Abstract
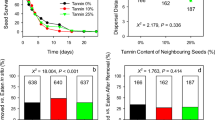
Scatter-hoarding rodents play important roles in seed dispersal and predation in many forest ecosystems. Existing studies have shown that the seed foraging preference of rodents is directly affected by seed traits and indirectly affected by the traits of other co-existing seeds nearby (i.e., neighbor effect). Plant seeds exhibit a combination of diverse seed traits, including seed size, chemical defense, and nutrient content. Therefore, it is difficult to evaluate the influence of each single seed trait on such neighbor effects. Here, by using artificial seeds, we investigated the impacts of contrasts in seed size, tannin content, and nutrient content on neighbor effects. We tracked 9000 tagged artificial seeds from 30 seed-seed paired treatments in a subtropical forest in southwest China. The contrast in seed size between paired seeds created obvious neighbor effects measured through three seed dispersal related indicators: the proportion of seeds being removed, the proportion of seeds cached, and the distance transported by rodents. However, the magnitudes and the signs of the neighbor effects differed among pairs, including both apparent mutualism and apparent competition, depending on the contrast in seed size between paired seeds. The contrasts of tannin and nutrient content between paired seeds showed relatively few neighbor effects. Our results suggest that the contrast in seed traits between the target seed and its neighboring seeds should be considered when studying rodent-seed interactions. Furthermore, we expect that similar complex neighbor effects may also exist in other plant-animal interactions, such as pollination and herbivory.


Similar content being viewed by others
Availability of data and material
The datasets used and/or analyzed during the current study are available from the corresponding author on reasonable request.
Code availability
Not applicable.
References
Barbosa P, Hines J, Kaplan I, Martinson H, Szczepaniec A, Szendrei Z (2009) Associational resistance and associational susceptibility: having right or wrong neighbors. Annu Rev Ecol Evol Syst 40:1–20. https://doi.org/10.1146/annurev.ecolsys.110308.120242
Bascompte J, Jordano P (2007) Plant-animal mutualistic networks: the architecture of biodiversity. Annu Rev Ecol Evol Syst 38:567–593. https://doi.org/10.1146/annurev.ecolsys.38.091206.095818
Cagnolo L, Salvo A, Valladares G (2011) Network topology: patterns and mechanisms in plant-herbivore and host-parasitoid food webs. J Anim Ecol 80:342–351. https://doi.org/10.1111/j.1365-2656.2010.01778.x
Cao L, Wang ZY, Yan C, Chen J, Guo C, Zhang ZB (2016) Differential foraging preferences on seed size by rodents result in higher dispersal success of medium-sized seeds. Ecology 97:3070–3078. https://doi.org/10.1002/ecy.1555
Cope OL, Becker Z, Ode PJ, Paul RL, Pearse IS (2020) Associational effects of plant ontogeny on damage by a specialist insect herbivore. Oecologia 193:593–602. https://doi.org/10.1007/s00442-020-04702-z
Feng L, Chen S, Wang B (2021) Fine-scale spatiotemporal variation in seed-rodent interactions: a potential contribution to species coexistence. For Ecol Manag 498:119566. https://doi.org/10.1016/j.foreco.2021.119566
Garzon-Lopez CX, Ballesteros-Mejia L, Ordonez A, Bohlman SA, Olff H, Jansen PA (2015) Indirect interactions among tropical tree species through shared rodent seed predators: a novel mechanism of tree species coexistence. Ecol Lett 18:752–760. https://doi.org/10.1111/ele.12452
Gong HD, Tang CZ, Wang B (2015) Post-dispersal seed predation and its relations with seed traits: a thirty-species-comparative study. Plant Spec Biol 30:193–201. https://doi.org/10.1111/1442-1984.12051
