Abstract
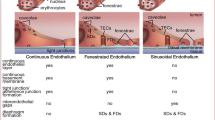
Endothelial fenestrae are the transcellular pores existing on the capillary walls which are organized in clusters referred to as sieve plates. They are also divided by a diaphragm consisting of plasmalemma vesicle-associated protein (PLVAP). In this study, we examined the involvement of fibronectin signaling in the formation of fenestra and diaphragm in endothelial cells. Results showed that Itga5 and Itgb1 were expressed in PECAM1-positive endothelial cells isolated from the anterior lobe (AL) of the rat pituitary, and integrin α5 was localized at the fenestrated capillaries of the rat pituitary and cultured PECAM1-positive endothelial cells isolated from AL (CECAL). Inhibition of both integrin α5β1 and FAK, a key molecule for integrin-microtubule signaling, respectively, by ATN-161 and FAK inhibitor 14, caused the delocalization of PLVAP at the sieve plates and depolymerization of microtubules in CECAL. Paclitaxel prevented the delocalization of PLVAP by the inhibition of integrin α5β1. Microtubule depolymerization induced by colcemid also caused the delocalization of PLVAP. Treatment of CECAL with ATN-161 and colcemid caused PLVAP localization at the Golgi apparatus. The localization of PLVAP at the sieve plates was inhibited by BFA treatment in a time-dependent manner and spread diffusely to the cytoplasm. These results indicate that a constant supply of PLVAP proteins by the endomembrane system via the Golgi apparatus is essential for the localization of PLVAP at sieve plates. In conclusion, the endomembrane transport pathway from the Golgi apparatus to sieve plates requires microtubule cytoskeletons, which are regulated by fibronectin-integrin α5β1 signaling.








Similar content being viewed by others
Abbreviations
- AL:
-
Anterior lobe
- BFA :
-
Brefeldin A
- BM :
-
Basement membrane
- BSA :
-
Bovine serum albumin
- CECAL :
-
Cultured endothelial cells isolated from the rat AL
- DAPI :
-
4′,6-Diamidino-2-phenylindole
- DMSO :
-
Dimethyl sulfoxide
- ECM :
-
Extracellular matrix
- ECMs :
-
Extracellular matrices
- FBS :
-
Fetal bovine serum
- GP :
-
Guinea pig
- IgG :
-
Immunoglobulin G
- PBS :
-
Phosphate-buffered saline
- PCR :
-
Polymerase chain reaction
- PECAM1 :
-
Platelet endothelial cell adhesion molecule
- PFA :
-
Paraformadehyde
- PLVAP :
-
Plasmalemma vesicle-associated protein
- RT :
-
Room temperature
- SEM :
-
Scanning electron microscopy
- VEGF-A :
-
Vascular endothelial growth factor-A
References
Akhmanova A, Steinmetz MO (2015) Control of microtubule organization and dynamics: two ends in the limelight. Nat Rev Mol Cell Biol 16:711–726
Alfaro-Aco R, Petry S (2015) Building the microtubule cytoskeleton piece by piece. J Biol Chem 290:17154–17162
Astrof S, Hynes RO (2009) Fibronectins in vascular morphogenesis. Angiogenesis 12:165–175
Braet F, Wisse E (2002) Structural and functional aspects of liver sinusoidal endothelial cell fenestrae: a review. Comp Hepatol 1:1–1
Colello D, Mathew S, Ward R, Pumiglia K, LaFlamme SE (2012) Integrins regulate microtubule nucleating activity of centrosome through mitogen-activated protein kinase/extracellular signal-regulated kinase kinase/extracellular signal-regulated kinase (MEK/ERK) signaling. J Biol Chem 287:2520–2530
Eremina V, Sood M, Haigh J, Nagy A, Lajoie G, Ferrara N, Gerber H-P, Kikkawa Y, Miner JH, Quaggin SE (2003) Glomerular-specific alterations of VEGF-A expression lead to distinct congenital and acquired renal diseases. J Clin Invest 111:707–716
Etienne-Manneville S, Hall A (2001) Integrin-mediated activation of Cdc42 controls cell polarity in migrating astrocytes through PKCζ. Cell 106:489–498
Farquhar MG (1961) Fine structure and function in capillaries of the anterior pituitary gland. Angiology 12:270–292
Francis SE, Goh KL, Hodivala-Dilke K, Bader BL, Stark M, Davidson D, Hynes RO (2002) Central roles of α5β1 integrin and fibronectin in vascular development in mouse embryos and embryoid bodies. Arterioscler Thromb Vasc Biol 22:927–933
