossbred heifers to rising temperature, with particular reference to sweating. Aust J Agric Res 13:165–179. https://doi.org/10.1071/AR9620165
Ammer S, Lambertz C, von Soosten D, Zimmer K, Meyer U, Dänicke S, Gauly M (2018) Impact of diet composition and temperature-humidity index on water and dry matter intake of high-yielding dairy cows. J Anim Physiol Anim Nutr (berl) 102(1):103–113. https://doi.org/10.1111/jpn.12664
Anastasiadi D, Esteve-Codina A, Piferrer F (2018) Consistent inverse correlation between DNA methylation of the first intron and gene expression across tissues and species. Epigenet Chromatin 29:11(1):37 https://doi.org/10.1186/s13072-018-0205-1
Baena MM, Tizioto PC, Meirelles SLC, Regitano LCA (2018) HSF1 and HSPA6 as functional candidate genes associated with heat tolerance in Angus cattle. Rev Bras Zootec 47https://doi.org/10.1590/rbz4720160390
Bakusic J, Ghosh M, Polli A, Bekaert B, Schaufeli W, Claes S, Godderis L (2020) Epigenetic perspective on the role of brain-derived neurotrophic factor in burnout. Transl Psychiatry 10(1):354. https://doi.org/10.1038/s41398-020-01037-4
Baumgard LH, Wheelock JB, Sanders SR, Moore CE, Green HB, Waldron MR, Rhoads RP (2011) Postabsorptive carbohydrate adaptations to heat stress and monensin supplementation in lactating Holstein cows. J Dairy Sci 94(11):5620–5633. https://doi.org/10.3168/jds.2011-4462
Bhanuprakash V, Singh U, Sengar G, Sajjanar B, Bhusan B, Raja TV, Alex R, Kumar S, Singh R, Kumar A, Alyethodi RR, Kumar S, Deb R (2016) Differential effect of thermal stress on HSP70 expression, nitric oxide production and cell proliferation among native and crossbred dairy cattle. J Therm Biol 59:18–25. https://doi.org/10.1016/j.jtherbio.2016.04.012
Bomfim MM, Andrade GM, Del Collado M, Sangalli JR, Fontes PK, Nogueira MFG, Meirelles FV, da Silveira JC, Perecin F (2017) Antioxidant responses and deregulation of epigenetic writers and erasers link oxidative stress and DNA methylation in bovine blastocysts. Mol Reprod Dev 84(12):1296–1305. https://doi.org/10.1002/mrd.22929
Brueckner B, Kuck D, Lyko F (2007) DNA methyltransferase inhibitors for cancer therapy. Cancer J 13(1):17–22. https://doi.org/10.1097/PPO.0b013e31803c7245
Ciliberti MG, Albenzio M, Inghese C, Santillo A, Marino R, Sevi A, Caroprese M (2017) Peripheral blood mononuclear cell proliferation and cytokine production in sheep as affected by cortisol level and duration of stress. J Dairy Sci 100(1):750–756. https://doi.org/10.3168/jds.2016-11688
Collier RJ, Collier JL, Rhoads RP, Baumgard LH (2008) Invited review: genes involvedin the bovine heat stress response. J Dairy Sci (2):445–454. https://doi.org/10.3168/jds.2007-0540
Cramer T, Rosenberg T, Kisliouk T, Meiri N (2019a) Early-life epigenetic changes along the corticotropin-releasing hormone (CRH) gene influence resilience or vulnerability to heat stress later in life. Mol Psychiatry 24(7):1013–1026. https://doi.org/10.1038/s41380-018-0280-5
Cramer T, Rosenberg T, Kisliouk T, Meiri N (2019b) PARP Inhibitor affects long-term heat-stress response via changes in DNA methylation. Neuroscience 399:65–76. https://doi.org/10.1016/j.neuroscience.2018.12.018
