Abstract
The significance of soil water redistribution by roots and nocturnal transpiration for nutrient acquisition were assessed for deep-rooted 3-year-old leguminous Aspalathus linearis shrubs of the Cape Floristic Region (South Africa). We hypothesised that hydraulic redistribution and nocturnal transpiration facilitate nutrient acquisition by releasing moisture in shallow soil to enable acquisition of shallow-soil nutrients during the summer drought periods and by driving water fluxes from deep to shallow soil powering mass-flow nutrient acquisition, respectively. A. linearis was supplied with sub-surface (1-m-deep) irrigation rates of 0, 2 or 4 L day−1 plant−1. Some plants were unfertilized, whilst others were surface- or deep-fertilized (1 m depth) with Na15NO3 and CaP/FePO4. We also supplied deuterium oxide (2H2O) at 1 m depth at dusk and measured its predawn redistribution to shallow soil and plant stems. Hydraulic redistribution of deep water was substantial across all treatments, accounting for 34–72 % of surface-soil predawn moisture. Fourteen days after fertilization, the surface-fertilized plants exhibited increased hydraulic redistribution and increased 15N and P acquisition with higher rates of deep-irrigation. Deep-fertilization also increased hydraulic redistribution to surface soils, although these plants additionally accumulated 2H2O in their stem tissue overnight, probably due to nocturnal transpiration. Plants engaged in nocturnal transpiration also increased 15N and P acquisition from deep fertilizer sources. Thus, both nocturnal transpiration and hydraulic redistribution increased acquisition of shallow soil N and P, possibly through a combination of increased nutrient availability and mobility.
Similar content being viewed by others
Introduction
The root systems of many plants explore large volumes of soils as a consequence encountering soil patches that differ greatly with respect to moisture and nutrients (Jackson and Caldwell 1993; Prieto et al. 2012). Under these circumstances, roots may serve as conduits for the movement of water from wetter to drier regions of the soil profile, a phenomenon known as ‘hydraulic redistribution’ (hereafter, HR; Burgess et al. 1998) that has been documented in more than 60 plant species, including trees, shrubs and grasses (Prieto et al. 2012; Neumann and Cardon 2012). Although HR is especially prevalent in species from seasonally droughted environments, it has also been reported in tropical forest (Jackson et al. 2000) and crop taxa (Pang et al. 2013; Sekiya et al. 2011; Sekiya and Yano 2004; Allen et al. 2004). Despite being common, the ecological and physiological benefits of HR remain unclear (Ryel 2004).
HR is powered by water potential gradients that exist between the roots and soil (e.g. Burgess and Bleby 2006; Hawkins et al. 2009). Thus, the magnitude and direction of HR is controlled by the location and relative strengths of sources of water and sinks for water, which are in turn influenced by a network of competing water potential gradients in the soil–plant-atmosphere continuum (Nadezhdina et al. 2010; Prieto et al. 2012). High nocturnal transpiration, for example, lowers leaf water potentials, thereby forcing water to flow towards the canopy and reducing HR towards the drier soil (Hultine et al. 2004; Howard et al. 2009; Prieto et al. 2010). At night, in the absence of strong nocturnal transpiration when stomatal conductance is low, water is moved by roots from wetter to drier soil patches to equilibrate the existing gradients in soil and plant water potentials (Prieto et al. 2012). Thus, HR may be modulated by conditions that alter plant and soil water potentials such as soil moisture and evaporative demand (Scholz et al. 2002). Water deep in the soil profile, for instance, provides the conditions for upward HR to shallow soil layers (Ludwig et al. 2003, 2004; Kurz-Besson et al. 2006; Muñoz et al. 2008; Burgess et al. 2000; Ryel et al. 2002, 2004).
HR of water from deep to shallow soil may enable access to nutrients (e.g. N and P) that accumulate in topsoil due to plant litterfall (Jobbágy and Jackson 2000, 2004). Indeed, HR has been found to enhance uptake of some nutrients; e.g. N (Matzner and Richards 1996; Dawson 1997; Leffler et al. 2004; Armas et al. 2012; Shen et al. 2011), Zn (Nambiar 1976) and PO4 3− (Wang et al. 2009). Other studies, however, have reported no HR-associated uptake of NO3 − (Snyder et al. 2008), PO4 3− (Rose et al. 2008; Wang et al. 2009) or Li (a tracer for K; Hawkins et al. 2009; Rose et al. 2008). Such inconsistencies have largely been attributed to lack of suitable control treatments, limited experimental time scales, or differences in the form of supplied nutrients (Armas et al. 2012). Moreover, the prevailing environmental conditions during these experiments also affect the magnitudes and the direction of HR fluxes (Prieto et al. 2012) resulting in downward, upward or lateral fluxes of water (e.g. Burgess et al. 1998; Hultine et al. 2003; Smart et al. 2005) and inconsistent effects on nutrient acquisition.
There are several mechanisms through which HR may function to promote nutrient acquisition: (1) through prolonging the life span of fine roots and maintaining root–soil contact in dry soils by redistributing water into the soil occupied by these roots (Dawson 1993; Caldwell et al. 1998; Bauerle et al. 2008); (2) through maintaining active microbial floras, including mycorrhizas (Querejeta et al. 2003; Warren et al. 2007; Lehto and Zwiazek 2011); (3) through increasing mineralisation and/or nutrient acquisition from the soil that is wetted by HR (e.g. Prieto et al. 2012; Armas et al. 2012); and (4) through facilitating root acquisition of nutrients in fertile, but dry soil patches through daily cycles of soil wetting and drying which improves nutrient mobility (Aanderud and Richards 2009; Cardon and Gage 2006; Matzner and Richards 1996; Leffler et al. 2004; Shen et al. 2011). Nutrient mobility in soils wetted by HR may be enhanced by both improved diffusion of nutrients (Barber 1995) and mass-flow of nutrients (Cramer et al. 2009).
Nutrients may in turn moderate water potential gradients through physiological controls on water flux or through determining the growth of plants. For example, root conductance changes with exposure to NO −3 (e.g. Gloser et al. 2007). Acquisition of NO −3 also modulates both day (Clarkson et al. 2000; Gorska et al. 2008; Cramer et al. 2008, 2009) and night transpiration (Kupper et al. 2012), possibly altering plant water potentials and consequently the magnitude of HR. Whilst HR is suggested to be a passive physical process that occurs even through the roots of fully senesced plants (e.g. Leffler et al. 2005), we envisage the nutritional control of transpiration to modulate HR, particularly where nutrients are leached to wetter deep soil layers. Increased transpiration suppresses HR; possibly because leaves become stronger sinks forcing water inflow from the soil through the stems towards the leaves, rather than releasing it back to the soil (Howard et al. 2009; Prieto et al. 2012). Modulation of HR may also occur secondarily through the NO −3 regulation of transpiration (Wilkinson et al. 2007; Cramer et al. 2009), which may alter water potential gradients and consequently HR. To our knowledge, there has been no report of nutrients modulating the magnitude or direction of HR.
