Abstract
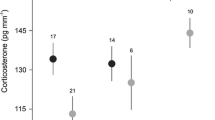
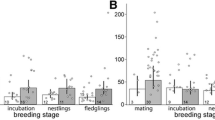
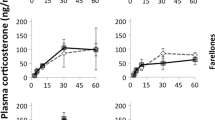
Because glucocorticoid (stress) hormones fundamentally affect various aspects of the behaviour, life history and fitness of free-living vertebrates, there is a need to understand the environmental factors shaping their variation in natural populations. Here, we examined whether spatial heterogeneity in breeding territory quality affected the stress of colonial king penguin (Aptenodytes patagonicus). We assessed the effects of local climate (wind, sun and ambient temperature) and social conditions (number of neighbours, distance to neighbours) on the baseline levels of plasma total corticosterone (CORT) in 77 incubating and 42 chick-brooding birds, breeding on territories of central or peripheral colony location. We also assessed the oxidative stress status of a sub-sample of central vs. peripheral chick-brooders to determine whether chronic stress arose from breeding on specific territories. On average, we found that brooders had 55 % higher CORT levels than incubators. Regardless of breeding status, central birds experienced greater social density (higher number of neighbours, shorter distance between territories) and had higher CORT levels than peripheral birds. Increasing social density positively explained 40 % of the variation in CORT levels of both incubators and brooders, but the effect was more pronounced in brooders. In contrast, climate was similar among breeding territories and did not significantly affect the CORT levels of breeding birds. In brooders, oxidative stress status was not affected by local density or weather conditions. These results highlight that local heterogeneity in breeding (including social) conditions may strongly affect the stress levels of breeding seabirds. The fitness consequences of such variation remain to be investigated.


Similar content being viewed by others
References
Angelier F, Shaffer SA, Weimerskirch H, Chastel O (2006) Effects of age, breeding experience and senescence on corticosterone and prolactin levels in a long-lived seabird: the wandering albatross. Gen Comp Endocr 149:1–9
Bernard SF, Mioskowski E, Groscolas R (2002) Blockade of fatty acid oxidation mimics phase II–phase III transition in a fasting bird, the king penguin. Am J Physiol Reg I 283:R144–R152
Bize P, Stocker A, Jenni-Eiermann S, Gasparini J, Roulin A (2010) Sudden weather deterioration but not brood size affects baseline corticosterone levels in nestling Alpine swifts. Horm Behav 58:591–598
Bókony V, Lendvai Á, Liker A, Angelier F, Wingfield JC, Chastel O (2009) Stress response and the value of reproduction: are birds prudent parents? Am Nat 173:589–598
Bonier F, Moore IT, Martin PR, Robertson RJ (2009) The relationship between fitness and baseline glucocorticoids in a passerine bird. Gen Comp Endocr 163:208–213
Boonstra R (2004) Coping with changing northern environments: the role of the stress axis in birds and mammals. Integr Comp Biol 44:95–108
Boonstra R (2013) Reality as the leading cause of stress: rethinking the impact of chronic stress in nature. Funct Ecol 27:11–23
Boonstra R, Hik D, Singleton GR, Tinnikov A (1998) The impact of predator-induced stress on the snowshoe hare cycle. Ecol Monogr 75:371–394
Breuner CW, Delehanty B, Boonstra R (2013) Evaluating stress in natural populations of vertebrates: total CORT is not good enough. Funct Ecol 27:24–36
Bried J, Jouventin P (2001) The king penguin Aptenodytes patagonicus, a non-nesting bird which selects its breeding habitat. Ibis 143:670–673
Burnham KP, Anderson DR (2002) Model selection and multimodel inference: a practical information-theoretic approach. Springer, New York
Budaev SV (2010) Using principal components and factor analyses in animal behavior research: caveats and guidelines. Ethology 116:472–480
Cherel Y, Robin J-P, Walch O, Karmann H, Netchitailo P, Le Maho Y (1988) Fasting in king penguin. I. Hormonal and metabolic changes during breeding. Am J Physiol Reg I 254:R170–R177
Clinchy M, Sheriff MJ, Zanette LY (2013) Predator-induced stress and the ecology of fear. Funct Ecol 27:56–65
Côté SD (2000) Aggressiveness in king penguins in relation to reproductive status and territory location. Anim Behav 59:813–821
Côté SD, Dewasmes G (1999) Do sleeping king penguins influence the movement of conspecifics through a colony? Polar Biol 22:13–16
Creel S, Dantzer B, Goymann W, Rubenstein DR (2013) The ecology of stress: effects of the social environment. Funct Ecol 27:66–80
