Abstract
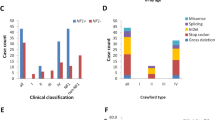
Up to 84% of patients with congenital pseudarthrosis of the tibia (CPT) present with neurofibromatosis type 1 (NF1) (NF1-CPT). However, the etiology of CPT not fulfilling the NIH diagnostic criteria for NF1 (non-NF1-CPT) is not well understood. Here, we collected the periosteum tissue from the pseudarthrosis (PA) site of 43 non-NF1-CPT patients and six patients with NF1-CPT, together with the blood or oral specimen of trios (probands and unaffected parents). Whole-exome plus copy number variation sequencing, multiplex ligation-dependent probe amplification (MLPA), ultra-high amplicon sequencing, and Sanger sequencing were employed to identify pathogenic variants. The result showed that nine tissues of 43 non-NF1-CPT patients (21%) had somatic mono-allelic NF1 inactivation, and five of six NF1-CPT patients (83.3%) had bi-allelic NF1 inactivation in tissues. However, previous literature involving genetic testing did not reveal somatic mosaicism in non-NF1-CPT patients so far. In NF1-CPT patients, when the results from earlier reports and the present study were combined, 66.7% of them showed somatic NF1 inactivation in PA tissues other than germline inactivation. Furthermore, no diagnostic variants from other known genes (GNAS, AKT1, PDGFRB, and NOTCH3) related to skeletal dysplasia were identified in the nine NF1 positive non-NF1-CPT patients and six NF1-CPT patients. In conclusion, we detected evident somatic mono-allelic NF1 inactivation in the non-NF1-CPT. Thus, for pediatric patients without NF1 diagnosis, somatic mutations in NF1 are important.

Similar content being viewed by others
Availability of data and materials
The novel variants found in this study were submitted to ClinVar (accession: SCV002014754-SCV002014759). Other datasets used during the current study are available from the corresponding authors on reasonable request.
Abbreviations
- CPT:
-
Congenital pseudarthrosis of the tibia
- NF1:
-
Neurofibromatosis type 1
- NF1-CPT:
-
CPT fulfilling the NIH diagnostic criteria for NF1
- Non-NF1-CPT:
-
CPT not fulfilling the NIH diagnostic criteria for NF1
- MNF1:
-
Mosaic NF1
- MLPA:
-
Multiplex ligation-dependent probe amplification
- PA:
-
Pseudarthrosis
- WES:
-
Whole-exome sequencing
- CNV:
-
Copy number variation
- CAL:
-
Café au lait macules
- PBS:
-
Phosphate-buffered saline
- SNV:
-
Single-nucleotide variations
References
Andersen LB, Ballester R, Marchuk DA, Chang E, Gutmann DH, Saulino AM, Collins FS (1993) A conserved alternative splice in the von Recklinghausen neurofibromatosis (NF1) gene produces two neurofibromin isoforms, both of which have GTPase-activating protein activity. Mol Cell Biol 13(1):487–495
Ballester R, Marchuk D, Boguski M, Saulino A, Letcher R, Wigler M, Collins F (1990) The NF1 locus encodes a protein functionally related to mammalian GAP and yeast IRA proteins. Cell 63(4):851–859
Basu TN, Gutmann DH, Fletcher JA, Glover TW, Collins FS, Downward J (1992) Aberrant regulation of ras proteins in malignant tumour cells from type 1 neurofibromatosis patients. Nature 356(6371):713–715. https://doi.org/10.1038/356713a0
Bollag G, Clapp DW, Shih S, Adler F, Zhang YY, Thompson P, Shannon K (1996) Loss of NF1 results in activation of the Ras signaling pathway and leads to aberrant growth in haematopoietic cells. Nat Genet 12(2):144–148. https://doi.org/10.1038/ng0296-144
Brekelmans C, Hollants S, De Groote C, Sohier N, Marechal M, Geris L, Brems H (2019) Neurofibromatosis type 1-related pseudarthrosis: Beyond the pseudarthrosis site. Hum Mutat 40(10):1760–1767. https://doi.org/10.1002/humu.23783
