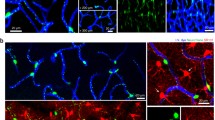
Abstract
This tribute article presents selected immunocytochemical and transmission electron microscope data on the location of ATP-gated P2X receptor in the rat brain, as studied in the 1990s in Prof G. Burnstock’s laboratory at University College London. There are examples of immuno-ultrastructural findings and introductory information about pre- and post-synaptic location of P2X receptors in the rat cerebellum and endocrine hypothalamus to support the concept of purinergic transmission in the central nervous system. Then findings of diverse immunoreactivity for P2X1, P2X2, P2X4, and P2X6 receptors associated with brain microvessels are shown, including vascular endothelium and pericytes as well as perivascular astrocytes and neuronal components. These findings imply the involvement of P2X receptors and hence purinergic signalling in the neurovascular unit, at least in microvessels in the rat cerebellum and hypothalamic paraventricular and supraoptic nuclei examined here. Various aspects of P2X receptors in brain microvessels are discussed.




Similar content being viewed by others
References
Araque A, Parpura V, Sanzgiri RP, Haydon PG (1999) Tripartite synapses: glia, the unacknowledged partner. Trends Neurosci 22:208–215
Armulik A, Abramsson A, Betsholtz C (2005) Endothelial/pericyte-interactions. Circ Res 97:512–523
Armulik A, Genove G, Mäe M, Nisancioglu MH, Wallgard E, Niaudet C, He L, Norlin J, Lindblom P, Strittmatter K, Johansson BR, Betsholtz C (2010) Pericytes regulate the blood–brain barrier. Nature 468:557–561
Bezzi P, Domercq M, Vesce S, Volterra A (1999) Neuron-astrocyte cross-talk during synaptic transmission: physiological and neuropathological implications. Progr Brain Res 132:255–265
Blanco VM, Stern JE, Filosa J (2008) Tone-dependent vascular responses to astrocyte-derived signals. Am J Physiol Heart Circ Physiol 294:H2855–H2863
Bobanovic LK, Royle SL, Murrell-Lagnado RD (2002) P2X receptor trafficking in neurons is subunit specific. J Neurosci 22:4814–4824
Bodin P, Burnstock G (1995) Synergistic effect of acute hypoxia on flow-induced release of ATP from cultured endothelial cells. Experientia 51:256–259
Bodin P, Burnstock G (2001) Evidence that release of adenosine triphosphate from endothelial cells during increased shear stress is vesicular. J Cardiovasc Pharmacol 38:900–908
Bodin P, Bailey DJ, Burnstock G (1991) Increased flow-induced ATP release from isolated vascular endothelial but not smooth muscle cells. Br J Pharmacol 103:1203–1205
Bredt DS, Hwang PM, Snyder SH (1990) Localization of nitric oxide synthase indicating a neural role for nitric oxide. Nature 347:768–770
Burnstock G (1990) Dual control of local blood flow by purines. Ann NY Acad Sci 603:31–44
Burnstock G (2007) Physiology and pathophysiology of purinergic neurotransmission. Physiol Rev 67:659–797. https://doi.org/10.1152/physrev.00043.2006
Burnstock G (2010) Control of vascular tone by purines and pyrimidines. Br J Pharmacol 161:527–529
Burnstock G (2017a) Purinergic signaling in the cardiovascular system. Circ Res 120:207–228
Burnstock G (2017b) Purinergic signalling and neurological diseases: an update. CNS Neurol Disord Drug Targets 16:257–265
Burnstock G (2017c) Purinergic signalling: therapeutic developments Front Pharmacol 8:661. https://doi.org/10.3389/fphar.2017.00661