Dado-Senn B, Skibiel AL, Fabris TF, Zhang Y, Dahl GE, Peñagaricano F, Laporta J (2018) RNA-Seq reveals novel genes and pathways involved in bovine mammary involution during the dry period and under environmental heat stress. Sci Rep 8(1):11096. https://doi.org/10.1038/s41598-018-29420-8
Dahl GE, Tao S, Thompson IM (2012) Lactation Biology Symposium: effects of photoperiod on mammary gland development and lactation. J Anim Sci 90(3):755–760. https://doi.org/10.2527/jas.2011-4630
Deb R, Sajjanar B, Singh U, Kumar S, Singh R, Sengar G, Sharma A (2014) Effect of heat stress on the expression profile of Hsp90 among Sahiwal (Bos indicus) and Frieswal (Bos indicus × Bos taurus) breed of cattle: a comparative study. Gene 536(2):435–440. https://doi.org/10.1016/j.gene.2013.11.086
Denoyelle L, de Villemereuil P, Boyer F, Khelifi M, Gaffet C, Alberto F, Benjelloun B, Pompanon F (2021) Genetic variations and differential DNA methylation to face contrasted climates in small ruminants: an analysis on traditionally-managed sheep and goats. Front Genet 12:745284. https://doi.org/10.3389/fgene.2021.745284
Dorts J, Falisse E, Schoofs E, Flamion E, Kestemont P, Silvestre F (2016) DNA methyltransferases and stress-related genes expression in zebrafish larvae after exposure to heat and copper during reprogramming of DNA methylation. Sci Rep 6:34254. https://doi.org/10.1038/srep34254
Dowling DF (1955) The hair follicle and apocrine gland population of Zebu (Bos indicus) and Shorthorn (B taurus) cattle skin. Aust J Agric Res 64:645–654
Fang H, Kang L, Abbas Z, Hu L, Chen Y, Tan X, Wang Y, Xu Q (2021) Identification of key genes and pathways associated with thermal stress in peripheral blood mononuclear cells of holstein dairy cattle. Front Genet 12:662080. https://doi.org/10.3389/fgene.2021.662080
Feeney A, Nilsson E, Skinner MK (2014) Epigenetics and transgenerational inheritance in domesticated farm animals. J Anim Sci Biotechnol 5(1):48. https://doi.org/10.1186/2049-1891-5-48
Fernandes JG, Oliveira H, Carvalheiro R, Cardoso DF, Fonseca L, Ventura R, Albuquerque L (2020) Whole-genome sequencing provides new insights into genetic mechanisms of tropical adaptation in Nellore (Bos primigenius indicus). Sci Rep. https://doi.org/10.1038/s41598-020-66272-7
Franci O, Amici A, Margarit R, Merendino N, Piccolella E (1996) Influence of thermal and dietary stress on immune response of rabbits. J Anim Sci 74(7):1523–1529. https://doi.org/10.2527/1996.7471523x
Gabriel JE, Ferro JA, Stefani RM, Ferro MI, Gomes SL, Macari M (1996) Effect of acute heat stress on heat shock protein 70 messenger RNA and on heat shock protein expression in the liver of broilers. Br Poult Sci 37(2):443–449. https://doi.org/10.1080/00071669608417875
Gan JK, Zhang DX, He DL, Zhang XQ, Chen ZY, Luo QB (2013) Promoter methylation negatively correlated with mRNA expression but not tissue differential expression after heat stress. Genet Mol Res 12(1):809–819. https://doi.org/10.4238/2013.March.15.1
Garner JB, Chamberlain AJ, Vander Jagt C, Nguyen TTT, Mason BA, Marett LC, Leury BJ, Wales WJ, Hayes BJ (2020) Gene expression of the heat stress response in bovine peripheral white blood cells and milk somatic cells in vivo. Sci Rep 10(1):19181. https://doi.org/10.1038/s41598-020-75438-2