Cichowski K, Jacks T (2001) NF1 tumor suppressor gene function: narrowing the GAP. Cell 104(4):593–604. https://doi.org/10.1016/s0092-8674(01)00245-8
DeClue JE, Cohen BD, Lowy DR (1991) Identification and characterization of the neurofibromatosis type 1 protein product. Proc Natl Acad Sci USA 88(22):9914–9918
DePristo MA, Banks E, Poplin R, Garimella KV, Maguire JR, Hartl C, Daly MJ (2011) A framework for variation discovery and genotyping using next-generation DNA sequencing data. Nat Genet 43(5):491–498. https://doi.org/10.1038/ng.806
El-Rosasy MA (2020) Congenital pseudarthrosis of the tibia: the outcome of a pathology-oriented classification system and treatment protocol. J Pediatr Orthop B 29(4):337–347. https://doi.org/10.1097/BPB.0000000000000660
Evans DG, Howard E, Giblin C, Clancy T, Spencer H, Huson SM, Lalloo F (2010) Birth incidence and prevalence of tumor-prone syndromes: estimates from a UK family genetic register service. Am J Med Genet A 152A(2):327–332. https://doi.org/10.1002/ajmg.a.33139
Evans DG, Bowers N, Burkitt-Wright E, Miles E, Garg S, Scott-Kitching V, Huson SM (2016) Comprehensive RNA analysis of the NF1 gene in classically affected NF1 affected individuals meeting NIH criteria has high sensitivity and mutation negative testing is reassuring in isolated cases with pigmentary features only. EBioMedicine 7:212–220. https://doi.org/10.1016/j.ebiom.2016.04.005
Faust GG, Hall IM (2014) SAMBLASTER: fast duplicate marking and structural variant read extraction. Bioinformatics 30(17):2503–2505. https://doi.org/10.1093/bioinformatics/btu314
Friedman JM, Birch PH (1997) Type 1 neurofibromatosis: a descriptive analysis of the disorder in 1728 patients. Am J Med Genet 70:138–143
Garcia-Romero MT, Parkin P, Lara-Corrales I (2016) Mosaic neurofibromatosis type 1: a systematic review. Pediatr Dermatol 33(1):9–17. https://doi.org/10.1111/pde.12673
Granchi D, Devescovi V, Baglio SR, Leonardi E, Donzelli O, Magnani M, Baldini N (2010) Biological basis for the use of autologous bone marrow stromal cells in the treatment of congenital pseudarthrosis of the tibia. Bone 46(3):780–788. https://doi.org/10.1016/j.bone.2009.10.044
Gutmann DH, Ferner RE, Listernick RH, Korf BR, Wolters PL, Johnson KJ (2017) Neurofibromatosis type 1. Nat Rev Dis Prim 3:17004. https://doi.org/10.1038/nrdp.2017.4
Hagel C, Zils U, Peiper M, Kluwe L, Gotthard S, Friedrich RE, Mautner VF (2007) Histopathology and clinical outcome of NF1-associated vs. sporadic malignant peripheral nerve sheath tumors. J Neurooncol 82(2):187–192. https://doi.org/10.1007/s11060-006-9266-2
Hefti F, Bollini G, Dungl P, Fixsen J, Grill F, Ippolito E, Wientroub S (2000) Congenital pseudarthrosis of the tibia: history, etiology, classification, and epidemiologic data. J Pediatr Orthop B 9(1):11–15
Hermanns-Sachweh B, Senderek J, Alfer J, Klosterhalfen B, Buttner R, Fuzesi L, Weber M (2005) Vascular changes in the periosteum of congenital pseudarthrosis of the tibia. Pathol Res Pract 201(4):305–312. https://doi.org/10.1016/j.prp.2004.09.013
Hutmacher DW, Sittinger M (2003) Periosteal cells in bone tissue engineering. Tissue Eng 9(Suppl 1):S45-64. https://doi.org/10.1089/10763270360696978
Ippolito E, Corsi A, Grill F, Wientroub S, Bianco P (2000) Pathology of bone lesions associated with congenital pseudarthrosis of the leg. J Pediatr Orthop B 9(1):3–10
Karczewski KJ, Weisburd B, Thomas B, Solomonson M, Ruderfer DM, Kavanagh D, MacArthur DG (2017) The ExAC browser: displaying reference data information from over 60 000 exomes. Nucleic Acids Res 45(D1):D840–D845. https://doi.org/10.1093/nar/gkw971