In this study, we used Aspalathus linearis (Burm. F) Dahlg. (Fabaceae) shrubs to test the hypotheses that (1) both nocturnal transpiration and HR promote nutrient acquisition by driving water fluxes and enabling mass-flow acquisition of nutrients from deep soil, and (2) that hydraulically redistributed water promotes nutrient uptake from shallow soil. To create a range of soil water potentials, three deep-soil irrigation levels were supplied to A. linearis plants in the field and a fertilizer containing 15N and P supplemented either in surface or deep soils.
Materials and methods
Site description and experimental set-up
The study site was on the farm Bloemfontein (South Africa; 31°44′04.96″S, 19°08′18.77″E) where A. linearis is cultivated ‘organically’ for commercial rooibos tea production. The ca. 1.5-m-tall shrub is native to the north-western to western portion of the Cape Floristic Region (CFR), South Africa, and forms cluster roots, arbuscular mycorrhizal associations and also rhizobial nodules that fix N2 (Hawkins et al. 2011). Aspalathus linearis has a tap root extending to 2 m or more in depth (Morton 1983), often reaching the ground water, which is why the plant can survive periods of drought (Gérard 2010). Our observations of root distribution down a soil profile of 1.63 m depth showed ca. 75 % of roots to be distributed in the fertile surface soil (ca. 30 cm). The vegetation is arid-fynbos (Moll et al. 1984) and the site receives 135 mm year−1 of rain and has a Mediterranean-type climate, the lowest monthly rainfall falling in January and the highest (43 mm per month) in June, with a negligible amount of rain falling between mid-November and mid-March (Fig. 1). The mean annual temperature for the nearby town of Nieuwoudtville is 19 °C. To monitor soil volumetric moisture at the beginning of the study, four 5-TE moisture sensors (Decagon Devices, Pullman, USA) were buried at 0.2, 0.4, 0.6 and 0.8 m depths at 0.3 m distance from the plant and interfaced with EM50 data loggers (Decagon Devices) that logged at 2-h intervals. The 5-TE moisture sensor is designed to measure the water content, electrical conductivity, and temperature of a soil based on a 5-point dielectric calibration that provides dielectric permittivity measurements. Three-year-old A. linearis (n = 72) growing in three rows 4 m apart were tagged, leaving single “guard” rows of non-experimental plants between the tagged rows. To supplement groundwater, holes (70 mm diameter) were augered to 1.2 m depth at 0.25 m from the stem of each plant and 1.2-m-long PVC access tubes (60 mm Ø) inserted. After allowing the roots to adjust to the soil disturbance for 14 days, an automated drip irrigation system was set up to supply the deep soil layers with 0, 2 or 4 L day−1 (per plant) through the access tubes, dripped from 1800 to 1900 hours. Nutrients were then supplied as a solid fertilizer, coated onto dried sand. We made the fertilizer from 65 kg of medium-grained commercial sand (grade 30/10; Consol Minerals, South Africa) mixed with 30 mg kg−1 of 96 % Ca3(PO4)2, 70 mg kg−1 FePO4·2H2O, 600 mg kg−1 of 99 % NaNO3, 5.12 mg kg−1 of 98 atom % Na15NO3 (Sigma-Aldrich, St. Louis, MO, USA) and 12 L distilled water. The slurry was mixed in a cement mixer for 1 h and then air-dried in a temperature-controlled greenhouse (25 °C). The fertilizer had a δ15N value of 200 ± 1.6 ‰ (n = 4).
Monthly rainfall and average temperatures at Bloemfontein Farm, Niewoudtville, during the period May 2010 to April 2011. There was no rain between 8 November 2010, when fertilizer treatments were supplied and 26 February 2011(at 110 days post-fertilization). Rainfall on 27 and 28 February 2011 was 0.6 and 2.6 mm, respectively, at 111–112 days post-fertilization
Eight tagged plants in each row were left unfertilized, the remainder being supplied with nutrients either in shallow (n = 8) or deep (n = 8) soil layers. To avoid cross-contamination between treatments, gaps of 6 m with two rows of untreated plants separated the treatments along each row. For the deep fertilization treatment, 0.8 kg of fertilizer–sand mix was poured through the PVC access pipes. The deep-fertilizer mix was released from the 1.2-m PVC pipes inserted into the augered holes by gently pulling the pipes 0.2 m above the soil surface. For shallow-soil fertilization, the litter layer was removed and 0.8 kg of fertilizer–sand mix poured in a circle of radius 0.3 m in surface-soil to 0.1 m depth.
Foliar and soil sampling before and after supply of 2H-enriched water
To determine the initial foliar elemental contents, samples (ca. 50 g FW) of young apical shoots (ca. 0.2 m long) were cut from all experimental plants prior to fertilization (n = 10). At 14 and 125 days post-fertilization, sets of other 50 g samples of foliage were similarly cut from each experimental plant. All foliar samples were oven-dried at 70 °C for 48 h and milled in a Wiley mill using a 0.5-mm mesh (Arthur H. Thomas, Philadelphia, CA, USA). The milled material was analysed for tissue elemental concentrations as described below.
To determine HR, we used 2H-enriched water (e.g. Dawson et al. 2002) prepared from 2-mL aliquots of 99.9 atom % 2H2O (Sigma-Aldrich) diluted in 20 L of H2O. The δ2H value after dilution was 496 ‰. Initial stem and soil δ2H values were determined at 14 days post-fertilization on stem samples (ca. 10 mm diameter × 60 mm long, n = 5) of suberized wood collected at dusk (1800 hours) from six plants of each treatment, while soil cores (400 g) were augered directly under the canopy of each plant to 0.1 m depth, away from fertilized patches. Following this dusk sampling, we supplied 1 L of the diluted 2H2O through each PVC-access pipe at 1815 hours. A second set of stem and soil samples were collected predawn (0600 hours). Samples of irrigation water (n = 4) were also collected from the dam used for irrigation, and groundwater (n = 4) was sampled from a nearby borehole located 35 m from the study site for δ2H analysis. To avoid fractionation, the stem, soil and water samples were collected directly into airtight borosilicate tubes (Kimax–Kimble, Vineland, USA), sealed with Parafilm M (Sigma-Aldrich) and transported in cooler boxes with dry ice. All samples were kept in a cold room (4 °C) 3 days prior to analysis. Gravimetric moisture of shallow soil was measured both at 14 and 125 days by weighing sub-samples of shallow soil before oven-drying them at 70 °C for ca. 48 h before re-weighing them.
Foliar nutritional analysis
Foliar P was extracted by ashing pulverized leaf material at 480 °C for 8 h before dissolving in 1:1 (v/v) of HCl (Kalra 1998). Elemental concentrations of P were then determined using inductively coupled plasma atomic emission spectrometry (Varian Vista MPX, Mulgrave, Australia). Foliar [N], [15N] and [13C] ratios were determined using mass spectrometry. Between 1.900 and 2.000 mg of ground leaf sample was weighed into a 5 × 9 mm tin capsule (Santis Analytical, Teufen, Switzerland). The tin capsules were then combusted in a Thermo Flash EA 1112 series elemental analyzer coupled to a Delta Plus XP isotope ratio mass spectrometer via a Thermo Finnigan Conflo III control unit (Thermo Electron, Milan, Italy). International Atomic Energy Authority standards were used to determine the values.