Gatta E, Saudagar V, Auta J, Grayson DR, Guidotti A (2021) Epigenetic landscape of stress surfeit disorders: Key role for DNA methylation dynamics. Int Rev Neurobiol 156:127–183.https://doi.org/10.1016/bs.irn.2020.08.002
Gorniak T, Meyer U, Südekum KH, Dänicke S (2014) Impact of mild heat stress on dry matter intake, milk yield and milk composition in mid-lactation Holstein dairy cows in a temperate climate. Arch Anim Nutr 68(5):358–369. https://doi.org/10.1080/1745039X.2014.950451
Gunn KM, Holly MA, Veith TL, Buda AR, Prasad R, Rotz CA, Soder KJ, Stoner AMK (2019) Projected heat stress challenges and abatement opportunities for U.S. milk production. PLoS One 14(3):e0214665. https://doi.org/10.1371/journal.pone.0214665
Hall LW, Villar F, Chapman JD, McLean DJ, Long NM, Xiao Y, Collier JL, Collier RJ (2018) An evaluation of an immunomodulatory feed ingredient in heat-stressed lactating Holstein cows: effects on hormonal, physiological, and production responses. J Dairy Sci 101(8):7095–7105. https://doi.org/10.3168/jds.2017-14210
Hammond AC, Olson TA, Chase CC Jr, Bowers EJ, Randel RD, Murphy CN, Vogt DW, Tewolde A (1996) Heat tolerance in two tropically adapted Bos taurus breeds, Senepol and Romosinuano, compared with Brahman, Angus, and Hereford cattle in Florida. J Anim Sci 74(2):295–303. https://doi.org/10.2527/1996.742295x
Hansen PJ (2004) Physiological and cellular adaptations of zebu cattle to thermal stress. Anim Reprod Sci 82–83:349–360. https://doi.org/10.1016/j.anireprosci.2004.04.011
Hao Y, Cui Y, Gu X (2016) Genome-Wide DNA Methylation Profiles Changes Associated with Constant Heat Stress in Pigs as Measured by Bisulfite Sequencing. Sci Rep 6:27507. https://doi.org/10.1038/srep27507
Hassan FU, Nawaz A, Rehman MS, Ali MA, Dilshad SMR, Yang C (2019) Prospects of HSP70 as a genetic marker for thermo-tolerance and immuno-modulation in animals under climate change scenario. Anim Nutr 5(4):340–350. https://doi.org/10.1016/j.aninu.2019.06.005
Heller G, Schmidt WM, Ziegler B, Holzer S, Müllauer L, Bilban M, Zielinski CC, Drach J, Zöchbauer-Müller S (2008) Genome-wide transcriptional response to 5-aza-2’-deoxycytidine and trichostatin a in multiple myeloma cells. Cancer Res 68(1):44–54. https://doi.org/10.1158/0008-5472.CAN-07-2531
Houtepen LC, Vinkers CH, Carrillo-Roa T, Hiemstra M, van Lier PA, Meeus W, Branje S, Heim CM, Nemeroff CB, Mill J, Schalkwyk LC, Creyghton MP, Kahn RS, Joëls M, Binder EB, Boks MP (2016) Genome-wide DNA methylation levels and altered cortisol stress reactivity following childhood trauma in humans. Nat Commun 7:10967. https://doi.org/10.1038/ncomms10967
Huang L, Mivechi NF, Moskophidis D (2001) Insights into regulation and function of the major stress-induced hsp70 molecular chaperone in vivo: analysis of mice with targeted gene disruption of the hsp70.1 or hsp70.3 gene. Mol Cell Biol 21(24):8575–8591. https://doi.org/10.1128/MCB.21.24.8575-8591.2001
Ibeagha-Awemu EM, Zhao X (2015) Epigenetic marks: regulators of livestock phenotypes and conceivable sources of missing variation in livestock improvement programs. Front Genet 6:302. https://doi.org/10.3389/fgene.2015.00302
