Abstract
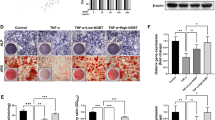
Our aim is to elucidate the effects of osteoproteogerin (OPG) on cartilage destruction in rats as a model of collagen-induced arthritis (CIA). To establish the CIA model, Sprague Dawley rats were injected with bovine type II collagen solution subcutaneously via the tails. Adenovirus-mediated OPG (Ad-OPG) was then injected intra-articularly either at the beginning of CIA (early OPG treatment) or one week after CIA establishment (late OPG treatment); vehicle or Ad-green fluorescent protein were injected as controls. The rats were killed 4 weeks after treatment. Ankle-joint sections were obtained for histology. Serum samples were collected for enzyme-linked immunosorbent assay. Safranin O staining showed that proteoglycan loss was inhibited in the early and late Ad-OPG groups. Terminal deoxynucleotidyl transferase-mediated dUTP nick-end labeling staining revealed that both early and late Ad-OPG treatments significantly prevented chondrocyte apoptosis in CIA rats. Furthermore, disintegrin and metalloproteinase with thrombospondin motif−5 expression decreased remarkably in the early and late OPG treatment groups. However, the cartilage destruction score, cartilage oligomeric matrix protein level and caspase-3 expression were only decreased in the early Ad-OPG treatment group. Additionally, ankle-joint swelling and the interleukin-1β expression level in CIA rats were not notably altered by Ad-OPG treatment. Taken together, our results suggest that early Ad-OPG treatment has potent protective effects against cartilage destruction during rheumatoid arthritis progression, mainly by reducing proteoglycan loss and chondrocyte apoptosis.






Similar content being viewed by others
References
Aigner T, Soeder S, Haag J (2006) IL-1beta and BMPs–interactive players of cartilage matrix degradation and regeneration. Eur Cell Mater 12:49–56
Aletaha D, Funovits J, Smolen JS (2011) Physical disability in rheumatoid arthritis is associated with cartilage damage rather than bone destruction. Ann Rheum Dis 70:733–739. doi:10.1136/ard.2010.138693corr1
Anderson JJ, Wells G, Verhoeven AC, Felson DT (2000) Factors predicting response to treatment in rheumatoid arthritis: the importance of disease duration. Arthritis Rheum 43:22–29
Audo R, Calmon-Hamaty F, Baeten D, Bruyer A, Combe B, Hahne M, Morel J (2011) Mechanisms and clinical relevance of TRAIL-triggered responses in the synovial fibroblasts of patients with rheumatoid arthritis. Arthritis Rheum 63:904–913. doi:10.1002/art.30181
Bellido M, Lugo L, Roman-Blas JA, Castaneda S, Caeiro JR, Dapia S, Calvo E, Largo R, Herrero-Beaumont G (2010) Subchondral bone microstructural damage by increased remodelling aggravates experimental osteoarthritis preceded by osteoporosis. Arthritis Res Ther 12:R152. doi:10.1186/ar3103
Bendele A (2001) Animal models of rheumatoid arthritis. J Musculoskelet Neuronal Interact 1:377–385
Bolon B, Carter C, Daris M, Morony S, Capparelli C, Hsieh A, Mao M, Kostenuik P, Dunstan CR, Lacey DL (2001) Adenoviral delivery of osteoprotegerin ameliorates bone resorption in a mouse ovariectomy model of osteoporosis&ast. Mol Ther 3:197–205
Bondeson J, Wainwright S, Hughes C, Caterson B (2008) The regulation of the ADAMTS4 and ADAMTS5 aggrecanases in osteoarthritis: a review. Clin Exp Rheumatol 26:139–145
Boyce BF, Xing L (2007) Biology of RANK, RANKL, and osteoprotegerin. Arthritis Res Ther 9 (Suppl 1):S1. doi:10.1186/ar2165