Karczewski KJ, Francioli LC, Tiao G, Cummings BB, Alfoldi J, Wang Q, MacArthur DG (2020) The mutational constraint spectrum quantified from variation in 141,456 humans. Nature 581(7809):434–443. https://doi.org/10.1038/s41586-020-2308-7
Kehrer-Sawatzki H, Mautner VF, Cooper DN (2017) Emerging genotype-phenotype relationships in patients with large NF1 deletions. Hum Genet 136(4):349–376. https://doi.org/10.1007/s00439-017-1766-y
Koliou X, Fedonidis C, Kalpachidou T, Mangoura D (2016) Nuclear import mechanism of neurofibromin for localization on the spindle and function in chromosome congression. J Neurochem 136(1):78–91. https://doi.org/10.1111/jnc.13401
Kopanos C, Tsiolkas V, Kouris A, Chapple CE, Albarca Aguilera M, Meyer R, Massouras A (2019) VarSome: the human genomic variant search engine. Bioinformatics 35(11):1978–1980. https://doi.org/10.1093/bioinformatics/bty897
Lammert M, Friedman JM, Kluwe L, Mautner VF (2005) Prevalence of neurofibromatosis 1 in German children at elementary school enrollment. Arch Dermatol 141(1):71–74. https://doi.org/10.1001/archderm.141.1.71
Lau N, Feldkamp MM, Roncari L, Loehr AH, Shannon P, Gutmann DH, Guha A (2000) Loss of neurofibromin is associated with activation of RAS/MAPK and PI3-K/AKT signaling in a neurofibromatosis 1 astrocytoma. J Neuropathol Exp Neurol 59(9):759–767. https://doi.org/10.1093/jnen/59.9.759
Layer RM, Chiang C, Quinlan AR, Hall IM (2014) LUMPY: a probabilistic framework for structural variant discovery. Genome Biol 15(6):R84. https://doi.org/10.1186/gb-2014-15-6-r84
Lee SM, Choi IH, Lee DY, Lee HR, Park MS, Yoo WJ, Cho TJ (2012) Is double inactivation of the Nf1 gene responsible for the development of congenital pseudarthrosis of the tibia associated with NF1? J Orthop Res 30(10):1535–1540. https://doi.org/10.1002/jor.22121
Legius E, Messiaen L, Wolkenstein P, Pancza P, Avery RA, Berman Y, Plotkin SR (2021) Revised diagnostic criteria for neurofibromatosis type 1 and Legius syndrome: an international consensus recommendation. Genet Med 23(8):1506–1513. https://doi.org/10.1038/s41436-021-01170-5
Leskela HV, Kuorilehto T, Risteli J, Koivunen J, Nissinen M, Peltonen S, Peltonen J (2009) Congenital pseudarthrosis of neurofibromatosis type 1: impaired osteoblast differentiation and function and altered NF1 gene expression. Bone 44(2):243–250. https://doi.org/10.1016/j.bone.2008.10.050
Li H, Durbin R (2010) Fast and accurate long-read alignment with Burrows-Wheeler transform. Bioinformatics 26(5):589–595. https://doi.org/10.1093/bioinformatics/btp698
Li Q, Wang K (2017) InterVar: clinical interpretation of genetic variants by the 2015 ACMG-AMP Guidelines. Am J Hum Genet 100(2):267–280. https://doi.org/10.1016/j.ajhg.2017.01.004
Li H, Handsaker B, Wysoker A, Fennell T, Ruan J, Homer N, Genome Project Data Processing, S (2009) The sequence alignment/map format and SAM tools. Bioinformatics 25(16):2078–2079. https://doi.org/10.1093/bioinformatics/btp352
Listernick R, Mancini AJ, Charrow J (2003) Segmental neurofibromatosis in childhood. Am J Med Genet A 121A(2):132–135. https://doi.org/10.1002/ajmg.a.20183
Liu J, Zheng Y, Huang J, Zhu D, Zang P, Luo Z, Lu X (2021) Expanding the genotypes and phenotypes for 19 rare diseases by exome sequencing performed in pediatric intensive care unit. Hum Mutat 42(11):1443–1460. https://doi.org/10.1002/humu.24266