IPCC (2007) Climate Change: Synthesis Report. Contribution of Working Groups I, II and III to the Fourth Assessment Report of the Intergovernmental Panel on Climate Change [Core Writing Team: Pachauri RK and Reisinger A (eds)]. IPCC, Geneva, Switzerland, 104
Ismaylova E, Lévesque ML, Pomares FB, Szyf M, Nemoda Z, Fahim C, Vitaro F, Brendgen M, Dionne G, Boivin M, Tremblay RE, Booij L (2018) Serotonin transporter promoter methylation in peripheral cells and neural responses to negative stimuli: a study of adolescent monozygotic twins. Transl Psychiatry 8(1):147. https://doi.org/10.1038/s41398-018-0195-6
Ito S, D’Alessio AC, Taranova OV, Hong K, Sowers LC, Zhang Y (2010) Role of Tet proteins in 5mC to 5hmC conversion, ES-cell self-renewal and inner cell mass specification. Nature 466(7310):1129–1133. https://doi.org/10.1038/nature09303
Janke O, Tzschentke B (2010) Long-lasting effect of changes in incubation temperature on heat stress induced neuronal hypothalamic c-Fos expression in chickens. Open Ornithol J 3:150–155. https://doi.org/10.2174/1874453201003010150
Joksimović-Todorović M, Davidović V, Hristov S, Stanković B (2011) Effect of heat stress on milk production in dairy cows. Biotechnol Anim Husb 27(3):1017-1023 27. https://doi.org/10.2298/BAH1103017J
Karpova N, Umemori J (2016) Protocol for methylated DNA Immuno precipitation (MeDIP) analysis. In: Karpova N (ed) Epigenetic methods in neuroscience research 105:97–114. https://doi.org/10.1007/978-1-4939-2754-8_6
Katz A, Meiri N (2006) Brain-derived neurotrophic factor is critically involved in thermal-experience-dependent developmental plasticity. J Neurosci 26(15):3899–3907. https://doi.org/10.1523/JNEUROSCI.0371-06.2006
Kim WS, Ghassemi Nejad J, Roh SG, Lee HG (2020) Heat-shock proteins gene expression in peripheral blood mononuclear cells as an indicator of heat stress in beef calves. Animals (Basel) 10(5):895. https://doi.org/10.3390/ani10050895
Kinoshita T, Seki M (2014) Epigenetic memory for stress response and adaptation in plants. Plant Cell Physiol 55(11):1859–1863. https://doi.org/10.1093/pcp/pcu125
Kisliouk T, Meiri N (2009) A critical role for dynamic changes in histone H3 methylation at the Bdnf promoter during postnatal thermotolerance acquisition. Eur J Neurosci 30(10):1909–1922. https://doi.org/10.1111/j.1460-9568.2009.06957.x
Kisliouk T, Cramer T, Meiri N (2017) Methyl CpG level at distal part of heat-shock protein promoter HSP70 exhibits epigenetic memory for heat stress by modulating recruitment of POU2F1-associated nucleosome-remodeling deacetylase (NuRD) complex. J Neurochem 141(3):358–372. https://doi.org/10.1111/jnc.14014
Kiziltepe T, Hideshima T, Catley L, Raje N, Yasui H, Shiraishi N, Okawa Y, Ikeda H, Vallet S, Pozzi S, Ishitsuka K, Ocio EM, Chauhan D, Anderson KC (2007) 5-Azacytidine, a DNA methyltransferase inhibitor, induces ATR-mediated DNA double-strand break responses, apoptosis, and synergistic cytotoxicity with doxorubicin and bortezomib against multiple myeloma cells. Mol Cancer Ther 6(6):1718–1727. https://doi.org/10.1158/1535-7163.MCT-07-0010