Bu R, McKeon R, Cao L, Blair H (2001) Osteoprotegerin (OPG) binds TNF family proteins including TNF alpha and protects osteoblasts from TNA alpha-induced apoptosis: a complementary function in addition to effects of OPG on osteoclast differentiation via RANKL. J Bone Miner Res S169-S169
Burr DB (2004) Anatomy and physiology of the mineralized tissues: role in the pathogenesis of osteoarthrosis. Osteoarthritis Cartilage 12 (Suppl A):S20-S30
Campo G, Avenoso A, Campo S, D’Ascola A, Traina P, Calatroni A (2008) Chondroitin-4-sulphate inhibits NF-kB translocation and caspase activation in collagen-induced arthritis in mice. Osteoarthritis Cartilage 16:1474–1483
Castaneda S, Roman-Blas JA, Largo R, Herrero-Beaumont G (2012) Subchondral bone as a key target for osteoarthritis treatment. Biochem Pharmacol 83:315–323. doi:10.1016/j.bcp.2011.09.018
Chappard C, Peyrin F, Bonnassie A, Lemineur G, Brunet-Imbault B, Lespessailles E, Benhamou CL (2006) Subchondral bone micro-architectural alterations in osteoarthritis: a synchrotron micro-computed tomography study. Osteoarthritis Cartilage 14:215–223. doi:10.1016/j.joca.2005.09.008
Dudler J, Renggli-Zulliger N, Busso N, Lotz M, So A (2000) Effect of interleukin 17 on proteoglycan degradation in murine knee joints. Ann Rheum Dis 59:529–532
Feng ZY, He ZN, Zhang B, Chen Z (2013) Osteoprotegerin promotes the proliferation of chondrocytes and affects the expression of ADAMTS-5 and TIMP-4 through MEK/ERK signaling. Mol Med Rep 8:1669–1679. doi:10.3892/mmr.2013.1717
Ferrari-Lacraz S, Ferrari S (2011) Do RANKL inhibitors (denosumab) affect inflammation and immunity? Osteoporosis Int 22:435–446. doi:10.1007/s00198-010-1326-y
Finckh A, Liang MH, van Herckenrode CM, de Pablo P (2006) Long-term impact of early treatment on radiographic progression in rheumatoid arthritis: a meta-analysis. Arthritis Care Res 55:864–872
Fuchs H, Kaye J, Callahan L, Nance E, Pincus T (1989) Evidence of significant radiographic damage in rheumatoid arthritis within the first 2 years of disease. J Rheumatol 16:585
Gilbert AM, Bursavich MG, Lombardi S, Georgiadis KE, Reifenberg E, Flannery CR, Morris EA (2008) N-((8-hydroxy-5-substituted-quinolin-7-yl)(phenyl)methyl)-2-phenyloxy/amino-aceta mide inhibitors of ADAMTS-5 (Aggrecanase-2). Bioorg Med Chem Lett 18:6454–6457. doi:10.1016/j.bmcl.2008.10.065
Glasson SS, Askew R, Sheppard B, Carito B, Blanchet T, Ma H-L, Flannery CR, Peluso D, Kanki K, Yang Z (2005) Deletion of active ADAMTS5 prevents cartilage degradation in a murine model of osteoarthritis. Nature 434:644–648
Gough AK, Lilley J, Eyre S, Holder RL, Emery P (1994) Generalised bone loss in patients with early rheumatoid arthritis. Lancet 344:23–27
Hayami T, Pickarski M, Wesolowski GA, McLane J, Bone A, Destefano J, Rodan GA, le Duong T (2004) The role of subchondral bone remodeling in osteoarthritis: reduction of cartilage degeneration and prevention of osteophyte formation by alendronate in the rat anterior cruciate ligament transection model. Arthritis Rheum 50:1193–1206. doi:10.1002/art.20124
Hilbers I, Hansen T, Petrow PK, Gaumann A, Bräuer R, Salzmann G, Gay RE, Kosmehl H, Kirkpatrick JC, Gay S (2003) Expression of the apoptosis accelerator Bax in rheumatoid arthritis synovium. Rheumatol Int 23:75–81
Holen I, Croucher PI, Hamdy FC, Eaton CL (2002) Osteoprotegerin (OPG) is a survival factor for human prostate cancer cells. Cancer Res 62:1619–1623