Burnstock G (2020) Introduction to purinergic signalling in the brain. In: Barańska J (ed) Glioma signaling. Advances in experimental medicine and biology, vol 1202. Springer, Cham, pp 1–12 https://doi.org/10.1007/978-3-030-30651-9_1
Burnstock G, Knight G (2018) The potential of P2X7 receptors as a therapeutic target, including inflammation and tumour progression. Purinergic Signal 14:1–18
Burnstock G, Ralevic V (2014) Purinergic signaling and blood vessels in health and disease. Pharmacol Rev 66:102–192
Burnstock G, Krugel U, Abbracchio MP, Illes P (2011) Purinergic signalling: from normal behaviour to pathological brain function. Prog Neurobiol 95:229–274
Burnstock G, Verkhratsky A (2010) Vas deferens - a model used to establish sympathetic cotransmission. Trends Pharmacol Sci 31:131–139
Cai C, Fordsmann JC, Jensen SH, Gesslein B, Lønstrup M, Hald BO, Zambach SA, Brodin B, Lauritzen MJ (2018) Stimulation-induced increases in cerebral blood flow and local capillary vasoconstriction depend on conducted vascular responses. PNAS 115:E5796–E5804
Cantelmo AR, Conradi L-C, Brajic A, Goveia J, Kalucka J, Pircher A, Chaturvedi P, Hol J, Thienpont B, Teuwen L-A, Schoors S, Boeckx B, Vriens J, Kuchnio A, Veys K, Cruys B, Finotto L, Treps L, Stav-Noraas TE, Bifari F, Stapor P, Decimo I, Kampen K, De Bock K, Haraldsen G, Schoonjans L, Rabelink T, Eelen G, Ghesquière B, Rehman J, Lambrechts D, Malik AB, Dewerchin M, Carmeliet P (2016) Inhibition of the glycolytic activator PFKFB3 in endothelium induces tumor vessel normalization, impairs metastasis, and improves chemotherapy. Cancer Cell 30:968–985
Chan CM, Unwin RJ, Oglesby IB, Ford APDW, Townsend-Nicholson A, Burnstock G (1998) Localization of the P2X1purinoceptors by autoradiography and immunochistochemistry in rat kid-neys. Am J Physiol 274:F799–F804
Chen ZP, Levy A, Lightman SL (1994) Activation of specific ATP receptors induces a rapid increase in intracellular calcium ions in rat hypothalamic neurons. Brain Res 641:249–256
Cheng J, Korte N, Nortley R, Sethi H, Tang Y, Attwell D (2018) Targeting pericytes for therapeutic approaches to neurological disorders. Acta Neuropathol 136:507–523
Coco S, Calegari F, Pravettoni E, Pozzi D, Taverna E, Rosa P, Matteoli M, Verderio C (2003) Storage and release of ATP from astrocytes in culture. Biol Chem 278:1354–1362. https://doi.org/10.1074/jbc.m209454200
Collo G, North RA, Kawashima E, Merlo-Pich E, Neidhart S, Surprenant A, Buell G (1996) Cloning of P2X5 and P2X6 receptors and the distribution and properties of an extended family of ATP-gated ion channels. J Neurosci 16:2495–2507
Cunha RA, Ribeiro JA (2000) ATP as a presynaptic modulator. Life Sci 68:119–137
Dalkara T, Alakron-Martinez L (2015) Cerebral microvascular pericytes and neurogliovascular gnaling in health and disease. Brain Res 1623:3–17
Dietrich HH, Horiuchi T, Xiang C, Hongo K, Falck JR, Dacey RG Jr (2009) Mechanism of ATP-induced local and conducted vasomotor responses in isolated rat cerebral penetrating arterioles. J Vasc Res 46:253–264
Domer FR, Alexander B, Milner P, Bodin P, Burnstock G (1993) Effect of changes in rate of vascular perfusion on release of substances into the effluent from the brain of the rabbit. Brain Res 630:88–94
Dore-Duffy P, Cleary K (2011) Morphology and properties of pericytes. Methods Mol Biol 686:49–68