Luo G, Kim J, Song K (2014) The C-terminal domains of human neurofibromin and its budding yeast homologs Ira1 and Ira2 regulate the metaphase to anaphase transition. Cell Cycle 13(17):2780–2789. https://doi.org/10.4161/15384101.2015.945870
Maes C, Kobayashi T, Selig MK, Torrekens S, Roth SI, Mackem S, Kronenberg HM (2010) Osteoblast precursors, but not mature osteoblasts, move into developing and fractured bones along with invading blood vessels. Dev Cell 19(2):329–344. https://doi.org/10.1016/j.devcel.2010.07.010
Margraf RL, VanSant-Webb C, Sant D, Carey J, Hanson H, D’Astous J, Mao R (2017) Utilization of whole-exome next-generation sequencing variant read frequency for detection of lesion-specific, somatic loss of heterozygosity in a neurofibromatosis type 1 cohort with tibial pseudarthrosis. J Mol Diagn 19(3):468–474. https://doi.org/10.1016/j.jmoldx.2017.01.008
McCaughan JA, Holloway SM, Davidson R, Lam WW (2007) Further evidence of the increased risk for malignant peripheral nerve sheath tumour from a Scottish cohort of patients with neurofibromatosis type 1. J Med Genet 44(7):463–466. https://doi.org/10.1136/jmg.2006.048140
Messiaen LM, Callens T, Mortier G, Beysen D, Vandenbroucke I, Van Roy N, Paepe AD (2000) Exhaustive mutation analysis of the NF1 gene allows identification of 95% of mutations and reveals a high frequency of unusual splicing defects. Hum Mutat 15(6):541–555. https://doi.org/10.1002/1098-1004(200006)15:6%3c541::AID-HUMU6%3e3.0.CO;2-N
Neurofibromatosis (1988) Neurofibromatosis. Conference statement. National Institutes of Health Consensus Development Conference. Arch Neurol 45(5):575–578
O’Donnell C, Foster J, Mooney R, Beebe C, Donaldson N, Heare T (2017) Congenital Pseudarthrosis of the Tibia. JBJS Rev 5(4):e3. https://doi.org/10.2106/JBJS.RVW.16.00068
Ozaki A, Tsunoda M, Kinoshita S, Saura R (2000) Role of fracture hematoma and periosteum during fracture healing in rats: interaction of fracture hematoma and the periosteum in the initial step of the healing process. J Orthop Sci 5(1):64–70. https://doi.org/10.1007/s007760050010
Paria N, Cho TJ, Choi IH, Kamiya N, Kayembe K, Mao R, Rios JJ (2014) Neurofibromin deficiency-associated transcriptional dysregulation suggests a novel therapy for tibial pseudoarthrosis in NF1. J Bone Miner Res 29(12):2636–2642. https://doi.org/10.1002/jbmr.2298
Parrozzani R, Clementi M, Frizziero L, Miglionico G, Perrini P, Cavarzeran F, Midena E (2015) In vivo detection of choroidal abnormalities related to NF1: feasibility and comparison with standard NIH diagnostic criteria in pediatric patients. Invest Ophthalmol vis Sci 56(10):6036–6042. https://doi.org/10.1167/iovs.14-16053
Richards S, Aziz N, Bale S, Bick D, Das S, Gastier-Foster J, Committee ALQA (2015) Standards and guidelines for the interpretation of sequence variants: a joint consensus recommendation of the American College of Medical Genetics and Genomics and the Association for Molecular Pathology. Genet Med 17(5):405–424. https://doi.org/10.1038/gim.2015.30
Riggs ER, Andersen EF, Cherry AM, Kantarci S, Kearney H, Patel A, Martin CL (2020) Technical standards for the interpretation and reporting of constitutional copy-number variants: a joint consensus recommendation of the American College of Medical Genetics and Genomics (ACMG) and the Clinical Genome Resource (ClinGen). Genet Med 22(2):245–257. https://doi.org/10.1038/s41436-019-0686-8
Roberts SJ, van Gastel N, Carmeliet G, Luyten FP (2015) Uncovering the periosteum for skeletal regeneration: the stem cell that lies beneath. Bone 70:10–18. https://doi.org/10.1016/j.bone.2014.08.007