Jiao K, Niu LN, Wang MQ, Dai J, Yu SB, Liu XD, Wang J (2011) Subchondral bone loss following orthodontically induced cartilage degradation in the mandibular condyles of rats. Bone 48:362–371. doi:10.1016/j.bone.2010.09.010
Jin CH, Chae SY, Kim TH, Yang HK, Lee EY, Song YW, Jo DG, Lee KC (2010) Effect of tumor necrosis factor-related apoptosis-inducing ligand on the reduction of joint inflammation in experimental rheumatoid arthritis. J Pharmacol Experiment Therapeut 332:858–865. doi:10.1124/jpet.109.159517
Kadri A, Ea HK, Bazille C, Hannouche D, Liote F, Cohen-Solal ME (2008) Osteoprotegerin inhibits cartilage degradation through an effect on trabecular bone in murine experimental osteoarthritis. Arthritis Rheum 58:2379–2386. doi:10.1002/art.23638
Keystone E, Haraoui B, Bykerk V (2003) Role of adalimumab in the treatment of early rheumatoid arthritis. Clin Exp Rheumatol 21(5, Suppl 31):S198–S199
Kim HA, Song YW (1999) Apoptotic chondrocyte death in rheumatoid arthritis. Arthritis Rheum 42:1528–1537
Kirwan J (1997) The relationship between synovitis and erosions in rheumatoid arthritis. Rheumatology 36:225–228
Kong Y-Y, Feige U, Sarosi I, Bolon B, Tafuri A, Morony S, Capparelli C, Li J, Elliott R, McCabe S (1999) Activated T cells regulate bone loss and joint destruction in adjuvant arthritis through osteoprotegerin ligand. Nature 402:43–47
Lee SW, Lee HJ, Chung WT, Choi SM, Rhyu SH, Kim DK, Kim KT, Kim JY, Kim JM, Yoo YH (2004) TRAIL induces apoptosis of chondrocytes and influences the pathogenesis of experimentally induced rat osteoarthritis. Arthritis Rheum 50:534–542. doi:10.1002/art.20052
Martel-Pelletier J, Boileau C, Pelletier JP, Roughley PJ (2008) Cartilage in normal and osteoarthritis conditions. Best Pract Res Clin Rheumatol 22:351–384. doi:10.1016/j.berh.2008.02.001
Moreland LW, Baumgartner SW, Schiff MH, Tindall EA, Fleischmann RM, Weaver AL, Ettlinger RE, Cohen S, Koopman WJ, Mohler K, Widmer MB, Blosch CM (1997) Treatment of rheumatoid arthritis with a recombinant human tumor necrosis factor receptor (p75)-Fc fusion protein. New Engl J Med 337:141–147. doi:10.1056/Nejm199707173370301
Muraoka T, Hagino H, Okano T, Enokida M, Teshima R (2007) Role of subchondral bone in osteoarthritis development: a comparative study of two strains of guinea pigs with and without spontaneously occurring osteoarthritis. Arthritis Rheum 56:3366–3374. doi:10.1002/art.22921
Nagase H, Kashiwagi M (2003) Aggrecanases and cartilage matrix degradation. Arthritis Res Ther 5:94–103
Pettersen I, Figenschau Y, Olsen E, Bakkelund W, Smedsrod B, Sveinbjornsson B (2002) Tumor necrosis factor-related apoptosis-inducing ligand induces apoptosis in human articular chondrocytes in vitro. Biochem Biophys Res Commun 296:671–676
Pettit AR, Ji H, von Stechow D, Muller R, Goldring SR, Choi YW, Benoist C, Gravallese EM (2001) TRANCE/RANKL knockout mice are protected from bone erosion in a serum transfer model of arthritis. Am J Pathol 159:1689–1699. doi:10.1016/S0002-9440(10)63016-7
Plaas A, Osborn B, Yoshihara Y, Bai Y, Bloom T, Nelson F, Mikecz K, Sandy JD (2007) Aggrecanolysis in human osteoarthritis: confocal localization and biochemical characterization of ADAMTS5-hyaluronan complexes in articular cartilages. Osteoarthritis Cartilage 15:719–734. doi:10.1016/j.joca.2006.12.008
Quinn M, Conaghan P, Emery P (2001) The therapeutic approach of early intervention for rheumatoid arthritis: what is the evidence? Rheumatology 40:1211–1220