Fujiwara T, Oda K, Yokota S, Takatsuki A, Ikehara Y (1988) Brefeldin A causes disassembly of the Golgi complex and accumulation of secretory proteins in the endoplasmic reticulum. J Biol Chem 263:18545–18552
George EL, Baldwin HS, Hynes RO (1997) Fibronectins are essential for heart and blood vessel morphogenesis but are dispensable for initial specification of precursor cells. Blood 90:3073–3081
George EL, Georges-Labouesse EN, Patel-King RS, Rayburn H, Hynes RO (1993) Defects in mesoderm, neural tube and vascular development in mouse embryos lacking fibronectin. Development 119:1079–1091
Gundersen GG, Gomes ER, Wen Y (2004) Cortical control of microtubule stability and polarization. Curr Opin Cell Biol 16:106–112
Herrnberger L, Seitz R, Kuespert S, Bösl MR, Fuchshofer R, Tamm ER (2012) Lack of endothelial diaphragms in fenestrae and caveolae of mutant Plvap-deficient mice. Histochem Cell Biol 138:709–724
Hirokawa N, Noda Y, Tanaka Y, Niwa S (2009) Kinesin superfamily motor proteins and intracellular transport. Nat Rev Mol Cell Biol 10:682–696
Horiguchi K, Nakakura T, Yoshida S, Tsukada T, Kanno N, Hasegawa R, Takigami S, Ohsako S, Kato T, Kato Y (2016) Identification of THY1 as a novel thyrotrope marker and THY1 antibody-mediated thyrotrope isolation in the rat anterior pituitary gland. Biochem Biophys Res Commun 480:273–279
Hynes RO (2002) Integrins: bidirectional, allosteric signaling machines. Cell 110:673–687
Ioannidou S, Deinhardt K, Miotla J, Bradley J, Cheung E, Samuelsson S, Ng Y-S, Shima DT (2006) An in vitro assay reveals a role for the diaphragm protein PV-1 in endothelial fenestra morphogenesis. Proc Natl Acad Sci U S A 103:16770–16775
Kapp TG, Rechenmacher F, Neubauer S, Maltsev OV, Cavalcanti-Adam EA, Zarka R, Reuning U, Notni J, Wester H-J, Mas-Moruno C, Spatz J, Geiger B, Kessler H (2017) A comprehensive evaluation of the activity and selectivity profile of ligands for RGD-binding integrins. Sci Rep 7:39805
Kim J, Han I, Kim Y, Kim S, Oh ES (2001) C-terminal heparin-binding domain of fibronectin regulates integrin-mediated cell spreading but not the activation of mitogen-activated protein kinase. Biochem J 360:239–245
LaFlamme SE, Mathew-Steiner S, Singh N, Colello-Borges D, Nieves B (2018) Integrin and microtubule crosstalk in the regulation of cellular processes. Cell Mol Life Sci 75:4177–4185
Lammert E, Gu G, McLaughlin M, Brown D, Brekken R, Murtaugh LC, Gerber HP, Ferrara N, Melton DA (2003) Role of VEGF-A in vascularization of pancreatic islets. Curr Biol 13:1070–1074
Larsson C (2006) Protein kinase C and the regulation of the actin cytoskeleton. Cell Signal 18:276–284
Lei L, Liu D, Huang Y, Jovin I, Shai S-Y, Kyriakides T, Ross RS, Giordano FJ (2008) Endothelial expression of β1 integrin is required for embryonic vascular patterning and postnatal vascular remodeling. Mol Cell Biol 28:794–802
Lombardi T, Montesano R, Furie MB, Silverstein SC, Orci L (1986) Endothelial diaphragmed fenestrae: in vitro modulation by phorbol myristate acetate. J Cell Biol 102:1965–1970
Marchand M, Monnot C, Muller L, Germain S (2019) Extracellular matrix scaffolding in angiogenesis and capillary homeostasis. Semin Cell Dev Biol 89:147–156
Murakami T, Kikuta A, Taguchi T, Ohtsuka A, Ohtani O (1987) Blood vascular architecture of the rat cerebral hypophysis and hypothalamus. A dissection/scanning electron microscopy of vascular casts. Arch Histol Jpn 50:133–176
Nakakura T, Suzuki T, Horiguchi K, Fujiwara K, Tsukada T, Asano-Hoshino A, Tanaka H, Arisawa K, Nishijima Y, Nekooki-Machida Y, Kiuchi Y, Hagiwara H (2017) Expression and localization of forkhead box protein FOXJ1 in S100β-positive multiciliated cells of the rat pituitary. Med Mol Morphol 50:59–67
Nakakura T, Suzuki T, Tanaka H, Arisawa K, Miyashita T, Nekooki-Machida Y, Kurosawa T, Tega Y, Deguchi Y, Hagiwara H (2020) Fibronectin is essential for formation of fenestrae in endothelial cells of the fenestrated capillary. Cell Tissue Res in press. https://doi.org/10.1007/s00441-00020-03273-y