Fernandez-Klett F, Brandt L, Fernández-Zapata C, Abuelnor B, Middeldorp J, Sluijs JA, Curtis M, Faull R, Harris LW, Bahn S, Hol EM, Prille J (2020) Denser brain capillary network with preserved pericytes in Alzheimer’s disease. Brain Pathol 2020:1–1. https://doi.org/10.1111/bpa.12897
Glass R, Burnstock G (2001) Immunohistochemical identification of cells expressing ATP-gated cation channels (P2X receptors) in the adult rat thyroid. J Anat 198:569–579
Glass R, Townsend-Nicholson A, Burnstock G (2000) P2 receptors in the thymus: expression of P2X and P2Y receptors in adult rats, an immunohistochemical and in situ hybridisation study. Cell Tissue Res 300:295–306
Glass R, Loesch A, Bodin† P, Burnstock G, (2002) P2X4 and P2X6 receptors associate with VE-cadherin in human endothelial cells. CMLS, Cell Mol Life Sci 59:870–881
Guo W, Sun J, Xu X, Bunstock G, He C, Xiang Z (2009) P2X receptors are differentially expressed on vasopressin and oxytocin-containing neurons in the supraoptic and paraventricular nuclei of rat hypothalamus. Histochem Cell Biol 131:29–41
Hall CN, Reynell C, Gesslein B, Hamilton NB, Mishra A, Sutherland BA, O’Farrell FM, Buchan AM, Lauritzen M, Attwell D (2014) Capillary pericytes regulate cerebral blood flow in health and disease. Nature 508:55–60
Hansen MA, Dutton JL, Barden BVJ, JA, Bennett MR, (1999) P, (purinergic) receptor distributions in rat blood vessels. J Anat Nerv Syst 75:147–155. https://doi.org/10.1016/S0165-1838(98)00189-1
Hayden MR (2019) Hypothesis: astrocyte foot processes detachment from the neurovascular unit in female diabetic mice may impair modulation of information processing-six degrees of separation. Brain Sci 9:83. https://doi.org/10.3390/brainsci9040083
Iadecola C, Beitz AJ, Renno W, Xu X, Mayer B, Zhang F (1993) Nitric oxide synthase-containing neural processes on large cerebral arteries and cerebral microvessels. Brain Res 606:148–155
Jensen LJ, Holstein-Rathlou NH (2013) The vascular conducted response in cerebral blood flow regulation. J Cereb Blood Flow Metab 33:649–656
Kapoor JR, Sladek CD (2000) Purinergic and adrenergic agonists synergize in stimulating vasopressin and oxytocin release. J Neurosci 20:8868–8875
Kawamura H, Sugiyama T, Wu DM, Kobayashi M, Yamanishi S, Katsumura K, Puro DG (2003) ATP: a vasoactive signal in the pericyte-containing microvasculature of the rat retina. J Physiol 551(Pt 3):787–799
Kirabali T, Rust R, Rigorri S, Siccoli A, Nitsch RM, Kulic L (2020) Distinct changes in all major components of the neurovascular unit across different neuropathological stages of Alzheimer’s disease. Brain Pathol. https://doi.org/10.1111/bpa.12895
Kovac A, Erickson MA, Banks WA (2011) Brain microvascular pericytes are immunoactive in culture: cytokine, chemokine, nitric oxide, and LRP-1 expression in response to lipopolysaccharide. J Neuroinflammation 8:139. https://doi.org/10.1186/1742-2094-8-139
Lê KT, Villeneuve P, Ramjaun AR, Mcpherson PS, Beaudet A, Séguéla P (1998) Sensory presynaptic and widespread somatodendritic immunolocalization of central ionotropic P2X ATP receptors. Neuroscience 83:177–190
Lemos JR, Custer EE, Ortiz-Miranda S (2018) Purinergic receptor types in the hypothalamic-neurohypophysial system. J Neuroendorrinol 30:1–8
Llewenllyn-Smith IJ, Burnstock G (1998) Ultrastructural localization of P2X3 receptors in rat sensory neurons. NeuroReport 9:2545–2550
Loesch A (2002) Perivascular nerves and vascular endothelium: recent advances. Histol Histopathol 17:591–597