Rohner E, Detert J, Kolar P, Hocke A, N’Guessan P, Matziolis G, Kanitz V, Bernimoulin JP, Kielbassa A, Burmester GR, Buttgereit F, Pischon N (2010) Induced apoptosis of chondrocytes by Porphyromonas gingivalis as a possible pathway for cartilage loss in rheumatoid arthritis. Calcif Tissue Int 87:333–340. doi:10.1007/s00223-010-9389-5
Romas E, Sims NA, Hards DK, Lindsay M, Quinn JW, Ryan PF, Dunstan CR, Martin TJ, Gillespie MT (2002) Osteoprotegerin reduces osteoclast numbers and prevents bone erosion in collagen-induced arthritis. Am J Pathol 161:1419–1427
Saidenberg-Kermanac’h N, Corrado A, Lemeiter D, Devernejoul M, Boissier M, Cohen-Solal M (2004) TNF-alpha antibodies and osteoprotegerin decrease systemic bone loss associated with inflammation through distinct mechanisms in collagen-induced arthritis. Bone 35:1200
Schett G, Redlich K, Hayer S, Zwerina J, Bolon B, Dunstan C, Gortz B, Schulz A, Bergmeister H, Kollias G, Steiner G, Smolen JS (2003) Osteoprotegerin protects against generalized bone loss in tumor necrosis factor-transgenic mice. Arthritis Rheum 48:2042–2051. doi:10.1002/art.11150
Scott D, Smith C, Kingsley G (2003) Joint damage and disability in rheumatoid arthritis: an updated systematic review. Clin Exp Rheumatol 21(5, Suppl 31):S20–S27
Shimizu S, Asou Y, Itoh S, Chung UI, Kawaguchi H, Shinomiya K, Muneta T (2007) Prevention of cartilage destruction with intraarticular osteoclastogenesis inhibitory factor/osteoprotegerin in a murine model of osteoarthritis. Arthritis Rheum 56:3358–3365. doi:10.1002/art.22941
Smolen JS, Han C, van der Heijde DMFM, Emery P, Bathon JM, Keystone E, Maini RN, Kalden JR, Aletaha D, Baker D, Han J, Bala M, St Clair EW, Active-Controlled Study of Patients Receiving Infliximab for the Treatment of Rheumatoid Arthritis of Early Onset (ASPIRE) Study Group (2009) Radiographic changes in rheumatoid arthritis patients attaining different disease activity states with methotrexate monotherapy and infliximab plus methotrexate: the impacts of remission and tumour necrosis factor blockade. Ann Rheum Dis 68:823–827. doi:10.1136/ard.2008.090019
Stanton H, Rogerson FM, East CJ, Golub SB, Lawlor KE, Meeker CT, Little CB, Last K, Farmer PJ, Campbell IK, Fourie AM, Fosang AJ (2005) ADAMTS5 is the major aggrecanase in mouse cartilage in vivo and in vitro. Nature 434:648–652. doi:10.1038/nature03417
Stolina M, Adamu S, Ominsky M, Dwyer D, Asuncion F, Geng Z, Middleton S, Brown H, Pretorius J, Schett G, Bolon B, Feige U, Zack D, Kostenuik PJ (2005) RANKL is a marker and mediator of local and systemic bone loss in two rat models of inflammatory arthritis. J Bone Miner Res 20:1756–1765. doi:10.1359/JBMR.050601
Stolina M, Schett G, Dwyer D, Vonderfecht S, Middleton S, Duryea D, Pacheco E, Van G, Bolon B, Feige U, Zack D, Kostenuik P (2009) RANKL inhibition by osteoprotegerin prevents bone loss without affecting local or systemic inflammation parameters in two rat arthritis models: comparison with anti-TNF alpha or anti-IL-1 therapies. Arthritis Res Ther 11:R187. doi:10.1186/Ar2879
Weissmann G (2006) The pathogenesis of rheumatoid arthritis. Bull NYU Hosp Jt Dis 64:12–15
Welsing PM, Van Gestel AM, Swinkels HL, Kiemeney LA, Van Riel PL (2001) The relationship between disease activity, joint destruction, and functional capacity over the course of rheumatoid arthritis. Arthritis Rheumat 44:2009–2017
Wolfe F, Flowers N, Anderson J (2000) Radiographic progression predicts substantial income loss and work disability in rheumatoid arthritis. Arthritis Rheum 43(Suppl 9):S403