Sabbagh A, Pasmant E, Imbard A, Luscan A, Soares M, Blanche H, Wolkenstein P (2013) NF1 molecular characterization and neurofibromatosis type I genotype-phenotype correlation: the French experience. Hum Mutat 34(11):1510–1518. https://doi.org/10.1002/humu.22392
Sakamoto A, Yoshida T, Yamamoto H, Oda Y, Tsuneyoshi M, Iwamoto Y (2007) Congenital pseudarthrosis of the tibia: analysis of the histology and the NF1 gene. J Orthop Sci 12(4):361–365. https://doi.org/10.1007/s00776-007-1142-1
Sant DW, Margraf RL, Stevenson DA, Grossmann AH, Viskochil DH, Hanson H, Mao R (2015) Evaluation of somatic mutations in tibial pseudarthrosis samples in neurofibromatosis type 1. J Med Genet 52(4):256–261. https://doi.org/10.1136/jmedgenet-2014-102815
Schindeler A, Little DG (2008) Recent insights into bone development, homeostasis, and repair in type 1 neurofibromatosis (NF1). Bone 42(4):616–622. https://doi.org/10.1016/j.bone.2007.11.006
Seminog OO, Goldacre MJ (2013) Risk of benign tumours of nervous system, and of malignant neoplasms, in people with neurofibromatosis: population-based record-linkage study. Br J Cancer 108(1):193–198. https://doi.org/10.1038/bjc.2012.535
Stevenson DA, Zhou H, Ashrafi S, Messiaen LM, Carey JC, D’Astous JL, Viskochil DH (2006) Double inactivation of NF1 in tibial pseudarthrosis. Am J Hum Genet 79(1):143–148. https://doi.org/10.1086/504441
Tadini G, Milani D, Menni F, Pezzani L, Sabatini C, Esposito S (2014) Is it time to change the neurofibromatosis 1 diagnostic criteria? Eur J Intern Med 25(6):506–510. https://doi.org/10.1016/j.ejim.2014.04.004
Tahaei SE, Couasnay G, Ma Y, Paria N, Gu J, Lemoine BF, Elefteriou F (2018) The reduced osteogenic potential of Nf1-deficient osteoprogenitors is EGFR-independent. Bone 106:103–111. https://doi.org/10.1016/j.bone.2017.10.012
Talevich E, Shain AH, Botton T, Bastian BC (2016) CNVkit: genome-wide copy number detection and visualization from targeted DNA sequencing. PLoS Comput Biol 12(4):e1004873. https://doi.org/10.1371/journal.pcbi.1004873
Thorvaldsdottir H, Robinson JT, Mesirov JP (2013) Integrative Genomics Viewer (IGV): high-performance genomics data visualization and exploration. Brief Bioinform 14(2):178–192. https://doi.org/10.1093/bib/bbs017
Upadhyaya M, Han S, Consoli C, Majounie E, Horan M, Thomas NS, Cooper DN (2004) Characterization of the somatic mutational spectrum of the neurofibromatosis type 1 (NF1) gene in neurofibromatosis patients with benign and malignant tumors. Hum Mutat 23(2):134–146. https://doi.org/10.1002/humu.10305
Uusitalo E, Leppavirta J, Koffert A, Suominen S, Vahtera J, Vahlberg T, Peltonen S (2015) Incidence and mortality of neurofibromatosis: a total population study in Finland. J Invest Dermatol 135(3):904–906. https://doi.org/10.1038/jid.2014.465
Vagge A, Nelson LB, Capris P, Traverso CE (2016) Choroidal freckling in pediatric patients affected by neurofibromatosis type 1. J Pediatr Ophthalmol Strabismus 53(5):271–274. https://doi.org/10.3928/01913913-20160719-05
Valentin T, Le Cesne A, Ray-Coquard I, Italiano A, Decanter G, Bompas E, Chevreau C (2016) Management and prognosis of malignant peripheral nerve sheath tumors: the experience of the French Sarcoma Group (GSF-GETO). Eur J Cancer 56:77–84. https://doi.org/10.1016/j.ejca.2015.12.015 (1879-0852 (Electronic))
Van Royen K, Brems H, Legius E, Lammens J, Laumen A (2016) Prevalence of neurofibromatosis type 1 in congenital pseudarthrosis of the tibia. Eur J Pediatr 175(9):1193–1198. https://doi.org/10.1007/s00431-016-2757-z
Vander Have KL, Hensinger RN, Caird M, Johnston C, Farley FA (2008) Congenital pseudarthrosis of the tibia. J Am Acad Orthop Surg 16(4):228–236