Loesch A, Burnstock G (1996) Ultrastructural study of perivascular nerve fibres and endothelial cells of the rat basilar artery immunolabelled with monoclonal antibodies to neuronal and endothelial nitric oxide synthase. J Neurocytol 25:525–534
Loesch A, Burnstock G (1998a) Electron-immunocytochemical localization of P2X1 receptors in the rat cerebellum. Cell Tissue Res 294:253–260
Loesch A, Burnstock G (1998b) Perivascular nerve fibres and endothelial cells of the rat basilar artery: immuno-gold labelling of antigenic sites for type I and type III nitric oxide synthase. J Neurocytol 27:197–204
Loesch A, Burnstock G (2000) Ultrastructural localisation of ATP-gated P2X2 receptor immunoreactivity in vascular endothelial cells in rat brain. Endothelium 7:93–98
Loesch A, Burnstock G (2001) Immunoreactivity to P2X6 receptors in the rat hypothalamo-neurohypophysial system: An ultrastructural study with extravidin and colloidal gold-silver labelling. Neuroscience 106:621–631
Loesch A, Miah S, Burnstock G (1999) Ultrastructural localisation of ATP-gated P2X2 receptor immunoreactivity in the rat hypothalamo-neurohypophysial system. J Neurocytol 28:495–504
Milner P, Bodin P, Loesch A, Burnstock G (1990) Rapid release of endothelin and ATP from isolated aortic endothelial cells exposed to increased flow. Biochem Biophys Res Commun 170:649–656
Milner P, Kirkpatrick KA, Ralevic V, Toothill V, Pearson JD, Burnstock G (1990) Endothelial cells cultured from human umbilical vein release ATP, substance P and acetylcholine in response to increased flow. Proc Roy Soc B 241:245–248
Milner P, Bodin P, Loesch A, Burnstock G (1992) Increased shear stress leads to differential release of endothelin and ATP from isolated endothelial cells from 4- and 12-month-old male rabbit aorta. J Vasc Res 29:420–425
Milner P, Bodin P, Loesch A, Burnstock G (1995) Interactions between sensory perivascular nerves and the endothelium in brain microvessels. Int J Microcirc 15:1–9
Mishra A, Reynolds JP, Chen Y, Gourine AV, Rusakov DA, Attwell D (2016) Astrocytes mediate neurovascular signaling to capillary pericytes but not to arteriols. Nat Neurosci 19:1619–1627
Mori M, Tsushima H, Matsuda T (1992) Antidiuretic effects of purinoceptor agonists injected into the hypothalamic paraventricular nucleus of water-loaded, ethanol-anesthetized rats. Neuropharmacol 31:585–592
Mulligan SJ, MacVicar BA (2004) Calcium transients in astrocyte endfeet cause cerebrovascular constrictions. Nature 431:195–199
Murphy S, Simmons ML, Agullo L, Garcia A, Feinstein DL, Galea E, Reis DJ, Minc-Golomb D, Schwartz JP (1993) Synthesis of nitric oxide in CNS glial cells. Trends Neurosci 16:323–328
Netto JP, Iliff J, Stanimirovic D, Krohn KA, Hamilton B, Varallyay C, Gahramanov S, Daldrup-Link H, d’Esterre C, Zlokovic B, Sair H, Lee Y, Taheri S, Jain R, Panigrahy A, Reich DS, Drewes LR, Castillo M, Neuwelt EA (2018) Neurovascular unit: basic and clinical imaging with emphasis on advantages of ferumoxytol. Neurosurgery 82:770–780
Nozaki K, Moskowitz MA, Maynard KI, Koketsu N, Dawson TM, Bredt DS, Snyder SH (1993) Possible origins and distribution of immunoreactive nitric oxide synthase-containing nerve fibers in cerebral arteries. J Cereb Blood Flow Metab 13:70–79
Nwadozi E, Rudnicki M, Haas TL (2020) Metabolic coordination of pericyte phenotypes: therapeutic implications. Front Cell Dev Biol 8:77. https://doi.org/10.3389/2Ffcell.2020.00077