Kojima D, Nakamura T, Banno M, Umemoto Y, Kinoshita T, Ishida Y, Tajima F (2017) Head-out immersion in hot water increases serum BDNF in healthy males. Int J Hyperth 34(6):834–839. https://doi.org/10.1080/02656736.2017.1394502
Kornicka K, Marycz K, Marędziak M, Tomaszewski KA, Nicpoń J (2017) The effects of the DNA methyltranfserases inhibitor 5-Azacitidine on ageing, oxidative stress and DNA methylation of adipose derived stem cells. J Cell Mol Med 21(2):387–401. https://doi.org/10.1111/jcmm.12972
Kuroda A, Rauch TA, Todorov I, Ku HT, Al-Abdullah IH, Kandeel F, Mullen Y, Pfeifer GP, Ferreri K (2009) Insulin gene expression is regulated by DNA methylation. PLoS ONE 4(9):e6953. https://doi.org/10.1371/journal.pone.0006953.Erratum.In:PLoSOne2009;4(10)
Lacetera N, Bernabucci U, Scalia D, Basiricò L, Morera P, Nardone A (2006) Heat stress elicits different responses in peripheral blood mononuclear cells from Brown Swiss and Holstein cows. J Dairy Sci 89(12):4606–4612. https://doi.org/10.3168/jds.S0022-0302(06)72510-3
Lallias D, Bernard M, Ciobotaru C, Dechamp N, Labbé L, Goardon L, Le Calvez JM, Bideau M, Fricot A, Prézelin A, Charles M, Moroldo M, Cousin X, Bouchez O, Roulet A, Quillet E, Dupont-Nivet M (2021) Sources of variation of DNA methylation in rainbow trout: combined effects of temperature and genetic background. Epigenetics 16(9):1031–1052. https://doi.org/10.1080/15592294.2020.1834924
Laurino D, Ferro J, Macari M, Rutz F, Furlan L, Tiraboschi F (2001) Efeito do estresse térmico agudo sobre os níveis da proteína e RNA mensageiro da Hsp70, em fígado e cérebro de pintos de corte de diferentes linhagens. R Bras Zootec 30(5):1506–1513. https://doi.org/10.1590/S1516-35982001000600018
Li L, Sun Y, Wu J, Li X, Luo M, Wang G (2015) The global effect of heat on gene expression in cultured bovine mammary epithelial cells. Cell Stress Chaperones 20(2):381–389. https://doi.org/10.1007/s12192-014-0559-7
Lister R, Mukamel EA, Nery JR, Urich M, Puddifoot CA, Johnson ND, Lucero J, Huang Y, Dwork AJ, Schultz MD, Yu M, Tonti-Filippini J, Heyn H, Hu S, Wu JC, Rao A, Esteller M, He C, Haghighi FG, Sejnowski TJ, Behrens MM, Ecker JR (2013) Global epigenomic reconfiguration during mammalian brain development. Science 341(6146):1237905. https://doi.org/10.1126/science.1237905
Littlejohn BP, Price DM, Neuendorff DA, Carroll JA, Vann RC, Riggs PK, Riley DG, Long CR, Welsh TH Jr, Randel RD (2018) Prenatal transportation stress alters genome-wide DNA methylation in suckling Brahman bull calves. J Anim Sci 96(12):5075–5099. https://doi.org/10.1093/jas/sky350
Livernois AM, Mallard BA, Cartwright SL, Cánovas A (2021) Heat stress and immune response phenotype affect DNA methylation in blood mononuclear cells from Holstein dairy cows. Sci Rep 11(1):11371. https://doi.org/10.1038/s41598-021-89951-5
Lozano-Villegas KJ, Rodríguez-Hernández R, Herrera-Sánchez MP, Uribe-García HF, Naranjo-Gómez JS, Otero-Arroyo RJ, Rondón-Barragán IS (2021) Identification of reference genes for expression studies in the whole-blood from three cattle breeds under two states of livestock weather safety. Animals (Basel) 11(11):3073. https://doi.org/10.3390/ani11113073