Nakakura T, Suzuki T, Torii S, Asano-Hoshino A, Nekooki-Machida Y, Tanaka H, Arisawa K, Nishijima Y, Susa T, Okazaki T, Kiuchi Y, Hagiwara H (2017) ATAT1 is essential for regulation of homeostasis-retaining cellular responses in corticotrophs along hypothalamic-pituitary-adrenal axis. Cell Tissue Res 370:169–178
Nakakura T, Yoshida M, Dohra H, Suzuki M, Tanaka S (2006) Gene expression of vascular endothelial growth factor-A in the pituitary during formation of the vascular system in the hypothalamic-pituitary axis of the rat. Cell Tissue Res 324:87–95
Palazzo AF, Eng CH, Schlaepfer DD, Marcantonio EE, Gundersen GG (2004) Localized stabilization of microtubules by integrin- and FAK-facilitated Rho signaling. Science 303:836–839
Pankov R, Yamada KM (2002) Fibronectin at a glance. J Cell Sci 115:3861–3863
Peng X, Ueda H, Zhou H, Stokol T, Shen T-L, Alcaraz A, Nagy T, Vassalli J-D, Guan J-L (2004) Overexpression of focal adhesion kinase in vascular endothelial cells promotes angiogenesis in transgenic mice. Cardiovasc Res 64:421–430
Rhodin JA (1962) The diaphragm of capillary endothelial fenestrations. J Ultrastruct Res 6:171–185
Roberts WG, Palade GE (1995) Increased microvascular permeability and endothelial fenestration induced by vascular endothelial growth factor. J Cell Sci 108:2369–2379
Rosse C, Linch M, Kermorgant S, Cameron AJM, Boeckeler K, Parker PJ (2010) PKC and the control of localized signal dynamics. Nat Rev Mol Cell Biol 11:103–112
Shen T-L, Park AY-J, Alcaraz A, Peng X, Jang I, Koni P, Flavell RA, Gu H, Guan J-L (2005) Conditional knockout of focal adhesion kinase in endothelial cells reveals its role in angiogenesis and vascular development in late embryogenesis. J Cell Biol 169:941–952
Stan RV, Kubitza M, Palade GE (1999) PV-1 is a component of the fenestral and stomatal diaphragms in fenestrated endothelia. Proc Natl Acad Sci U S A 96:13203–13207
Stan RV, Tse D, Deharvengt SJ, Smits NC, Xu Y, Luciano MR, McGarry CL, Buitendijk M, Nemani KV, Elgueta R, Kobayashi T, Shipman SL, Moodie KL, Daghlian CP, Ernst PA, Lee HK, Suriawinata AA, Schned AR, Longnecker DS, Fiering SN, Noelle RJ, Gimi B, Shworak NW, Carriere C (2012) The diaphragms of fenestrated endothelia: gatekeepers of vascular permeability and blood composition. Dev Cell 23:1203–1218
Tanaka S, Nakakura T, Jansen EJR, Unno K, Okada R, Suzuki M, Martens GJM, Kikuyama S (2013) Angiogenesis in the intermediate lobe of the pituitary gland alters its structure and function. Gen Comp Endocrinol 185:10–18
Tanjore H, Zeisberg EM, Gerami-Naini B, Kalluri R (2008) β1 integrin expression on endothelial cells is required for angiogenesis but not for vasculogenesis. Dev Dyn 237:75–82
Xu J, Shi G-P (2014) Vascular wall extracellular matrix proteins and vascular diseases. Biochim Biophys Acta 1842:2106–2119
Zhao X, Guan J-L (2011) Focal adhesion kinase and its signaling pathways in cell migration and angiogenesis. Adv Drug Deliv Rev 63:610–615
Funding
This work was supported in part by JSPS KAKENHI (C) Grant Number JP19K07257, a research grant from Takeda Science Foundation, Hokuto Foundation for Bioscience, and Teikyo University.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflicts of interest.
Ethical approval
This study was approved by the Laboratory Animal Ethics Committee of Teikyo University (Tokyo, Japan) and conducted according to its guidelines. The document ID of the approval is 17-008. This article does not contain any studies with human participants.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Nakakura, T., Suzuki, T., Horiguchi, K. et al. Fibronectin-integrin signaling regulates PLVAP localization at endothelial fenestrae by microtubule stabilization. Cell Tissue Res 384, 449–463 (2021). https://doi.org/10.1007/s00441-020-03326-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00441-020-03326-2