δ2H analysis of water
Water was extracted from the soil and stem samples using the cryogenic vacuum distillation method of West et al. (2006). After extraction, the water samples were analysed for δ2H values using closed tube Zn reduction method (Coleman et al. 1982). Between 100 and 105 mg of Zn was loaded into oven-dried 6-mm flame-sealed break-seal vials before connecting them to an H2 sample preparation line developed after Coleman et al. (1982). The break-seal vials loaded with Zn were heated at 450 °C for 5 min with an HE2300 Metabo heat gun (Nürtingen, Germany), whilst evacuating them to 10−4 torr. The vials were then loaded with 2 × 10−6 L aliquots of water samples using Hirschmann micro-capillary pipettes (Eberstadt, Germany) and quickly returned to the vacuum line and frozen (ca. −200 °C) with liquid-N2 for 5 min. After evacuating impurities from the frozen samples to 10−4 torr, the vials were flame-sealed with the oxyacetylene torch. The loaded vials were then combusted in a furnace at 450 °C for 1 h to release H2 gas. The H2 gas was analysed with an isotopic ratio mass spectrometer (Finnigan Mat 252, Bremen, Germany). Internal standards (CTMP and DML ice) were run to calibrate the measurements relative to standard Mean Ocean Water (V-SMOW) and to correct for drift in the reference gas. The enrichment (Δ2H values) of shallow soil and stems after supplying 2H2O-tracer at dusk was calculated as the difference in δ2H values of dusk and predawn samples across the soil moisture and nutrient treatments. HR was inferred from predawn δ2H enrichment of shallow soil water, whilst nocturnal transpiration was inferred from the predawn δ2H enrichment of stem water samples.
The contributions of groundwater, 2H2O-tracer, irrigation and dusk shallow-soil water to the predawn shallow-soil water were estimated using the Isosource isotope mixing model (version 1.3.1; http://www.epa.gov/wed/pages/models.htm). In this model, all possible combinations of each source contribution (0–100 %) are examined in small increments. Combinations that sum to the observed mixture isotopic signatures within a small tolerance are considered to be feasible solutions, from which the frequency and range of potential source contributions can be determined (Phillips et al. 2005). We used predawn shallow-soil δ2H values as the “mixture” isotopic signatures, with source water having δ2H values of 496 ‰ for 2H2O, −48 ‰ for irrigation water, −65 ‰ for groundwater and the measured shallow-soil dusk δ2H values. We used an increment of 1 % and a tolerance of ±0.1 ‰ for estimating the proportions contributed by source waters to the predawn shallow-soil water. Since δ2H assays of shallow-soil waters were not done prior to irrigation of the experiment, the Isosource isotope mixing model could not be reliably used for estimating the proportions contributed by source waters prior to adding the 2H2O-tracer.
Statistical analysis
A 3 × 3 factorial ANOVA was used to test the effect of nutrient location, and irrigation levels using Statistica 10 (Statsoft, Tulsa, OK, USA). For comparing variables at 14 days with those at 125 days post-fertilization, a repeated measures ANOVA was used. A post hoc test using Fisher’s LSD test was used to identify significantly different means.
Results
Water-related measures
No rain fell at the study site for 110 days post-fertilization (Fig. 1). Although, 3.2 mm fell on 27 and 28 February 2011, this did not override the treatment differences in gravitational water content of shallow soil measured 125 days post-fertilization (see below). The volumetric water content at the beginning of the study was lower in the shallow soil than in the deeper soil (Fig. 2), suggesting the existence of a water potential gradient that could drive HR. Dam-water that was used for deep irrigation for 14 days prior to the experimental treatments had a δ2H value of −48 ± 1.8 ‰ (n = 4) and the δ2H value for rain was ca. −75.1 ‰, which is the volume-weighted average for the year (e.g. Jaeschke et al. 2011). The shallow-soil water at dusk, before the application of 2H2O, had different δ2H values for non-irrigated and irrigated treatments, the differences varying in response to fertilization (Fig. 3). For unfertilized plants and the deep-fertilized plants, the dusk δ2H values were higher with irrigation at 4 L plant−1 day−1, whilst for surface-fertilized plants the values became more negative with increased irrigation. After supply of 2H2O at dusk, the δ2H values of shallow-soil water at predawn were enriched to varying levels, expressed as Δ2H, calculated by subtracting predawn from dusk δ2H values (see Fig. 4a). The unfertilized and the deep-fertilized plants had higher shallow-soil Δ2H at lower rates of irrigation. In contrast, the Δ2H of shallow-soil under surface-fertilized plants increased with irrigation (Fig. 4a). Unlike soil Δ2H, stem predawn Δ2H showed no clear trend with irrigation, except for deep-fertilized plants where Δ2H increased with irrigation (Fig. 4b).
Volumetric soil moisture at varying soil depth as measured using EchoTE sensors at the beginning of the study. Soil moisture was measured at 2-h intervals using 5-TE moisture sensors buried at 0.2, 0.4, 0.6 and 0.8 m depths and 0.3 m from Aspalathus linearis stems. The values from each depth are averaged for the day before treatment application (n = 12)
δ2H values of surface-soil of unfertilized, surface-fertilized or the deep-fertilized Aspalathus linearis, receiving sub-surface (1 m) irrigation at 0, 2 or 4 L plant−1 day−1 at dusk and predawn. Symbols and error bars represent mean ± SE. Different letters indicate significantly different means (P < 0.05) as determined using a three way ANOVA with post hoc Fischer’s LSD tests. Rain had δ2H = −75 ‰, groundwater −65 ‰, irrigation water from an open earth dam had δ2H = −48 ‰; deuterium oxide had δ2H = 496 ‰
Enrichment (Δ2H) values of a surface-soil and b stems of unfertilized, surface-fertilized or the deep-fertilized Aspalathus linearis, receiving sub-surface (1 m) irrigation at 0, 2 or 4 L plant−1 day−1. Δ2H was calculated by subtracting dusk δ2H values prior to supply of 2H2O from predawn values 14 days post-fertilization. Symbols and error bars represent mean ± SE. Different letters indicate significantly different means (P < 0.05) as determined using two-way ANOVA with post hoc Fischer’s LSD tests. The dotted horizontal line represents zero enrichment. In the absence of significant irrigation × fertilizer interactions, values above lines in (b) show the average stem nocturnal Δ2H for plants supplied with different fertilizer treatments
The estimated proportional contribution of 2H2O-tracer to predawn surface-soil moisture ranged between 2 and 7 % (Table 1). This contribution followed the trends in shallow-soil Δ2H values (Fig. 4a). Although the proportion of tracer lifted to surface-soil by HR was relatively small, the estimated HR of deep water (sum of groundwater, irrigation water and tracer) was large, accounting for between 34 and 72 % of predawn surface-soil moisture (Table 1). In unfertilized and surface-fertilized plants, HR of deep water increased with moderate irrigation (i.e. 2 L plant−1 day−1), whereas with deep fertilization it changed rather little. The deep water contributed a smaller proportion of shallow-soil water of the unfertilized and unirrigated plants, compared to the unirrigated surface- and deep-fertilized plants.