Madkour M, Aboelenin MM, Aboelazab O, Elolimy AA, El-Azeem NA, El-Kholy MS, Alagwany M, Shourrap M (2021) Hepatic expression responses of DNA methyltransferases, heat shock proteins, antioxidant enzymes, and NADPH 4 to early life thermal conditioning in broiler chickens. Ital J Anim Sci 20:433–446. https://doi.org/10.1080/1828051X.2021.1890645
Matosin N, Cruceanu C, Binder EB (2017) Preclinical and clinical evidence of DNA methylation changes in response to trauma and chronic stress. Chronic Stress (thousand Oaks) 1:2470547017710764. https://doi.org/10.1177/2470547017710764
Maunakea AK, Nagarajan RP, Bilenky M, Ballinger TJ, D’Souza C, Fouse SD, Johnson BE, Hong C, Nielsen C, Zhao Y, Turecki G, Delaney A, Varhol R, Thiessen N, Shchors K, Heine VM, Rowitch DH, Xing X, Fiore C, Schillebeeck M, Jones SJ, Haussler D, Marra MA, Hirst M, Wang T, Costello JF (2010) Conserved role of intragenic DNA methylation in regulating alternative promoters. Nature 466(7303):253–257. https://doi.org/10.1038/nature09165
Murgo AJ (2005) Innovative approaches to the clinical development of DNA methylation inhibitors as epigenetic remodeling drugs. Semin Oncol 32(5):458–464. https://doi.org/10.1053/j.seminoncol.2005.07.00
Naranjo-Gómez JS, Uribe-García HF, Herrera-Sánchez MP, Lozano-Villegas KJ, Rodríguez-Hernández R, Rondón-Barragán IS (2021) Heat stress on cattle embryo: gene regulation and adaptation. Heliyon 7(3):e06570. https://doi.org/10.1016/j.heliyon.2021.e065708
Odunuga OO, Longshaw VM, Blatch GL (2004) Hop: more than an Hsp70/Hsp90 adaptor protein. BioEssays 26(10):1058–1068. https://doi.org/10.1002/bies.20107
Olson TA, Lucena CJ, Chase C, Hammond AC (2003) Evidence of a major gene influencing hair length and heat tolerance in Bos taurus cattle. J Anim Sci 81(1):80–90. https://doi.org/10.2527/2003.81180x
Paula-Lopes FF, Chase CC Jr, Al-Katanani YM, Krininger CE 3rd, Rivera RM, Tekin S, Majewski AC, Ocon OM, Olson TA, Hansen PJ (2003) Genetic divergence in cellular resistance to heat shock in cattle: differences between breeds developed in temperate versus hot climates in responses of preimplantation embryos, reproductive tract tissues and lymphocytes to increased culture temperatures. Reproduction 125(2):285–294. https://doi.org/10.1530/rep.0.1250285
Rhoads ML, Rhoads RP, VanBaale MJ, Collier RJ, Sanders SR, Weber WJ, Crooker BA, Baumgard LH (2009) Effects of heat stress and plane of nutrition on lactating Holstein cows: I. Production, metabolism, and aspects of circulating somatotropin. J Dairy Sci 92(5):1986–1997. https://doi.org/10.3168/jds.2008-1641
Rhoads RP, La Noce AJ, Wheelock JB, Baumgard LH (2011) Alterations in expression of gluconeogenic genes during heat stress and exogenous bovine somatotropin administration. J Dairy Sci 94(4):1917–1921. https://doi.org/10.3168/jds.2010-3722
Rhoads RP, Baumgard LH, Suagee JK (2013) Metabolic priorities during heat stress with an emphasis on skeletal muscle. J Anim Sci 91(6):2492–2503. https://doi.org/10.2527/jas.2012-6120
Salces-Ortiz J, González C, Bolado-Carrancio A, Rodríguez-Rey JC, Calvo JH, Muñoz R, Serrano MM (2015) Ovine HSP90AA1 gene promoter: functional study and epigenetic modifications. Cell Stress Chaperones 20(6):1001–1012. https://doi.org/10.1007/s12192-015-0629-5