Xie C, Ma L, Liu J, Li X, Pei H, Xiang M, Chen L (2013) SKLB023 blocks joint inflammation and cartilage destruction in arthritis models via suppression of nuclear factor-kappa B activation in macrophage. PLoS One 8:e56349. doi:10.1371/journal.pone.0056349
Yatsugi N, Tsukazaki T, Osaki M, Koji T, Yamashita S, Shindo H (2000) Apoptosis of articular chondrocytes in rheumatoid arthritis and osteoarthritis: correlation of apoptosis with degree of cartilage destruction and expression of apoptosis-related proteins of p53 and c-myc. J Orthopaed Sci 5:150–156. doi:10.1007/s007760000050150.776
Zwerina J, Hayer S, Tohidast-Akrad M, Bergmeister H, Redlich K, Feige U, Dunstan C, Kollias G, Steiner G, Smolen J (2004) Single and combined inhibition of tumor necrosis factor, interleukin-1, and RANKL pathways in tumor necrosis factor–induced arthritis: effects on synovial inflammation, bone erosion, and cartilage destruction. Arthritis Rheum 50:277–290
Acknowledgments
This study was supported by the Natural Science Fund of the Ningbo Science and Technology Bureau (No. 201101A6110108).
Competing interest
The authors declare no competing interests.
Author information
Authors and Affiliations
Corresponding author
Additional information
Zhi-yun Feng and Zhen-nian He contributed equally to this work.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Fig. S1
Expression level of OPG was significantly higher in chondrocytes infected with adenovirus-OPG than in those infected with adenovirus-GFP (9.2-fold). Glyceraldehyde-3-phosphate dehydrogenate (GAPDH) was used as an internal control. (GIF 7 kb)
Fig. S2
Another 15 collagen-induced arthritis (CIA) rats were established. Each group of three was killed after 1, 7, 14, 21 and 28 days. Sections were stained with Fast Green-safranin O and TUNEL (terminal dUTP nick end-labeling) to show the state of proteoglycan loss and chondrocyte apoptosis in the evolution of CIA. As shown, compared with the normal control, a significant loss of proteoglycan can be observed at the onset of CIA (day 1, a) and becomes severe at day 7 (b). Cartilage destruction, such as the irregularity of articular surface and thinning of cartilage, was initially noted at day 14 (c). The integrity of the cartilage is compromised on day 21 and 28, with the formation of large fissures and focals (d,). At the same time, sparse safarain O staining on the cartilage occurs at day 14 and later (c–e). The number of TUNEL-positive chondrocytes at the beginning of arthritis (28.0%, f, k) was comparable with the normal control (24.1%), became more prevalent at day 7 (52.1%, g, l) and mounted by day 14 (h, m) and day 21 (i, n). Finally, it became overwhelming at day 28 (j, o). Quantitative data showed that the proteoglycan loss score was 1.6 (mild to severe) at the onset of arthritis and increased to 2.3 (severe) after 7 days (p). The cartilage destruction score was higher than 2 at day 14 (q), indicating structural changes of cartilage according to the modified criterion. Magnification: ×100 (a-j), ×400 (k-o, box in f-j). (GIF 269 kb)
Rights and permissions
About this article
Cite this article
Feng, Zy., He, Zn., Zhang, B. et al. Adenovirus-mediated osteoprotegerin ameliorates cartilage destruction by inhibiting proteoglycan loss and chondrocyte apoptosis in rats with collagen-induced arthritis. Cell Tissue Res 362, 187–199 (2015). https://doi.org/10.1007/s00441-015-2194-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00441-015-2194-8