Oglesby IB, Lachnit WG, Burnstock G, Ford APDW (1999) Subunit specificity of polyclonal antisera to the carboxy terminal regions of the P2X receptors, P2X1 through P2X7. Drug Dev Res 47:189–195
Ozerdem U, Grako KA, Danhil-Huppe K, Monsov E, Stallcup WB (2001) NG2 proteoglycan is expressed exclusively by mural cells during vascular morphogenesis. Dev Dyn 222:218–227
Peppiatt CM, Howarth C, Mobbs P, Attwell D (2006) Bidirectional control of CNS capillary diameter by pericytes. Nature 443:700–704
Poelchen W, Sieler D, Wirkner K, Illes P (2001) Co-transmitter function of ATP in central catecholaminergic neurons of the rat. Neuroscience 102:593–602
Porter JT, McCarthy KD (1996) Hippocampal astrocytes in situ respond to glutamate released from synaptic terminals. J Neurosci 16:5073–5081
Ralevic V, Burnstock G (1998) Receptors for purines and pyrimidines. Pharmacol Rev 50:413–492
Ralevic V, Milner P, Kirkpatrick KA, Burnstock G (1992) Flow-induced release of adenosine 5’-triphosphate from endothelial cells of the rat mesenteric arterial bed. Experientia 48:31–34
Renaud LP, Bourquet CW (1991) Neurophysiology and neuropharmacology of hypothalamic magnocellular neurons secreting vasopressin and oxytocin. Progr Neurobiol 36:131–169
Robinson LE, Murrell-Lagnado R (2013) The trafficking and targeting of P2X receptors. Front. Cell. Neurosci., 22 November 2013 https://doi.org/10.3389/fncel.2013.00233
Sperlágh B, Sershen H, Lajtha A, Vizi ES (1997) Co-release of endogenous ATP and [3H]noradrenaline from rat hypothalamic slices: origin and modulation by α2-adrenoceptors. Neuroscience 82:511–520
Sperlágh B, Mergl Z, Juranyi Z, Vizi ES, Makara GB (1999) Local regulation of vasopressin and oxytocin secretion by extracellular ATP in the isolated posterior lobe of the rat hypophysis. J Endocrinol 160:343–350
Vanhoutte PM (2000) Say NO to ET. J Auton Nerv Syst 81:271–277
Verderio C, Matteoli M (2001) ATP Mediates calcium signaling between astrocytes and microglial cells: modulation by IFN-gamma. J Immunol 166:6383–6391
Verkhratsky A, Zimmermann H, Abbracchio MP, Illes P, Di Virgilio F (2020) Invited Obituary. In memoriam Geoffrey Burnstock: Creator of purinergic signaling. FUNCTION, 2020, 1(1): zqaa006. https://doi.org/10.1093/function/zqaa006
Xiang Z, Bo ZX, Burnstock G (1998) Localization of ATP-gated P2X receptor immunoreactivity in rat sensory and sympathetic ganglia. Neurosci Lett 256:105–108
Zonta M, Angulo MC, Gobbo S, Rosengarten B, Hossmann KA, Pozzan T, Carmignoto G (2003) Neuron-to-astrocyte signaling is central to the dynamic control of brain microcirculation. Nature Neurosci 6:43–50
Acknowledgments
I would like to thank Ms Tania Raquel Assuncao Martins for her editorial assistance.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The author declares that there is no conflict of interest.
Ethical approval
The studies described were carried following ethical approval. All rats were treated in accordance with the UK Animals (Scientific Procedures) Act 1986.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Loesch, A. On P2X receptors in the brain: microvessels. Dedicated to the memory of the late Professor Geoffrey Burnstock (1929–2020). Cell Tissue Res 384, 577–588 (2021). https://doi.org/10.1007/s00441-021-03411-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00441-021-03411-0