Sammad A, Umer S, Shi R, Zhu H, Zhao X, Wang Y (2020a) Dairy cow reproduction under the influence of heat stress. J Anim Physiol Anim Nutr (berl) 104(4):978–986. https://doi.org/10.1111/jpn.13257
Sammad A, Wang YJ, Umer S, Lirong H, Khan I, Khan A, Ahmad B, Wang Y (2020b) Nutritional physiology and biochemistry of dairy cattle under the influence of heat stress: consequences and opportunities. Animals (basel) 10(5):793. https://doi.org/10.3390/ani10050793
Sato D, Yamashiro K, Yamazaki Y, Tsubaki A, Onishi H, Takehara N, Maruyama A (2017) Site specificity of changes in cortical oxyhaemoglobin concentration induced by water immersion. Adv Exp Med Biol 977:233–240. https://doi.org/10.1007/978-3-319-55231-6_32
Saunderson EA, Spiers H, Mifsud KR, Gutierrez-Mecinas M, Trollope AF, Shaikh A, Mill J, Reul JM (2016) Stress-induced gene expression and behavior are controlled by DNA methylation and methyl donor availability in the dentate gyrus. Proc Natl Acad Sci USA 113(17):4830–4835. https://doi.org/10.1073/pnas.1524857113
Secco D, Wang C, Shou H, Schultz MD, Chiarenza S, Nussaume L, Ecker JR, Whelan J, Lister R (2015) Stress induced gene expression drives transient DNA methylation changes at adjacent repetitive elements. Elife 4:e09343. https://doi.org/10.7554/eLife.09343
Sigdel A, Abdollahi-Arpanahi R, Aguilar I, Peñagaricano F (2019) Whole genome mapping reveals novel genes and pathways involved in milk production under heat stress in US Holstein cows. Front Genet 10:928. https://doi.org/10.3389/fgene.2019.00928
Singh K, Molenaar AJ, Swanson KM, Gudex B, Arias JA, Erdman RA, Stelwagen K (2012) Epigenetics: a possible role in acute and transgenerational regulation of dairy cow milk production. Animal 6(3):375–381. https://doi.org/10.1017/S1751731111002564
Singh AK, Upadhyay RC, Chandra G, Kumar S, Malakar D, Singh SV, Singh MK (2020) Genome-wide expression analysis of the heat stress response in dermal fibroblasts of Tharparkar (zebu) and Karan-Fries (zebu × taurine) cattle. Cell Stress Chaperones 25(2):327–344. https://doi.org/10.1007/s12192-020-01076-2
Skibiel AL, Peñagaricano F, Amorín R, Ahmed BM, Dahl GE, Laporta J (2018) In utero heat stress alters the offspring epigenome. Sci Rep 8(1):14609. https://doi.org/10.1038/s41598-018-32975-1
Spiers D, Spain J, Sampson J, Rhoads R (2004) Use of physiological parameters to predict milk yield and feed intake in heat-stressed dairy cows. J Therm Biol 29:759–764. https://doi.org/10.1016/j.jtherbio.2004.08.051
Stankiewicz AR, Lachapelle G, Foo CP, Radicioni SM, Mosser DD (2005) Hsp70 inhibits heat-induced apoptosis upstream of mitochondria by preventing Bax translocation. J Biol Chem 280(46):38729–38739. https://doi.org/10.1074/jbc.M509497200
Steel GJ, Fullerton DM, Tyson JR, Stirling CJ (2004) Coordinated activation of Hsp70 chaperones. Science 303(5654):98–101. https://doi.org/10.1126/science.1092287
Stefanski V, Engler H (1998) Effects of acute and chronic social stress on blood cellular immunity in rats. Physiol Behav 64(5):733–741. https://doi.org/10.1016/s0031-9384(98)00127-9
Summer A, Lora I, Formaggioni P, Gottardo F (2018) Impact of heat stress on milk and meat production. Anim Front 9(1):39–46. https://doi.org/10.1093/af/vfy026