Hahn PG, Orrock JL (2016) Neighbor palatability generates associational effects by altering herbivore foraging behavior. Ecology 97:2103–2111. https://doi.org/10.1002/ecy.1430
Hirsch BT, Kays R, Pereira VE, Jansen PA (2012) Directed seed dispersal towards areas with low conspecific tree density by a scatter-hoarding rodent. Ecol Lett 15:1423–1429. https://doi.org/10.1111/ele.12000
Jordano P, Bascompte J, Olesen JM (2003) Invariant properties in coevolutionary networks of plant-animal interactions. Ecol Lett 6:69–81. https://doi.org/10.1046/j.1461-0248.2003.00403.x
Koerner SE, Smith MD, Burkepile DE, Hanan NP, Avolio ML, Collins SL et al (2018) Change in dominance determines herbivore effects on plant biodiversity. Nat Ecol Evol 2:1925–1932. https://doi.org/10.1038/s41559-018-0696-y
Kuprewicz EK, García-Robledo C (2019) Deciphering seed dispersal decisions: size, not tannin content, drives seed fate and survival in a tropical forest. Ecosphere 10:e02551. https://doi.org/10.1002/ecs2.2551
Lai J, Zou Y, Zhang S, Zhang X, Mao L (2022) glmm.hp: an R package for computing individual effect of predictors in generalized linear mixed models. J Plant Ecol 15:1302–1307. https://doi.org/10.1093/jpe/rtac096
Lichti NI, Steele MA, Zhang H, Swihart RK (2014) Mast species composition alters seed fate in North American rodent-dispersed hardwoods. Ecology 95:1746–1758. https://doi.org/10.1890/13-1657.1
Lichti NI, Steele MA, Swihart RK (2017) Seed fate and decision-making processes in scatter-hoarding rodents. Biol Rev 92:474–504. https://doi.org/10.1111/brv.12240
Moore JE, McEuen AB, Swihart RK, Contreras TA, Steele MA (2007) Determinants of seed removal distance by scatter-hoarding rodents in deciduous forests. Ecology 88:2529–2540. https://doi.org/10.1890/07-0247.1
Ostoja SM, Schupp EW, Durham S, Klinger R (2013) Seed harvesting is influenced by associational effects in mixed seed neighbourhoods, not just by seed density. Funct Ecol 27:775–785. https://doi.org/10.1111/1365-2435.12091
Proesmans W, Albrecht M, Gajda A, Neumann P, Paxton RJ, Pioz M, Polzin C, Schweiger O, Settele J, Szentgyörgyi H, Thulke HH, Vanbergen AJ (2021) Pathways for novel epidemiology: plant-pollinator-pathogen networks and global change. Trends Ecol Evol 36:623–636. https://doi.org/10.1016/j.tree.2021.03.006
Shimada T, Takahashi A, Shibata M, Yagihashi T (2015) Effects of within-plant variability in seed weight and tannin content on foraging behaviour of seed consumers. Funct Ecol 29:1513–1521. https://doi.org/10.1111/1365-2435.12464
Takahashi A, Shimada T (2008) Selective consumption of acorns by the Japanese wood mouse according to tannin content: a behavioral countermeasure against plant secondary metabolites. Ecol Res 23:1033–1038. https://doi.org/10.1007/s11284-008-0473-5
Vander Wall SB (2010) How plants manipulate the scatter-hoarding behaviour of seed-dispersing animals. Philos Trans R Soc B 365:989–997. https://doi.org/10.1098/rstb.2009.0205
Vander Wall SB, Kuhn KM, Beck MJ (2005) Seed removal, seed predation, and secondary dispersal. Ecology 86:801–806. https://doi.org/10.1890/04-0847
Wang B (2020) Neighbour effects do not always show consistent patterns, contrast of seed trait matters: evidence from a seed-rodent mutualism study. Behav Ecol Sociobiol 74:1–10. https://doi.org/10.1007/s00265-020-02900-6
Wang B, Chen J (2008) Tannin concentration enhances seed caching by scatter-hoarding rodents: an experiment using artificial ‘seeds.’ Acta Oecol 34:379–385. https://doi.org/10.1016/j.actao.2008.07.001