Across the fertilization treatments, the gravimetric soil moisture of shallow-soil at 14 days was increased by deep irrigation (Fig. 5a). In contrast, at 125 days post-fertilization, the surface-soil moisture was reduced by deep irrigation (Fig. 5b). There was no interaction between irrigation and fertilizer treatments with regard to soil moisture content. Overall, surface-soil moisture was higher at 125 days than at 14 days post-fertilization (F (1,100) = 134.06; P < 0.001), possibly because of the rain that fell a few days before sample collection. Deep irrigation generally resulted in more negative foliar δ13C (Fig. 6a, b). Plant δ13C was used to provide a time-integrated estimate of intercellular to ambient CO2 mol fractions (C i /C a ), which are a long-term proxy for WUE (Farquhar et al. 1982, 1989; Brugnoli and Farquhar 2000). There was no significant effect of fertilizer treatment on δ13C values at either 14 days or 125 days post-fertilization.
Surface-soil gravimetric water content at dusk of unfertilized, surface-fertilized and deep-fertilized (1 m depth) Aspalathus linearis receiving sub-surface irrigation at 0, 2 or 4 L plant−1 day−1 at 1 m depth after a 14 days and b 125 days post-fertilization. Symbols and error bars are mean ± SE. Different letters indicate significantly different means (P < 0.05) as determined using two-way ANOVA with post hoc Fischer’s LSD tests. In the absence of significant irrigation × fertilizer interactions, inserted tables show the average soil H2O concentrations across irrigation levels
Foliar δ13C of unfertilized, surface-fertilized or deep-fertilized (1 m depth) Aspalathus linearis receiving sub-surface irrigation at 0, 2 or 4 L plant−1 day−1 at 1 m depth after a 14 days and b 125 days post-fertilization. Symbols and error bars represent mean ± SE. Different letters indicate significantly different means (P < 0.05) as determined using two-way ANOVA with post hoc Fischer’s LSD tests. In the absence of significant irrigation × fertilizer interactions, inserted tables show the average foliar δ13C across irrigation levels. Values above lines show the average foliar δ13C for plants supplied with different fertilizer treatments
Foliar nutrients
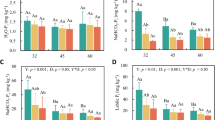
Foliar [N] 14 days post-fertilization (Fig. 7a) increased with higher rates of irrigation, but there was no significant effect of fertilization on foliar [N]. At 125 days post-fertilization, however, there were no significant differences in foliar [N] (Fig. 7b). Unlike foliar [N], which originates from soil sources, nodules and the supplied fertilizer, the changes in foliar δ15N values predominantly reflect the uptake of 15N-labelled fertilizer. At both 14 and 125 days post-fertilization (Fig. 8a, b), δ15N values were higher in both deep-fertilized and surface-fertilized plants than in the unfertilized plants, indicating uptake of the supplied 15N label. At 14 days post-fertilization, plants supplied with fertilization took up more 15N when irrigated, except in the case of those receiving surface-fertilization and 2 L plant−1 day−1. At 125 days post-fertilization, all fertilized plants had incorporated large amounts of 15N, especially the unirrigated surface-fertilized plants. In contrast to these surface-fertilized plants, δ15N increased with irrigation of the deep-fertilized plants.
Foliar N concentration of unfertilized, surface-fertilized or deep-fertilized (1 m depth) Aspalathus linearis receiving sub-surface irrigation at 0, 2 or 4 L plant−1 day−1 at 1 m depth after a 14 days and b 125 days post-fertilization. Symbols and error bars represent mean ± SE. Different letters indicate significantly different means (P < 0.05) as determined using two-way ANOVA with post hoc Fischer’s LSD tests. In the absence of significant irrigation × fertilizer interactions, inserted table in (a) shows the average foliar [N] across irrigation levels
Foliar δ15N of unfertilized, surface-fertilized or deep-fertilized (1 m depth) Aspalathus linearis receiving sub-surface irrigation at 0, 2 or 4 L plant−1 day−1 at 1 m depth after a 14 days and b 125 days post-fertilization. Each symbol and bar represents a mean ± SE. Different letters indicate significantly different means (P < 0.05) as determined using two-way ANOVA with post hoc Fischer’s LSD tests after log transformation
Overall, foliar [P] 14 days post-fertilization increased with deep irrigation in response to the added fertilizer with similar increases in foliar [P] with both surface- and deep-fertilization (Fig. 9a). At 125 days post-fertilization, however, foliar [P] did not vary with irrigation or fertilization (Fig. 9b). However, the overall foliar [P] of 291 ± 9 mg kg−1 measured at 125 days was significantly (F (1,54) = 82.53; P < 0.001) lower than 465 ± 21 mg kg−1 measured at 14 d post-fertilization.
Foliar P of unfertilized, surface-fertilized or deep-fertilized (1 m depth) Aspalathus linearis receiving sub-surface irrigation at 0, 2 or 4 L plant−1 day−1 at 1 m depth after a 14 days and b 125 days post-fertilization. Each circle and bar represents a mean ± SE. In the absence of significant irrigation × fertilizer interactions, inserted tables show the average foliar P across irrigation levels. Different letters indicate significantly different means (P < 0.05) as determined using two-way ANOVA with post hoc Fischer’s LSD tests. Inserted table shows foliar P across irrigation levels. In the absence of significant irrigation × fertilizer interactions, inserted table in (a) shows the average foliar [P] across irrigation levels
Discussion
We used 2H2O as a tracer for HR (e.g. Dawson 1993; Brooks et al. 2002) and found evidence for the occurrence of HR and nocturnal transpiration in the arid fynbos legume, A. linearis. Since 2H2O uptake responded to irrigation and fertilization treatments, it is unlikely that capillary flow through the sands explains the overnight redistribution of 2H2O, as also concluded previously in a similar edaphic context (Hawkins et al. 2009).
The differences in surface-soil δ2H values at dusk 14 days post-fertilization were caused by a combination of differences in HR and fractionation of residual shallow-soil water exposed to daytime fractionation via evaporative losses. Soil water may be variably bound to soil particles (Sun et al. 2009; Lund 1959; Tyree 2003), particularly clay (Kovda 1983; Xianliang and Yunsheng 1983) protecting it from evaporative fractionation. The dusk δ2H values are therefore a product of a combination of HR during the preceding night, the utilisation of surface-soil water by plants during the day and the evaporative fractionation of residual soil water not bound to particles. Unlike surface-soil natural abundance δ2H values and gravimetric moisture, which both may accumulate over time, the redistributed 2H2O-tracer indicates the short-term (<12 h) magnitude and direction of HR and was used to estimate the contribution of HR to surface-soil moisture (Fig. 4). We estimated that nocturnal HR contributed the bulk of surface-soil water in deep-irrigated plants and that HR was increased in response to both surface- and deep fertilization of unirrigated treatments (Table 1).