Varan A, Sen H, Aydin B, Yalcin B, Kutluk T, Akyuz C (2016) Neurofibromatosis type 1 and malignancy in childhood. Clin Genet 89(3):341–345. https://doi.org/10.1111/cge.12625
Wang K, Li M, Hakonarson H (2010) ANNOVAR: functional annotation of genetic variants from high-throughput sequencing data. Nucleic Acids Res 38(16):e164. https://doi.org/10.1093/nar/gkq603
Xu GF, Lin B, Tanaka K, Dunn D, Wood D, Gesteland R, Tamanoi F (1990a) The catalytic domain of the neurofibromatosis type 1 gene product stimulates ras GTPase and complements ira mutants of S. cerevisiae. Cell 63(4):835–841. https://doi.org/10.1016/0092-8674(90)90149-9
Xu GF, O’Connell P, Viskochil D, Cawthon R, Robertson M, Culver M et al (1990b) The neurofibromatosis type 1 gene encodes a protein related to GAP. Cell 62(3):599–608. https://doi.org/10.1016/0092-8674(90)90024-9
Yang Y, Zheng Y, Li W, Li L, Tu M, Zhao L, Zhu Y (2019) SMAD6 is frequently mutated in nonsyndromic radioulnar synostosis. Genet Med 21(11):2577–2585. https://doi.org/10.1038/s41436-019-0552-8
Young H, Hyman S, North K (2002) Neurofibromatosis 1: clinical review and exceptions to the rules. J Child Neurol 17:613–621
Zhang J, Tong H, Fu X, Zhang Y, Liu J, Cheng R, Yao Z (2015) Molecular characterization of NF1 and neurofibromatosis type 1 genotype-phenotype correlations in a Chinese population. Sci Rep 5:2045–2322. https://doi.org/10.1038/srep11291
Zhu G, Zheng Y, Liu Y, Yan A, Hu Z, Yang Y, Mei H (2019) Identification and characterization of NF1 and non-NF1 congenital pseudarthrosis of the tibia based on germline NF1 variants: genetic and clinical analysis of 75 patients. Orphanet J Rare Dis 14(1):221. https://doi.org/10.1186/s13023-019-1196-0
Acknowledgements
We thank all the participated patients and their families for supporting our research. We also would like to express our sincere gratitude to all those who helped us in this study.
Funding
This work was supported by the Hunan Province Natural Science Foundation of China (Grant number: 2020JJ8005), the Health Commission of Hunan Province of China (Grant number: B2019020), Key Research and Development Program of Hunan Province (Grant number: 2020SK2113), and Clinical Research Center for Limb Deformity of Children in Hunan Province (Grant number: 2019SK4006).
Author information
Authors and Affiliations
Contributions
Conceptualization and investigation: ZH and HM, Funding acquisition, investigation, supervision, resources acquisition, methodology, and validation: HM, ZH, and YZ. Original draft writing and data analysis: YZ. Resources collection, idea refining, and data validation: GZ, YL, WZ, YY, ZL, and YF. Manuscript review and editing: ZH and HM. All authors read and approved the final manuscript.
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare that they have no competing interests.
Ethical approval and consent to participate
This study was approved by the Ethics Committee of Hunan Children's Hospital (Approval No. HCHLL-2020-106). The samples were obtained appropriate informed consent from all participants.
Consent for publication
All authors read and approved the final manuscript for publication.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Zheng, Y., Zhu, G., Liu, Y. et al. Case series of congenital pseudarthrosis of the tibia unfulfilling neurofibromatosis type 1 diagnosis: 21% with somatic NF1 haploinsufficiency in the periosteum. Hum Genet 141, 1371–1383 (2022). https://doi.org/10.1007/s00439-021-02429-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00439-021-02429-2