Wang B, Chen J (2009) Seed size, more than nutrient or tannin content, affects seed caching behavior of a common genus of old world rodents. Ecology 90:3023–3032. https://doi.org/10.1890/08-2188.1
Wang B, Chen J (2011) Scatter-hoarding rodents prefer slightly astringent food. PLoS One 6:e26424. https://doi.org/10.1371/journal.pone.0026424
Wang B, Ives AR (2017) Tree-to-tree variation in seed size and its consequences for seed dispersal versus predation by rodents. Oecologia 183:751–762. https://doi.org/10.1007/s00442-016-3793-0
Wang B, Yang XL (2014) Teasing apart the effects of seed size and energy content on rodent scatter-hoarding behavior. PLoS One 9:e111389. https://doi.org/10.1371/journal.pone.0111389
Wang B, Wang G, Chen J (2012) Scatter-hoarding rodents use different foraging strategies for seeds from different plant species. Plant Ecol 213:1329–1336. https://doi.org/10.1007/s11258-012-0091-8
Wang B, Ye CX, Cannon CH, Chen J (2013) Dissecting the decision making process of scatter-hoarding rodents. Oikos 122:1027–1034. https://doi.org/10.1111/j.1600-0706.2012.20823.x
Wang B, Phillips JS, Tomlinson KW (2018) Tradeoff between physical and chemical defense in plant seeds is mediated by seed mass. Oikos 127:440–447. https://doi.org/10.1111/oik.04867
Wang ZY, Wang B, Yan C, Yuan SD, Cao L (2020) Neighborhood effects on the tannin-related foraging decisions of two rodent species under semi-natural conditions. Integr Zool 15:569–577. https://doi.org/10.1111/1749-4877.12473
Xiao ZS, Holyoak M, Krebs CJ, Huang XQ (2022) Palatability and profitability of co-occurring seeds alter indirect interactions among rodent-dispersed trees. Integr Zool 17:206–216. https://doi.org/10.1111/1749-4877.12543
Yang XF, Yan C, Gu HF, Zhang ZB (2020) Interspecific synchrony of seed rain shapes rodent-mediated indirect seed-seed interactions of sympatric tree species in a subtropical forest. Ecol Lett 23:45–54. https://doi.org/10.1111/ele.13405
Yi XF, Wang ZY (2015) Context-dependent seed dispersal determines acorn survival of sympatric oak species. Plant Ecol 216:123–132. https://doi.org/10.1007/s11258-014-0421-0
Zhang ZB, Wang ZY, Chang G, Yi XF, Lu JQ, Xiao ZS, Zhang HM, Cao L, Wang FS, Li HJ, Yan C (2016) Trade-off between seed defensive traits and impacts on interaction patterns between seeds and rodents in forest ecosystems. Plant Ecol 217:253–265. https://doi.org/10.1007/s11258-016-0566-0
Acknowledgements
We thank Ailaoshan Station of Subtropical Forest Ecosystem Studies for logistical support in the field work, Dr. Jiangshan Lai for statistical support, and two anonymous reviewers for insightful comments on early manuscript drafts.
Funding
This study was funded by the National Natural Science Foundation of China (31971444 and 32171533) and Anhui Provincial Natural Science Foundation (2208085J28).
Author information
Authors and Affiliations
Contributions
BW conceived and designed the experiments. HL and JZ performed the experiments. BW and HL analyzed the data and wrote first draft of the manuscript; other authors provided editorial advice.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflicts of interest.
Ethical approval
Not applicable.
Consent to participate
Not applicable.
Consent for publication
Not applicable.
Additional information
Communicated by Caroline Müller.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Liu, H., Zhang, J. & Wang, B. Contrasting seed traits of co-existing seeds lead to a complex neighbor effect in a seed-rodent interaction. Oecologia 201, 1017–1024 (2023). https://doi.org/10.1007/s00442-023-05365-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00442-023-05365-2