Decreased surface-soil Δ2H in unfertilized plants with increased rates of deep irrigation may be due to a combination of dilution of the supplied label by unlabelled irrigation water and plant uptake of surface water. The estimation of total redistribution of deep water (Table 1) and the increase in shallow-soil moisture with deep irrigation (Fig. 5a), however, indicates that irrigation did increase HR. Furthermore, the increased deep irrigation led to more negative δ13C values, also suggesting increased daytime transpiration and decreased WUE (Fig. 6). Plant uptake of surface-soil moisture may also have been increased, however, in response to deep irrigation. Indeed, the stem length increment (cm day−1) over 14 days for unfertilized plants was 0.10 ± 0.09, 0.19 ± 0.1 and 0.49 ± 0.20 for the 0, 2 and 4 L plant−1 day−1 irrigation rates, respectively. This increased growth due to the greater availability of water may contribute to utilisation of shallow-soil moisture. Thus deep irrigation increased HR, but probably also increased the utilisation of surface-soil moisture due to increased growth of the plants.
Supply of fertilizers to surface and deep soil increased HR of deep water in unirrigated plants. The estimated proportion of surface-soil water lifted overnight by HR by deep-irrigated plants was, however, not increased by surface- or deep-fertilization. Thus, HR was increased primarily by deep irrigation, and nutrition only increased HR in the absence of additional irrigation water (Table 1; Fig. 4a). The results also suggest that the placement of fertilizers (surface versus deep) influenced the engagement of HR and nocturnal transpiration (Fig. 4a, b). Irrigated plants supplied with deep fertilization engaged in both HR and nocturnal transpiration. In contrast, there was no evidence for uptake of 2H2O-labelled irrigation water through nocturnal transpiration in unfertilized or surface-fertilized plants, either directly from deep soil or indirectly from surface soil. During the following diurnal period, however, uptake of this water is likely to have occurred (not measured). In situations where nutrients are most abundant in surface soils, it may be strategic to favour HR and recharge of surface moisture, which would be compromised by simultaneous nocturnal transpirational uptake of surface-soil moisture. A trade-off between HR and nocturnal transpiration has been reported previously (Richards and Caldwell 1987; Bauerle et al. 2008; Howard et al. 2009; Prieto et al. 2010) and may operate when water supplies are limited.
During the summer drought period, the increased HR of surface-fertilized plants 14 days post-fertilization was associated with increased acquisition of shallow-soil nutrients. This was partially evident from higher δ15N values and tissue [P]. Because of the generally low P in CFR soils (e.g. Witkowski and Mitchells 1987), the lower foliar P at 125 days post-fertilization compared to 14 days possibly indicates the exhaustion of the relatively small amount of fertilizer-P supplied to the rhizosphere. Alternatively, chemical change in the forms of phosphate and fixation by the soil minerals may partly explain the decrease of P at 125 days relative to 14 days. Increased acquisition of these inorganic fertilizer forms is independent of mineralisation and must reflect access either through increased mobility of the nutrients in the soil or increased root interception of nutrients. Increased nutrient mobility due to higher soil moisture includes both diffusive and mass-flow mobility. Light rains (3.2 mm) preceding the harvest of plant material at 125 days may have facilitated the striking accumulation of 15N into young leaves of unirrigated shallow-fertilized plants, whereas the faster-growing irrigated and shallow-fertilized plants may have partially depleted 15N surface soils prior to the rain.
There was also evidence that the deep-fertilized plants increased their uptake of 15N and P in response to irrigation. As with surface-fertilized plants, this could be due to increased nutrient mobility or root growth. Since we interpret the stem Δ2H as indicating greater nocturnal water flux in these plants, we suggest that nutrient uptake may have been increased by mass-flow driven by nocturnal water flux (i.e. transpiration and HR) from deep soils. The increased availability of soil water with irrigation could have also increased nutrient diffusion and mass-flow during the day, particularly given the low WUE associated with the more negative foliar δ13C values (Fig. 6). Although diffusion and mass-flow of N is well known, P mobility has been considered limited in most soils (e.g. Bieleski 1973; Lambers et al. 1998) owing to low diffusion coefficients (Barber and Olson 1968; Clarkson 1981). P-mobility may, however, be relatively high in low P-binding soils. Examples of such soils are the low-clay, highly weathered and leached soils of the CFR (Witkowski and Mitchell 1987; Rebelo 1996; Rebelo et al. 2006; Goldblatt 1997) to which A. linearis is native. Plant water flux has also been linked to P acquisition in tropical species (Cernusak et al. 2011). Thus, plants supplied with deep fertilization apparently increased both nocturnal transpiration and HR, although the combination of these two processes was only manifested with irrigation.
If HR, diurnal and nocturnal transpiration have functional roles in nutrient acquisition through mass-flow, it is likely that these physiological activities will be regulated by nutrient availability. Evidence is accruing for nutritional regulation of diurnal transpiration (Clarkson et al. 2000; Gorska et al. 2008) and this has been linked to mass-flow acquisition of nutrients (Cramer et al. 2008, 2009; Matimati et al. 2014). The induction of HR by fertilizers in the shallow soil also indicates that this process is inducible by nutrition. This is at odds with the view that HR is a passive process, dependent only on the water potential gradient within the soil. Instead, we suggest a role for nutritional regulation of HR, possibly functioning through aquaporin regulation of water uptake from the soil or release to the soil (e.g. Carvajal et al. 1996; Hoarau et al. 1996; Clarkson et al. 2000) or indirectly through interactions with nocturnal transpiration (Prieto et al. 2010; Howard et al. 2009). Since the placement of fertilizer in the soil modulated stem Δ2H, we conclude that nocturnal transpiration is regulated by nutrient availability, as also reported by Kupper et al. (2012). The fertilizer supplied included NO −3 , which is known to partially regulate stomatal conductance (Wilkinson et al. 2007; Gloser et al. 2007; Gorska et al. 2008; Garrish et al. 2010). Further work is required to ascertain the physiological mechanism through which nutrients regulate HR and nocturnal transpiration and how these processes may interact.
Variation in the functional significance of HR and nocturnal transpiration in acquiring nutrients situated in shallow versus deep soil profiles accords with the conclusion of Prieto et al. (2012) that it is the selective placement of roots in nutrient-rich patches and deeper moist soils that most strongly determine nutrient capture and plant performance during summer drought. Many plants employ mixed strategies, in which the development of deep roots is prioritized to ensure access to deep water and leached nutrients (Lynch 2013), but with some shallow root development being maintained to enable the acquisition of shallow-soil nutrients (Lynch and Brown 2001; Lynch 2011; Richardson et al. 2011), possibly with participation of HR.
Conclusion
We conclude that A. linearis utilised transpiration and HR to increase acquisition of both shallow and deep soil nutrients. We suggest that both diurnal and nocturnal transpiration are particularly important for promoting mass-flow of nutrients by these plants growing in nutrient poor sands. HR responded to both surface fertilization and deep fertilization, indicating that this process may have dual functionality in increasing uptake of deep nutrients by driving water fluxes at night, and also by mobilizing surface nutrients. The fact that both nocturnal transpiration and HR responded to fertilizer placement indicates that these processes are partially regulated by nutrient availability.
