Abstract
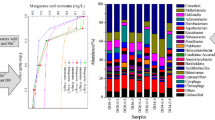
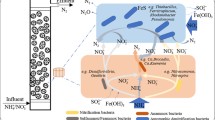
A continuous-upflow biofilter packed with sponge iron was constructed for nitrate removal under an anaerobic atmosphere. Microbacterium sp. W5, a nitrate reducing and Fe(II) oxidizing strain, was added to the biofilter as an inoculum. The best results were achieved when NO3 −-N concentration was 30 mg/L and Fe2+ was 800 mg/L. Nitrite in influent would inhibit nitrate removal and aqueous Fe2+ resulted in encrustation. Fe(II)EDTA would prevent cells from encrustation and the maximum nitrogen removal efficiency was about 90 % with Fe(II)EDTA level of 1100 mg/L. Nitrate reduction followed first-order reaction kinetics. Characteristics of biofilms were analyzed by X-ray fluorescence spectroscopy.






Similar content being viewed by others
References
Ghafari S, Hasan M, Aroua MK (2008) Bio-electrochemical removal of nitrate from water and wastewater—a review. Bioresour Technol 99:3965–3974
Karanasios KA, Vasiliadou IA, Pavlou S, Vayenas DV (2010) Hydrogenotrophic denitrification of potable water: a review. J Hazard Mater 180:20–37
Shrimali M, Singh KP (2001) New methods of nitrate removal from water. Environ Pollut 112:351–359
Foglar L, Briski F, Sipos L, Vukovic M (2005) High nitrate removal from synthetic wastewater with the mixed bacterial culture. Bioresour Technol 96:879–888
Chung J, Amin K, Kim S, Yoon S, Kwon K, Bae W (2014) Autotrophic denitrification of nitrate and nitrite using thiosulfate as an electron donor. Water Res 58:169–178
Chakraborty A, Picardal F (2013) Neutrophilic, nitrate-dependent, Fe(II) oxidation by a Dechloromonas species. World J Microbiol Biotechnol 29:617–623
Zhang M, Zheng P, Wang R, Li W, Lu H, Zhang J (2014) Nitrate-dependent anaerobic ferrous oxidation (NAFO) by denitrifying bacteria: a perspective autotrophic nitrogen pollution control technology. Chemosphere 117:604–609
Weber KA, Achenbach LA, Coates JD (2006) Microorganisms pumping iron: anaerobic microbial iron oxidation and reduction. Nat Rev Microbiol 4:752–764
Canfield DE, Rosing MT, Bjerrum C (2006) Early anaerobic metabolisms. Philos Trans R Soc Lond Ser A Math Phys Eng Sci 361:1819–1834
Straub KL, Schonhuber WA, Buchholz-Cleven B, Schink B (2004) Diversity of ferrous iron-oxidizing, nitrate-reducing bacteria and their involvement in oxygen-independent iron cycling. Geomicrobiol J 21:371–378
Carlson HK, Clark IC, Blazewicz SJ, Iavarone AT, Coates JD (2013) Fe(II) oxidation is an innate capability of nitrate-reducing bacteria that involves abiotic and biotic reactions. J Bacteriol 195:3260–3268
Kappler A, Johnson CM, Crosby HA, Beard BL, Newman DK (2010) Evidence for equilibrium iron isotope fractionation by nitrate-reducing iron(II)-oxidizing bacteria. Geochim Cosmochim Acta 74:2826–2842
Chakraborty A, Roden EE, Schieber J, Picardal F (2011) Enhanced growth of Acidovorax sp. strain 2AN during nitrate-dependent Fe(II) oxidation in batch and continuous-flow systems. Appl Environ Microbiol 77:8548–8556
Kappler A, Schink B, Newman DK (2005) Fe(III) mineral formation and cell encrustation by the nitrate-dependent Fe(II)-oxidizer strain BoFeN1. Geobiology 3:235–245
Finneran KT, Housewright ME, Lovley DR (2002) Multiple influences of nitrate on uranium solubility during bioremediation of uranium-contaminated subsurface sediments. Environ Microbiol 4:510–516
Li B, Tian C, Zhang D, Pan X (2014) Anaerobic nitrate-dependent iron (II) oxidation by a novel autotrophic bacterium, Citrobacter freundii strain PXL1. Geomicrobiol J 31:138–144
Oshiki M, Ishii S, Yoshida K, Fujii N, Ishiguro M, Satoh H, Okabe S (2013) Nitrate-dependent ferrous iron oxidation by anaerobic ammonium oxidation (Anammox) bacteria. Appl Environ Microbiol 79:4087–4093
Weber KA, Hedrick DB, Peacock AD, Thrash JC, White DC, Achenbach LA, Coates JD (2009) Physiological and taxonomic description of the novel autotrophic, metal oxidizing bacterium, Pseudogulbenkiania sp. strain 2002. Appl Microbiol Biotechnol 83:555–565
Byrne-Bailey KG, Weber KA, Chair AH, Bose S, Knox T, Spanbauer TL, Chertkov O, Coates JD (2010) Completed genome sequence of the anaerobic iron-oxidizing bacterium Acidovorax ebreus strain TPSY. J Bacteriol 192:1475–1476
