Abstract
There is increasing evidence that primary hypertension, coronary heart disease, and other aspects of the so-called metabolic syndrome that develop in adulthood are primed in fetal life or early postnatally. The identification of this phenomenon, also known as prenatal or fetal programming, and the detailed characterization of the underlying pathomechanisms will greatly influence the understanding of these diseases. The present paper reviews recent experimental and clinical evidence that low nephron number, found in patients with renal dysplasia and low birth weight, is a risk factor for cardiovascular disease in later life. Therefore, it is important to identify children at risk as early as possible in order to treat them early and to prevent the development of end-organ damage. This could be an important goal for pediatrics in the near future.



Similar content being viewed by others
References
Luft FC (2004) Present status of genetic mechanisms in hypertension. Med Clin North Am 88:1–18
Rettig R, Folberth C, Stauss H, Kopf D, Waldherr R, Unger T (1990) Role of the kidney in primary hypertension: a renal transplantation study in rats. Am J Physiol 258:F606–F611
Strandgaard S, Hansen U (1986) Hypertension in renal allograft recipients may be conveyed by cadaveric kidneys from donors with subarachnoidal haemorrhage. BMJ 292:1041–1044
Curtis JJ, Luke RG, Dustan HP, Kashgarian M, Whelchel JD, Jones P, Diethelm AG (1983) Remission of essential hypertension after renal transplantation. N Engl J Med 309:1009–1015
Mei-Zahav M, Korzets Z, Cohen I, Kessler O, Rathaus V, Wolach B, Pomeranz A (2001) Ambulatory blood pressure monitoring in children with a solitary kidney—a comparison between unilateral renal agenesis and uninephrectomy. Blood Press Monit 6:263
Brenner BM, Garcia DL, Anderson S (1988) Glomeruli and blood pressure: less of one, more of the other? Am J Hypertens 1:335–347
Woods LL (1999) Neonatal uninephrectomy causes hypertension in adult rats. Am J Physiol 276:R974–R978
Moritz KM, Wintour EM, Dodic M (2002) Fetal uninephrectomy leads to postnatal hypertension and compromised renal function. Hypertension 39:1071–1076
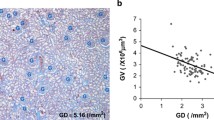
Keller G, Zimmer G, Mall G, Ritz E, Amann K (2003) Nephron number in patients with primary hypertension. N Engl J Med 348:101–108
Ingelfinger JR (2003) Is microanatomy destiny? N Engl J Med 348:99–100
Cullen-McEwen LA, Kett MM, Dowling J, Anderson WP, Bertram JF (2003) Nephron number, renal function, and arterial pressure in aged GDNF heterozygous mice. Hypertension 41:335–340
Merlet-Benichou C (1999) Influence of fetal environment on kidney development. Int J Dev Biol 43:453–456
Wintour EM, Moritz KM, Johnson K, Ricardo S, Samuel CS, Dodic M (2003) Reduced nephron number in adult sheep, hypertensive as a result of prenatal glucocorticoid treatment. J Physiol 549:929–935
Amri K, Freund N, Van Huyen JP, Merlet-Benichou C, Lelievre-Pegorier M (2001) Altered nephrogenesis due to maternal diabetes is associated with increased expression of IGF-II/mannose-6-phosphate receptor in the fetal kidney. Diabetes 50:1069–1075
Regina S, Lucas R, Miraglia SM, Zaladek Gil F, Machado Coimbra T (2001) Intrauterine food restriction as a determinant of nephrosclerosis. Am J Kidney Dis 37:467–476
Mitchell EK, Louey S, Cock ML, Harding R, Black MJ (2004) Nephron endowment and filtration surface area in the kidney after growth restriction of fetal sheep. Pediatr Res 55:769–773
Vehaskari MV, Aviles DH, Manning J (2001) Prenatal programming of adult hypertension in the rat. Kidney Int 59:238–245
Wintour EM, Johnson K, Koukoulas I, Moritz K, Tersteeg M, Dodic M (2003) Programming the cardiovascular system, kidney and the brain—a review. Placenta 24 [Suppl A]:S65–S71
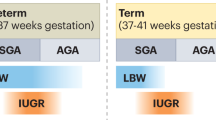
Manalich R, Reyes L, Herrera M, Melendi C, Fundora I (2000) Relationship between weight at birth and the number and size of renal glomeruli in humans: a histomorphometric study. Kidney Int 58:770–773
Hughson M, Farris AB 3rd, Douglas-Denton R, Hoy WE, Bertram JF (2003) Glomerular number and size in autopsy kidneys: the relationship to birth weight. Kidney Int 63:2113–2122
Battista MC, Oligny LL, St-Louis J, Brochu M (2002) Intrauterine growth restriction in rats is associated with hypertension and renal dysfunction in adulthood. Am J Physiol Endocrinol Metab 283:E124–E131
Manning J, Vehaskari VM (2001) Low birth weight-associated adult hypertension in the rat. Pediatr Nephrol 16:417–422
Bertram C, Trowern AR, Copin N, Jackson AA, Whorwood CB (2001) The maternal diet during pregnancy programs altered expression of the glucocorticoid receptor and type 2 11beta-hydroxysteroid dehydrogenase: potential molecular mechanisms underlying the programming of hypertension in utero. Endocrinology 142:2841–2853
Bassan H, Trejo LL, Kariv N, Bassan M, Berger E, Fattal A, Gozes I, Harel S (2000) Experimental intrauterine growth retardation alters renal development. Pediatr Nephrol 5:192–195