References
Aanderud ZT, Richards JH (2009) Hydraulic redistribution may stimulate decomposition. Biogeochemistry 95:323–333. doi:10.1007/s10533-009-9339-3
Allen SC, Jose S, Nair PKR, Brecke BJ, Nkedi-kizza P, Ramsey CL (2004) Safety-net role of tree roots: evidence from a pecan (Carya illinoensis K. Koch)—cotton (Gossypium hirsutum L.) alley cropping system in the southern United States. Forest Ecol Manag 192:395–407. doi:10.1016/j.foreco.2004.02.009
Armas C, Kim JH, Bleby TM, Jackson RB (2012) The effect of hydraulic lift on organic matter decomposition, soil nitrogen cycling, and nitrogen acquisition by a grass species. Oecologia 168:11–22. doi:10.1007/s00442-011-2065-2
Barber SA (1995) Soil nutrient bioavailability: a mechanistic approach, (2nd edn). Wiley, New York
Barber SA, Olson RA (1968) Fertilizer use on corn. In: Nelson LB, McVickar MH, Munson RD, Seatz LF, Tisdale SL, White WC (eds) Changing patterns in fertilizer use. Soil Science Society of America, Madison, pp 163–188
Bauerle TL, Richards JH, Smart DR, Eissenstat DM (2008) Importance of internal hydraulic redistribution for prolonging the lifespan of roots in drying soil. Plant Cell Environ 31:177–186. doi:10.1111/j.1365-3040.2007.01749.x
Bieleski RL (1973) Phosphate pools, phosphate transport, and phosphate availability. Ann Rev Plant Physiol 24:225–252. doi:10.1146/annurev.pp.24.060173.001301
Brooks JR, Meinzer FC, Coulombe R (2002) Hydraulic redistribution of soil water during summer drought in two contrasting Pacific Northwest coniferous forests. Tree Physiol 22:1107–1117. doi:10.1093/treephys/22.15-16.1107
Brugnoli E, Farquhar GD (2000) Photosynthetic fractionation of carbon isotopes, in: Leegood RC, Sharkey TD, von Caemmerer S (eds) Photo-synthesis: physiology and metabolism. Kluwer, The Netherlands, pp 399–434
Burgess SSO, Bleby TM (2006) Redistribution of soil water by lateral roots mediated by stem tissues. J Exp Bot 57:3283–3291. doi:10.1093/jxb/erl085
Burgess SSO, Adams MA, Turner NC, Ong CK (1998) The redistribution of soil water by tree root systems. Oecologia 115:306–311. doi:10.1007/s004420050521
Burgess SSO, Adams MA, Bleby TM (2000) Measurement of sap flow in roots of woody plants: a commentary. Tree Physiol 20:909–913. doi:10.1093/treephys/20.13.909
Caldwell MM, Dawson TE, Richards JH (1998) Hydraulic lift: consequences of water efflux from the roots of plants. Oecologia 113:151–161. doi:10.1007/s004420050363
Cardon ZG, Gage DJ (2006) Resource exchange in the rhizosphere: molecular tools and the microbial perspective. Ann Rev Ecol Evol Syst 37:459–488. doi:10.1146/annurev.ecolsys.37.091305.110207
Carvajal M, Cooke DT, Clarkson DT (1996) Responses of wheat plants to nutrient deprivation may involve the regulation of water-channel function. Planta 199:372–381. doi:10.1093/jexbot/51.342.61
Cernusak LA, Winter K, Turner BL (2011) Transpiration modulates phosphorus acquisition in tropical tree seedlings. Tree Physiol 31:878–885. doi:10.1093/treephys/tpr077
Clarkson DT (1981) Nutrient interception and transport by root systems. In: Johnson CB (ed) Physiological factors limiting plant productivity. Butterworths, London, pp 307–314
Clarkson DT, Carvajal M, Henzler T, Waterhouse RN, Smyth AJ, Cooke DT, Steudle E (2000) Root hydraulic conductance: diurnal aquaporin expression and the effects of nutrient stress. J Exp Bot 342:61–70. doi:10.1093/jexbot/51.342.61
Coleman ML, Shepherd TJ, Durham JJ, Rouse JE, Moore GR (1982) Reduction of water with zinc for hydrogen isotope analysis. Anal Chem 54:993–995. doi:10.1021/ac00243a035
Cramer MD, Hoffmann V, Verboom GA (2008) Nutrient availability moderates transpiration in Ehrharta calycina. New Phytol 179:1048–1057
Cramer MD, Hawkins H-J, Verboom AG (2009) The importance of nutritional regulation of plant water flux. Oecologia 161:15–24. doi:10.1007/s00442-009-1364-3
Dawson TE (1993) Hydraulic lift and water use by plants: implications for water balance, performance and plant–plant interactions. Oecologia 95:565–574. doi:10.1007/BF00317442
Dawson TE (1997) Water loss from tree influences soil water nutrient status and plant performance. In: Flores EH, Lynch JP, Eissenstat D (eds) Radical biology: advances and perspectives on the function of plant roots, vol 18. American Society of Plant Physiologists, Rockville, pp 235–250
Dawson TE, Mambelli S, Plamboeck AH, Templer PH, Tu KP (2002) Stable isotopes in plant ecology. Annu Rev Ecol Syst 33:507–559. doi:10.1146/annurev.ecolsys.33.020602.095451
Farquhar GD, O’Leary MH, Berry JA (1982) On the relationship between carbon isotope discrimination and the intercellular carbon dioxide concentration in leaves. Aust J Plant Physiol 9:121–137. doi:10.1071/PP9820121
Farquhar GD, Ehleringer JR, Hubick KT (1989) Carbon isotope discrimination and photosynthesis. Annu Rev Plant Phys 40:503–537. doi:10.1146/annurev.pp.40.060189.002443
Garrish V, Cernusak LA, Winter K, Turner BL (2010) Nitrogen to phosphorus ratio of plant biomass versus soil solution in a tropical pioneer tree, Ficus insipida. J Exp Bot 61:3735–3748. doi:10.1093/jxb/erq183
Gérard A (2010) Habitat conditions of wild Rooibos tea (Aspalathus linearis): Environmental abiotic and biotic drivers of its performance. Ms thesis, Universität Hamburg
Gloser V, Zwieniecki MA, Orians C, Holbrook NM (2007) Dynamic changes in root hydraulic properties in response to nitrate availability. J Exp Bot 58:2409–2415. doi:10.1093/jxb/erm118
Goldblatt P (1997) Floristic diversity in the Cape Flora of South Africa. Biodivers Conserv 6:359–377. doi:10.1023/A:1018360607299
Gorska A, Ye Q, Holbrook NM, Zwieniecki MA (2008) Nitrate control of root hydraulic properties in plants: translating local information to whole plant response. Plant Physiol 148:1159–1167. doi:10.1104/pp.108.122499