Dantzer B, Newman AEM, Boonstra R, Palme R, Boutin S, Humprhies MM, McAdam AG (2013) Density triggers maternal hormones that increase adaptive offspring growth in a wild mammal. Science 340:1215–1217
Descamps S, Gauthier-Clerc M, Le Bohec C, Gendner J-P, Le Maho Y (2005) Impact of predation on king penguin Aptenodytes patagonicus in Crozet Archipelago. Polar Biol 28:303–310
Descamps S, Le Bohec C, Le Maho Y, Gendner J-P, Gauthier-Clerc M (2009) Relating demographic performance to breeding-site location in king penguin. Condor 111:81–87
Gauthier-Clerc M, Le Maho Y, Gendner J-P, Durant J, Handrich Y (2001) State-dependent decisions in long-term fasting king penguins, Aptenodytes patagonicus, during courtship and incubation. Anim Behav 62:661–669
Geiger S, Le Vaillant M, Lebard T, Reichert S, Stier A, Le Maho Y, Criscuolo F (2012) Catching-up but telomere loss: half-opening the black box of growth and ageing trade-off in wild king penguin chicks. Mol Ecol 21:1500–1510
Groscolas R, Decrock F, Thil M-A, Fayolle C, Boissery C, Robin J-P (2000) Refeeding signal in fasting-incubating king penguins: changes in behavior and egg temperature. Am J Physiol Reg I 279:R2104–R2112
Guinet C, Jouventin P, Malacamp J (1995) Satellite remote sensing in monitoring change of seabirds: use of Spot Image in king penguin population increase at Ile aux Cochons. Crozet Archipelago Polar Biol 15:511–515
Halliwell B, Gutteridge J (2007) Free radicals in biology and medicine. Oxford University Press, Oxford
Hau M, Ricklefs RE, Wikelski M, Lee KA, Brawn JD (2010) Corticosterone, testosterone and life-history strategies of birds. Proc R Soc B 277:3203–3212
Haussmann MF, Longenecker AS, Marchetto NM, Juliano SA, Bowden RM (2012) Embryonic exposure to corticosterone modifies the juvenile stress response, oxidative stress and telomere length. Proc R Soc B 279:1447–1456
Hayward LS, Wingfield JC (2004) Maternal corticosterone is transferred to avian yolk and may alter offspring growth and adult phenotype. Gen Comp Endocr 135:365–371
Heidinger BJ, Nisbet ICT, Ketterson ED (2008) Changes in adrenal capacity contribute to a decline in the stress response with age in a long-lived seabird. Gen Comp Endocr 156:564–568
Jessop TS, Limpus CJ, Whittier JM (2002) Nocturnal activity in the green sea turtle alters daily profiles of melatonin and corticosterone. Horm Behav 41:357–365
Kitaysky AS, Wingfield JC, Piatt JF (1999) Dynamics of food availability, body condition and physiological response in breeding black-legged kittiwakes. Funct Ecol 13:577–585
Kitaysky AS, Kitaiskaia EV, Piatt JF, Wingfield JC (2003) Benefits and costs of increased levels of corticosterone in seabird chicks. Horm Behav 43:140–149
Koolhaas JM, Korte SM, De Boer SF, Van Der Vegt BJ, Van Reenen CG, Hopster H, De Jong IC, Ruis MAW, Blokhuis HJ (1999) Coping styles in animals: current status in behavior and stress physiology. Neurosci Biobehav R 23:925–935
Kotrschal A, Ilmonen P, Penn DJ (2007) Stress impacts telomere dynamics. Biol Lett 3:128–130
Landys MM, Ramenofsky M, Wingfield JC (2006) Actions of glucocorticoids at a seasonal baseline as compared to stress-related levels in the regulation of periodic life processes. Gen Comp Endocr 148:132–149
McCormick MI (2006) Mothers matter: Crowding leads to stressed mothers and smaller offspring in marine fish. Ecology 87:1104–1109
Ménard J–J (1998) Conséquences hormonales et métaboliques du stress de contention chez le manchot royal (Aptenodytes patagonicus). Veterinary thesis, Université Paul Sabatier, Toulouse
Miyashita T, Yamaguchi T, Motoyama K, Unno K, Nakano Y, Shimoi K (2006) Social stress increases biopyrrins, oxidative metabolites of bilirubin, in mouse urine. Biochem Biophys Res Comm 349:775–780
Nishio Y, Nakano Y, Deguchi Y, Terato H, Ide H, Ito C, Ishida H, Takagi K, Tsuboi H, Kinae N, Shimoi K (2007) Social stress induces oxidative DNA damage in mouse peripheral blood cells. Genes Environ 29:17–22
Raouf SA, Smith LC, Brown MB, Wingfield JC, Brown CR (2006) Glucocorticoid hormone levels increase with group size and parasite load in cliff swallows. Anim Behav 71:39–48
Romero LM (2002) Seasonal changes in plasma glucocorticoid concentrations in free-living vertebrates. Gen Comp Endocrinol 128:1–24
Romero LM, Reed JM (2005) Collecting baseline corticosterone samples in the field: is under 3 min good enough? Comp Biochem Physiol Part A 140:73–79
Romero LM, Reed JM, Wingfield JC (2000) Effects of weather on corticosterone responses in wild free-living passerine birds. Gen Comp Endocr 118:113–122