Tian F, Zhan F, VanderKraats ND, Hiken JF, Edwards JR, Zhang H, Zhao K, Song J (2013) DNMT gene expression and methylome in Marek’s disease resistant and susceptible chickens prior to and following infection by MDV. Epigenetics 8(4):431–444. https://doi.org/10.4161/epi.24361
Triantaphyllopoulos KA, Ikonomopoulos I, Bannister AJ (2016) Epigenetics and inheritance of phenotype variation in livestock. Epigenet Chromatin 9:31. https://doi.org/10.1186/s13072-016-0081-5
Vinoth A, Thirunalasundari T, Shanmugam M, Uthrakumar A, Suji S, Rajkumar U (2018) Evaluation of DNA methylation and mRNA expression of heat shock proteins in thermal manipulated chicken. Cell Stress Chaperones 23(2):235–252. https://doi.org/10.1007/s12192-017-0837-2
Wan X, He X, Liu Q, Wang X, Ding X, Li H (2020) Frequent and mild scrotal heat stress in mice epigenetically alters glucose metabolism in the male offspring. Am J Physiol Endocrinol Metab 319(2):E291–E304. https://doi.org/10.1152/ajpendo.00038.2020
Weber M, Schübeler D (2007) Genomic patterns of DNA methylation: targets and function of an epigenetic mark. Curr Opin Cell Biol 19(3):273–280. https://doi.org/10.1016/j.ceb.2007.04.011
West JW (2003) Effects of heat-stress on production in dairy cattle. J Dairy Sci 86(6):2131–2144. https://doi.org/10.3168/jds.S0022-0302(03)73803-X
Wheelock JB, Rhoads RP, Vanbaale MJ, Sanders SR, Baumgard LH (2010) Effects of heat stress on energetic metabolism in lactating Holstein cows. J Dairy Sci 93(2):644–655. https://doi.org/10.3168/jds.2009-2295
Yang Y, Li J, Geng Y, Liu L, Li D (2021) Azacitidine regulates DNA methylation of GADD45γ in myelodysplastic syndromes. J Clin Lab Anal 35(2):e23597. https://doi.org/10.1002/jcla.23597
Zachut M, Kra G, Livshitz L, Portnick Y, Yakoby S, Friedlander G, Levin Y (2017) Seasonal heat stress affects adipose tissue proteome toward enrichment of the Nrf2-mediated oxidative stress response in late-pregnant dairy cows. J Proteomics 158:52–61. https://doi.org/10.1016/j.jprot.2017.02.011
Acknowledgements
Authors acknowledge the Director, ICAR-IVRI for extending the facilities to carry over the present work.
Funding
This work was supported by the SERB-DST, Gov’t of India, CRG Grant no: CRG/2019/005515.
Author information
Authors and Affiliations
Contributions
BS, GS, TD, and BPM designed the work. SM, MTA, OK, and GNT performed the experiments. RKG, SD, and PKG involved in analyzing the data. SM and MTA wrote the manuscript and BS and GS edited the final draft. All the authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Ethics approval
All the experiments were approved by the Institutional Animal Ethics Committee (IAEC) of Indian Veterinary Research Institute (IVRI).
Consent to participate
Not applicable.
Consent to publish
Not applicable.
Conflict of interest
The authors declare no competing interests.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Masroor, S., Aalam, M.T., Khan, O. et al. Effect of acute heat shock on stress gene expression and DNA methylation in zebu (Bos indicus) and crossbred (Bos indicus × Bos taurus) dairy cattle. Int J Biometeorol 66, 1797–1809 (2022). https://doi.org/10.1007/s00484-022-02320-3
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00484-022-02320-3