Hawkins H-J, Hettasch H, West AG, Cramer MD (2009) Hydraulic redistribution by Protea “Sylvia” (Proteaceae) facilitates soil water replenishment and water acquisition by an understorey grass and shrub. Funct Plant Biol 36:752–760. doi:10.1071/FP09046
Hawkins H-J, Malgas R, Biénabe E (2011) Ecotypes of wild rooibos (Aspalathus linearis (Burm. F) Dahlg., Fabaceae) are ecologically distinct. S Afr J Bot 77:360–370. doi:10.1016/j.sajb.2010.09.014
Hoarau J, Barthes L, Bousser A, Deleens E, Prioul JL (1996) Effect of nitrate on water transfer across roots of nitrogen pre-starved maize seedlings. Planta 200:405–415. doi:10.1007/BF00231396
Howard AR, Van Iersel MW, Richards JH, Donovan LA (2009) Nocturnal transpiration can decrease hydraulic redistribution. Plant Cell Environ 32:1060–1070. doi:10.1111/j.1365-3040.2009.01988.x
Hultine KR, Cable WL, Burgess SSO, Williams DG (2003) Hydraulic redistribution by deep roots of a Chihuahuan Desert phreatophyte. Tree Physiol 23:353–360. doi:10.1093/treephys/23.5.353
Hultine KR, Scott RL, Cable WL, Goodrich DC, Williams DG (2004) Hydraulic redistribution by a dominant, warm-desert phreatophyte: seasonal patterns and response to precipitation pulses. Funct Ecol 18:530–538. doi:10.1111/j.0269-8463.2004.00867.x
Jackson RB, Caldwell MM (1993) The scale of nutrient heterogeneity around individual plants and its quantification with geostatistics. Ecol 74:612–614. doi:10.2307/1939320
Jackson RB, Sperry JS, Dawson TE (2000) Root water uptake and transport: using physiological processes in global predictions. Trends Plant Sci 5:482–488. doi:10.1016/S1360-1385(00)01766-0
Jaeschke JB, Scholl MA, Cozzarelli IM, Masoner JR, Christenson S, Qi H (2011) Stable-isotope ratios of hydrogen and oxygen in precipitation at Norman, Oklahoma, 1996–2008: U.S. Geological Survey Scientific Investigations Report, 2011–5262
Jobbágy EG, Jackson RB (2000) The vertical distribution of soil organic carbon and its relation to climate and vegetation. Ecol Appl 10:423–436. doi:10.1890/1051-0761(2000)010[0423:TVDOSO]2.0.CO;2
Jobbágy EG, Jackson RB (2004) The uplift of soil nutrients by plants: biogeochemical consequences across scales. Ecology 85:2380–2389. doi:10.1890/03-0245
Kalra YP (1998) Handbook of standard methods of plant analysis. CRC Press, Boca Raton
Kovda BA (1983) Agrology principle. Science Press, Beijing, p 17
Kupper P, Rohula G, Saksing L, Sellin A, Lõhmus K, Ostonen I, Helmisaari H-S, Sõber A (2012) Does soil nutrient availability influence nocturnal water flux of aspen saplings? Environ Exp Bot 82:37–42. doi:10.1016/j.envexpbot.2012.03.013
Kurz-Besson C, Otieno D, Lobo do Vale R, Siegwolf R, Schmidt M, Herd A, Nogueira C, David TS, David JS, Tenhunen J, Pereira JS, Chaves M (2006) Hydraulic lift in cork oak trees in a savannah-type Mediterranean ecosystem and its contribution to the local water balance. Plant Soil 282:361–378. doi:10.1007/s11104-006-0005-4
Lambers H, Chapin III FS, Pons TL (1998) Plant physiological ecology. Springer, New York, pp 239–298. doi:10.1007/978-1-4757-2855-2
Leffler AJ, Ivans CY, Ryel RJ, Caldwell MM (2004) Gas exchange and growth responses of the desert shrubs Artemisia tridentata and Chrysothamnus nauseosus to shallow- vs. deep-soil water in a glasshouse experiment. Environ Exp Bot 51:9–19. doi:10.1016/S0098-8472(03)00041-8
Leffler AJ, Peek MS, Ryel RJ, Ivans CY, Caldwell MM (2005) Hydraulic redistribution through the root systems of senesced plants. Ecol 86:633–642. doi:10.1890/04-0854
Lehto T, Zwiazek JJ (2011) Ectomycorrhizas and water relations of trees: a review. Mycorrhiza 21:71–90. doi:10.1007/s00572-010-0348-9
Ludwig F, Dawson TE, Kroon H, Berendse F, Prins HHT (2003) Hydraulic lift in Acacia tortilis trees on an East African savanna. Oecologia 134:293–300. doi:10.1007/s00442-002-1119-x
Ludwig F, Dawson TE, Prins HHT, Berendse F, de Kroon H (2004) Below-ground competition between trees and grasses may overwhelm the facilitative effects of hydraulic lift. Ecol Lett 7:623–631. doi:10.1111/j.1461-0248.2004.00615.x
Lund ZF (1959) Available water-holding capacity of alluvial soils in Louisiana. Soil Sci Soc Am J 23:1–3. doi:10.2136/sssaj1959.03615995002300010009x
Lynch JP (2011) Root phenes for enhanced soil exploration and phosphorus acquisition: tools for future crops. Plant Physiol 156:1041–1049. doi:10.1104/pp.111.175414
Lynch JP (2013) Steep, cheap and deep: an ideotype to optimize water and N acquisition by maize root systems. Ann Bot 112:347-357. doi:10.1093/aob/mcs293
Lynch JP, Brown KM (2001) Topsoil foraging—an architectural adaptation of plants to low phosphorus availability. Plant Soil 237:225–237. doi:10.1023/A:1013324727040
Matimati I, Verboom GA, Cramer MD (2014) Nitrogen regulation of transpiration controls mass-flow acquisition of nutrients. J Exp Bot 65:159–168. doi:10.1093/jxb/ert367
Matzner SL, Richards JH (1996) Sagebrush (Artemisia tridentata Nutt.) roots maintain nutrient uptake capacity under water stress. J Exp Bot 47:1045–1056. doi:10.1093/jxb/47.8.1045
Moll EJ, Campbell BM, Cowling RM, Bossi L, Jarman ML, Boucher C (1984) Description of major vegetation categories in and adjacent to the Fynbos biome. National Scientific Programmes Unit: CSIR, SANSP Report 83, Mar 1984
Morton JF (1983) Rooibos tea, Aspalathus linearis a caffeinless, low-tannin beverage. Econ Bot 37:164–173
Muñoz MR, Squeo F, León MF, Tracol Y, Gutiérrez JR (2008) Hydraulic lift in three shrub species from the Chilean coastal desert. J Arid Environ 72:624–632. doi:10.1016/j.jaridenv.2007.09.006
Nadezhdina N, David TS, David JS, Ferreira MA, Dohnal M, Tesa M, Gartner K, Leitgeb E, Nadezhdin V, Cermak J et al (2010) Trees never rest: the multiple facets of hydraulic redistribution. Ecohydrol 3:431–444. doi:10.1002/eco.148
Nambiar EKS (1976) Uptake of Zn65 from dry soil by plants. Plant Soil 44:267–271. doi:10.1007/BF00016978