Kopf SH, Henny C, Newman DK (2013) Ligand-enhanced abiotic iron oxidation and the effects of chemical versus biological iron cycling in anoxic environments. Environ Sci Technol 47:2602–2611
Hegler F, Schmidt C, Schwarz H, Kappler A (2010) Does a low-pH microenvironment around phototrophic FeII-oxidizing bacteria prevent cell encrustation by FeIII minerals? FEMS Microbiol Ecol 74:592–600
APHA (2005) Standard Methods for the examination of water and wastewater. Association A.P.H, Washington, DC
Zhang M, Zheng P, Li W, Wang R, Ding S, Abbas G (2015) Performance of nitrate-dependent anaerobic ferrous oxidizing (NAFO) process: a novel prospective technology for autotrophic denitrification. Bioresour Technol 179:543–548
Weber KA, Pollock J, Cole KA, O’Connor SM, Achenbach LA, Coates JD (2006) Anaerobic nitrate-dependent iron(II) bio-oxidation by a novel lithoautotrophic betaproteobacterium, strain 2002. Appl Environ Microbiol 72:686–694
Waki M, Yasuda T, Yokoyama H, Hanajima D, Ogino A, Suzuki K, Yamagishi T, Suwa Y, Tanaka Y (2009) Nitrogen removal by co-occurring methane oxidation, denitrification, aerobic ammonium oxidation, and anammox. Appl Microbiol Biotechnol 84:977–985
Adav SS, Lee D, Lai JY (2010) Enhanced biological denitrification of high concentration of nitrite with supplementary carbon source. Appl Microbiol Biotechnol 85:773–778
Coby AJ, Picardal FW (2005) Inhibition of NO3− and NO2− reduction by microbial Fe(III) reduction: evidence of a reaction between NO2− and cell surface-bound Fe2+. Appl Environ Microbiol 71:5267–5274
Cooper DC, Picardal FW, Schimmelmann A, Coby AJ (2003) Chemical and biological interactions during nitrate and goethite reduction by Shewanella putrefaciens 200. Appl Environ Microbiol 69:3517–3525
Schaedler S, Burkhardt C, Hegler F, Straub KL, Miot J, Benzerara K, Kappler A (2009) Formation of cell-iron-mineral aggregates by phototrophic and nitrate-reducing anaerobic Fe(II)-oxidizing bacteria. Geomicrobiol J 26:93–103
Dippon U, Pantke C, Porsch K, Larese-Casanova P, Kappler A (2012) Potential function of added minerals as nucleation sites and effect of humic substances on mineral formation by the nitrate-reducing Fe(II)-oxidizer Acidovorax sp. BoFeN1. Environ Sci Technol 46:6556–6565
Miot J, Benzerara K, Morin G, Kappler A, Bernard S, Obst M, Ferard C, Skouri-Panet F, Guigner J, Posth N, Galvez M Jr, Brown GE, Guyot F (2009) Iron biomineralization by anaerobic neutrophilic iron-oxidizing bacteria. Geochim Cosmochim Acta 73:696–711
Dong X, Zhang Y, Zhou J, Chen M, Wang X, Shi Z (2013) Fe(II)EDTA–NO reduction coupled with Fe(II)EDTA oxidation by a nitrate- and Fe(III)-reducing bacterium. Bioresour Technol 138:339–344
Straub KL, Benz M, Schink B, Widdel F (1996) Anaerobic, nitrate-dependent microbial oxidation of ferrous iron. Appl Environ Microbiol 62:1458–1460
Muehe EM, Gerhardt S, Schink B, Kappler A (2009) Ecophysiology and the energetic benefit of mixotrophic Fe(II) oxidation by various strains of nitrate-reducing bacteria. FEMS Microbiol Ecol 70:335–343
Nguyen TAH, Ngo HH, Guo WS, Nguyen TV, Zhang J, Liang S, Chen SS, Nguyen NC (2014) A comparative study on different metal loaded soybean milk by-product ‘okara’ for biosorption of phosphorus from aqueous solution. Bioresour Technol 169:291–298
Acknowledgments
This work was financially supported by the National Natural Science Foundation of China (NSFC) (No. 51008239 and 51378400), the Natural Science Foundation of Hubei Province, China (No. 2013CFB289 and 2013CFB308) and, the National "Twelfth Five-Year" Plan for Science and Technology Pillar Program (2014BAL04B04, 2015BAL01B02).
Author information
Authors and Affiliations
Corresponding authors
Rights and permissions
About this article
Cite this article
Zhou, J., Wang, H., Yang, K. et al. Autotrophic denitrification by nitrate-dependent Fe(II) oxidation in a continuous up-flow biofilter. Bioprocess Biosyst Eng 39, 277–284 (2016). https://doi.org/10.1007/s00449-015-1511-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00449-015-1511-7