Woods LL, Weeks DA, Rasch R (2004) Programming of adult blood pressure by maternal protein restriction: role of nephrogenesis. Kidney Int 65:1339–1348
Manning J, Beutler K, Knepper MA, Vehaskari VM (2002) Upregulation of renal BSC1 and TSC in prenatally programmed hypertension. Am J Physiol Renal Physiol 283:F202–F206
Bauer R, Walter B, Ihring W, Kluge H, Lampe V, Zwiener U (2000) Altered renal function in growth-restricted newborn piglets. Pediatr Nephrol 14:735–739
Bauer R, Walter B, Bauer K, Klupsch R, Patt S, Zwiener U (2002) Intrauterine growth restriction reduces nephron number and renal excretory function in newborn piglets. Acta Physiol Scand 176:83–90
Langley-Evans SC, Sherman RC, Welham SJ, Nwagwu MO, Gardner DS, Jackson AA (1999) Intrauterine programming of hypertension: the role of the renin-angiotensin system. Biochem Soc Trans 27:88–93
Kingdom JC, Hayes M, McQueen J, Howatson AG, Lindop GB (1999) Intrauterine growth restriction is associated with persistent juxtamedullary expression of renin in the fetal kidney. Kidney Int 55:424–429
Houang M, Morineau G, Bouc Y le, Fiet J, Gourmelen M (1999) The cortisol-cortisone shuttle in children born with intrauterine growth retardation. Pediatr Res 46:189–193
Masuzaki H, Paterson J, Shinyama H, Morton NM, Mullins JJ, Seckl JR, Flier JS (2001) A transgenic model of visceral obesity and the metabolic syndrome. Science 294:2166–2170
Haynes WG, Sivitz WI, Morgan DA, Walsh SA, Mark AL (1997) Sympathetic and cardiorenal actions of leptin. Hypertension 30 [Suppl]:619–623
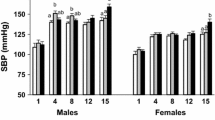
Barker DJ, Osmond C, Law CM (1989) The intrauterine and early postnatal origins of cardiovascular disease and chronic bronchitis. J Epidemiol Community Health 43:237–240
Law CM, Shiell AW, Newsome CA, Syddall HE, Shinebourne EA, Fayers PM, Martyn CN, Swiet M de (2002) Fetal, infant, and childhood growth and adult blood pressure: a longitudinal study from birth to 22 years of age. Circulation 105:1088–1092
Zhang J, Brenner RA, Klebanoff MA (2001) Differences in birth weight and blood pressure at age 7 years among twins. Am J Epidemiol 153:779–782
Williams S, Poulton R (2002) Birth size, growth, and blood pressure between the ages of 7 and 26 years: failure to support the fetal origins hypothesis. Am J Epidemiol 155:849–852
Fattal-Valevski A, Bernheim J, Leitner Y, Redianu B, Bassan H, Harel S (2001) Blood pressure values in children with intrauterine growth retardation. Isr Med Assoc J 3:805–808
Whincup PH, Bredow M, Payne F, Sadler S, Golding J (1999) Size at birth and blood pressure at 3 years of age. The Avon Longitudinal Study of Pregnancy and Childhood (ALSPAC). Am J Epidemiol 149:730–739
Uiterwaal CS, Anthony S, Launer LJ, Witteman JC, Trouwborst AM, Hofman A, Grobbee DE (1997) Birth weight, growth, and blood pressure: an annual follow-up study of children aged 5 through 21 years. Hypertension 30:267–271
Law CM, Shiell AW, Newsome CA, Syddall HE, Shinebourne EA, Fayers PM, Martyn CN, Swiet M de (2002) Fetal, infant, and childhood growth and adult blood pressure: a longitudinal study from birth to 22 years of age. Circulation 105:1088–1092
Horta BL, Barros FC, Victora CG, Cole TJ (2003) Early and late growth and blood pressure in adolescence. J Epidemiol Community Health 57:226–230
Huxley RR, Shiell AW, Law CM (2000) The role of size at birth and postnatal catch-up growth in determining systolic blood pressure: a systematic review of the literature. J Hypertens 18:815–831
Plagemann A (2003) Fetale Programmierung und Funktionelle Teratologie: Zur perinatalen Prägung dauerhaft erhöhter Disposition für das Metabolische Syndrom X. In: Zabransky S (ed) SGA-Syndrom Small for gestational Age. Jonag Verlag, Marburg, pp 49–59
Rostand SG (2003) Oligonephronia, primary hypertension and renal disease: ‘is the child father to the man?’. Nephrol Dial Transplant 18:1434–1438
Tulassay T, Vasarhelyi B (2002) Birth weight and renal function. Curr Opin Nephrol Hypertens 11:347–352
Ingelfinger JR (2004) Pathogenesis of perinatal programming. Curr Opin Nephrol Hypertens 13:459–464
Roseboom TJ, Meulen JHP van der, Ravelli ACJ, Osmond C, Barker DJP, Bleker OP (2001): Effects of prenatal exposure to the Dutch famine on adult disease in later life: an overview. J Mol Cell Endocrinol 185:93–98
Bouret SG, Draper SJ, Simerly RB (2004) Trophic action of leptin on hypothalamic neurons that regulate feeding. Science 304:108–115
Dötsch J, Rascher W, Meissner U (2004) Perinatal programming of appetite control by leptin? Eur J Endocrinol 151:1–3
Acknowledgement
This study was supported by the Deutsche Forschungsgemeinschaft (SFB 423, projects B8 and B9).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Amann, K., Plank, C. & Dötsch, J. Low nephron number—a new cardiovascular risk factor in children?. Pediatr Nephrol 19, 1319–1323 (2004). https://doi.org/10.1007/s00467-004-1643-5
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00467-004-1643-5