Neumann RB, Cardon ZG (2012) The magnitude of hydraulic redistribution by plant roots: a review and synthesis of empirical and modelling studies. New Phytol 194:337–352. doi:10.1111/j.1469-8137.2012.04088.x
Pang J, Wang Y, Lambers H, Tibbett M, Siddique KH, Ryan MH (2013) Commensalism in an agroecosystem: hydraulic redistribution by deep-rooted legumes improves survival of a droughted shallow-rooted legume companion. Physiol Plant. doi:10.1111/ppl.12020
Phillips DL, Gregg JW (2003) Source partitioning using stable isotopes: coping with too many sources. Oecologia 136:261–269. doi:10.1007/s00442-003-1218-3
Phillips DL, Newsome SD, Gregg JW (2005) Combining sources in stable isotope mixing models: alternative methods. Oecologia 144:520–527. doi:10.1007/s00442-004-1816-8
Prieto I, Kikvidze Z, Pugnaire FI (2010) Hydraulic lift: soil processes and transpiration in the Mediterranean leguminous shrub Retama sphaerocarpa (L.) Boiss. Plant Soil 329:447–456. doi:10.1007/s11104-009-0170-3
Prieto I, Armas C, Pugnaire FI (2012) Water release through plant roots: new insights into its consequences at the plant and ecosystem level. New Phytol 193:830–841. doi:10.1111/j.1469-8137.2011.04039.x
Querejeta JI, Egerton-Warburton LM, Allen MF (2003) Direct nocturnal water transfer from oaks to their mycorrhizal symbionts during severe soil drying. Oecologia 134:55–64. doi:10.1007/s00442-002-1078-2
Rebelo AG (1996) Fynbos biome. In: Low AB, Rebelo AG (eds) Vegetation of South Africa, Lesotho and Swaziland. Department of Environmental Affairs and Tourism, South Africa, pp 62–74
Rebelo AG, Boucher C, Helme N, Mucina L, Rutherford MC (2006) Fynbos biome. In: Mucina L, Rutherford MC (eds) The vegetation of South Africa Lesotho and Swaziland. Strelitzia, vol. 19, pp 53–219
Richards JH, Caldwell MM (1987) Hydraulic lift: substantial nocturnal water transport between soil layers by Artemisia tridentata roots. Oecologia 73:486–489. doi:10.1007/BF00379405
Richardson AE, Lynch JP, Ryan PR et al (2011) Plant and microbial strategies to improve the phosphorus efficiency of agriculture. Plant Soil 349:121–156. doi:10.1007/s11104-011-0950-4
Rose TJ, Rengel Z, Ma Q et al (2008) Hydraulic lift by canola plants aids P and K uptake from dry topsoil. Aust J Agric Res 59:38–45. doi:10.1071/AR07146
Ryel RJ (2004) Hydraulic redistribution. In: Esser K, Lüttge U, Beyschlag W (eds) Prog Bot, vol. 65, pp 413–435. doi: 10.1007/978-3-642-18819-0_17
Ryel RJ, Caldwell MM, Yoder CK, Or D, Leffler AJ (2002) Hydraulic redistribution in a stand of Artemisia tridentata: evaluation of benefits to transpiration assessed with a simulation model. Oecologia 130:173–184. doi:10.1007/s004420100794
Ryel RJ, Leffler AJ, Peek MS, Ivans CY, Caldwell MM (2004) Water conservation in Artemisia tridentata through redistribution of precipitation. Oecologia 141:335–345. doi:10.1007/s00442-003-1421-2
Scholz FG, Bucci SJ, Goldstein G, Meinzer FC, Franco AC (2002) Hydraulic redistribution of soil water by neotropical savanna trees. Tree Physiol 22:603–612. doi:10.1093/treephys/22.9.603
Sekiya N, Yano K (2004) Do pigeon pea and sesbania supply groundwater to intercropped maize through hydraulic lift?-Hydrogen stable isotope investigation of xylem waters. Field Crops Res 86:167–173. doi:10.1016/j.fcr.2003.08.007
Sekiya N, Araki H, Yano K (2011) Applying hydraulic lift in an agroecosystem: forage plants with shoots removed supply water to neighbouring vegetable crops. Plant Soil 341:39–50. doi:10.1007/s11104-010-0581-1
Shen Y, Zhang Y, Li S (2011) Nutrient effects on diurnal variation and magnitude of hydraulic lift in winter wheat. Agric Water Manag 98:1589–1594. doi:10.1016/j.agwat.2011.05.012
Smart DR, Carlisle E, Goebel M, Nuñez BA (2005) Transverse hydraulic redistribution by a grapevine. Plant Cell Environ 28:157–166. doi:10.1111/j.1365-3040.2004.01254.x
Snyder KA, James JJ, Richards JH, Donovan LA (2008) Does hydraulic lift or nighttime transpiration facilitate nitrogen acquisition? Plant Soil 306:159–166. doi:10.1007/s11104-008-9567-7
Sun G, Sun J, Zhou G (2009) Editorial: water and carbon dynamics in selected ecosystems in China. Agric For Meteorol 149:1789–1790
Tyree MT (2003) Matric potential. Encyclopaedia of water science. Marcel Dekker, Inc., New York, pp 615–617. http://www.nrs.fs.fed.us/pubs/jrnl/pristine/2003/ne_2003_tyree_007p.pdf
Wang X, Tang C, Guppy CN, Sale PWG (2009) The role of hydraulic lift and subsoil P placement in P uptake of cotton (Gossypium hirsutum L.). Plant Soil 325:263–275. doi:10.1007/s11104-009-9977-1
Warren JM, Meinzer FC, Brooks JR, Domec JC, Coulombe R (2007) Hydraulic redistribution of soil water in two old-growth coniferous forests: quantifying patterns and controls. New Phytol 173:753–765. doi:10.1111/j.1469-8137.2006.01963.x
West AG, Patrickson SJ, Ehleringer JR (2006) Water extraction times for plant and soil materials used in stable isotope analysis. Rapid Commun Mass Sp 20:1317–1321. doi:10.1002/rcm.2456
Wilkinson S, Bacon MA, Davies WJ (2007) Nitrate signalling to stomata and growing leaves: interactions with soil drying, ABA and xylem sap pH in maize. J Exp Bot 58:1705–1716. doi:10.1002/rcm.2456
Witkowski ETF, Mitchell DT (1987) Variations in soil phosphorus in the fynbos biome. S Afr J Ecol 75:1159–1171. doi:10.2307/2260320
Xianliang Y, Yunsheng C (1983) Soil Physics. Agriculture Press, Beijing, pp 1–14
Author information
Authors and Affiliations
Corresponding author
Additional information
Communicated by Todd E Dawson.
The authors have contributed equally to this paper.
Rights and permissions
About this article
Cite this article
Matimati, I., Anthony Verboom, G. & Cramer, M.D. Do hydraulic redistribution and nocturnal transpiration facilitate nutrient acquisition in Aspalathus linearis?. Oecologia 175, 1129–1142 (2014). https://doi.org/10.1007/s00442-014-2987-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00442-014-2987-6