Romero LM, Dickens MJ, Cyr NE (2009) The reactive scope model—a new model integrating homeostasis, allostasis, and stress. Horm Behav 55:375–389
Sapolsky RM (2002) Endocrinology of the stress response. In: Becker JB, Breedlove SM, Crews D, McCarthy MM (eds) Behavioral endocrinology. MIT Press, Cambridge, pp 409–450
Selman C, Blount JD, Nussey DH, Speakman JR (2012) Oxidative damage, ageing, and life-history evolution: where now? Trends Ecol Evol 27:570–577
Shultz MT, Kitaysky AS (2008) Spatial and temporal dynamics of corticosterone and corticosterone binding globulin are driven by environmental heterogeneity. Gen Comp Endocr 155:717–728
Stier KS, Almasi B, Gasparini J, Piault R, Roulin A, Jenni L (2009) Effects of corticosterone on innate and humoral immune functions and oxidative stress in barn owl nestlings. J Exp Biol 212:2085–2091
Stier A, Reichert S, Massemin S, Bize P, Criscuolo F (2012) Constraint and cost of oxidative stress on reproduction: correlative evidence in laboratory mice and review of the literature. Front Zool 9:37
Stier A, Viblanc VA, Massemin-Challet S, Handrich Y, Zahn S, Rojas ER, Saraux C, Le Vaillant M, Prud’Homme O, Grosbellet E, Robin J-P, Bize P, Criscuolo F (2014) Starting with a handicap: phenotypic differences between early- and late-born king penguin chicks and their survival correlates. Funct Ecol. doi:10.1111/1365-2435.12204
Stonehouse B (1960) The king penguin Aptenodytes patagonica of South Georgia. Sci Rep Falk Isl Depend Surv 23:1–81
Valle S, Li W, Qin SJ (1999) Selection of the number of principal components: the variance of the reconstruction error criterion with a comparison to other methods. Ind Eng Chem Res 38:4389–4401
Viblanc VA, Bize P, Criscuolo F, Le Vaillant M, Saraux C, Pardonnet S, Gineste B, Kauffmann M, Prud’homme O, Handrich Y, Massemin S, Groscolas R, Robin J-P (2012) Body girth as an alternative to body mass for establishing condition indexes in field studies: a validation in the king penguin. Physiol Biochem Zool 85:533–542
Viera VM, Le Bohec C, Côté SD, Groscolas R (2006) Massive breeding failures following a tsunami in a colonial seabird. Polar Biol 29:713–716
Viera VM, Viblanc VA, Filippi-Codaccioni O, Côté SD, Groscolas R (2011) Active territory defence at a low energy cost in a colonial seabird. Anim Behav 82:69–76
von Zglinicki T (2002) Oxidative stress shortens telomeres. Trends Biochem Sci 27:339–344
Washburn BE, Morris DL, Millspaugh JJ, Faaborg J, Schulz JH (2002) Using a commercially available radioimmunoassay to quantify corticosterone in avian plasma. Condor 104:558–563
Weimerskirch H, Stahl JC, Jouventin P (1992) The breeding biology and population dynamics of king penguins Aptenodytes patagonica on the Crozet Islands. Ibis 134:107–117
Wingfield JC, Romero LM (2001) Adrenocortical responses to stress and their modulation in free-living vertebrates. In: McEwen B (ed) Coping with the environment: neural and endocrine mechanisms. Oxford University Press, New York, pp 211–236
Wingfield JC, Moore MC, Farner DS (1983) Endocrine responses to inclement weather in naturally breeding populations of white-crowned sparrows (Zonotrichia leucophrys pugetensis). Auk 100:56–62
Wingfield JC, Maney DL, Breuner CW, Jacobs JD, Lynn S, Ramenofsky M, Richardson RD (1998) Ecological bases of hormone-behavior interactions: the ‘emergency life history stage’. Am Zool 38:191–206
Zafir A, Banu N (2009) Modulation of in vivo oxidative status by exogenous corticosterone and restraint stress in rats. Stress 12:167–177
Acknowledgments
We are sincerely grateful to R. Boonstra and C. Saraux for enlightened comments on a previous draft of the paper. Three anonymous reviewers provided insightful comments on the manuscript. We wish to thank M. Kauffmann and C. Zimmer for their help during fieldwork, and P. Ohlmann for providing facilities for CORT measurements. This research was funded by the French Polar Institute (IPEV-Research Program 119) and the National Center for Scientific Research (CNRS-INEE). Logistic support in the field was provided by the Terres Australes et Antarctiques Françaises. V. A. V. was the recipient of a post-doctoral fellowship from the AXA foundation.
Author information
Authors and Affiliations
Corresponding author
Additional information
Communicated by Pawel Koteja.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Viblanc, V.A., Gineste, B., Stier, A. et al. Stress hormones in relation to breeding status and territory location in colonial king penguin: a role for social density?. Oecologia 175, 763–772 (2014). https://doi.org/10.1007/s00442-014-2942-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00442-014-2942-6