Abstract
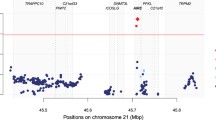
Primary Sjögren’s syndrome (PSS) is an autoimmune disease targeting exocrine glands. It ten times more dominantly affects women than men with an onset peak at menopause. The genetic factor predisposing women to PSS remains unclear. Therefore, we aimed to identify susceptibility loci for PSS in women. We performed genome-wide association study (GWAS) using 242 female PSS patients and 1444 female control in Han Chinese population residing in Taiwan. Replication was conducted in an independent cohort of 178 female PSS and 14,432 control subjects. We identified rs117026326 on GTF2I with GWAS significance (P = 1.10 × 10−15) and rs13079920 on RBMS3 with suggestive significance (P = 2.90 × 10−5) associating with PSS in women. The association of RBMS3 was further evidenced by imputation in which rs13072846 (P = 4.89 × 10−5) was identified and confirmed as female PSS associating SNP within the same LD with rs13079920. PSS pathogenesis involves both immune (effector) and exocrine (target) system. We suggested that while GTF2I is a previously reported associating gene which may function in immune system, RBMS3 is a novel susceptibility gene that predisposes women to PSS potentially through modulating acinar apoptosis and TGF-β signaling in target exocrine system.


Similar content being viewed by others
References
Burbelo PD, Ambatipudi K, Alevizos I (2014) Genome-wide association studies in Sjogren’s syndrome: what do the genes tell us about disease pathogenesis? Autoimmun Rev 13:756–761. doi:10.1016/j.autrev.2014.02.002
Callaghan R, Prabu A, Allan RB, Clarke AE, Sutcliffe N, Pierre YS, Gordon C, Bowman SJ, Group UKSsI (2007) Direct healthcare costs and predictors of costs in patients with primary Sjogren’s syndrome. Rheumatology (Oxford) 46:105–111. doi:10.1093/rheumatology/kel155
Chen J, Kwong DL, Zhu CL, Chen LL, Dong SS, Zhang LY, Tian J, Qi CB, Cao TT, Wong AM, Kong KL, Li Y, Liu M, Fu L, Guan XY (2012) RBMS3 at 3p24 inhibits nasopharyngeal carcinoma development via inhibiting cell proliferation, angiogenesis, and inducing apoptosis. PLoS ONE 7:e44636. doi:10.1371/journal.pone.0044636
Chen CH, Chen HC, Chang CC, Peng YJ, Lee CH, Shieh YS, Hung YJ, Lin YF (2015) Growth arrest-specific 6 protein in patients with Sjogren syndrome: determination of the plasma level and expression in the labial salivary gland. PLoS ONE 10:e0139955. doi:10.1371/journal.pone.0139955
Chisholm DM, Mason DK (1968) Labial salivary gland biopsy in Sjogren’s disease. J Clin Pathol 21:656–660
Forsblad-d’Elia H, Carlsten H, Labrie F, Konttinen YT, Ohlsson C (2009) Low serum levels of sex steroids are associated with disease characteristics in primary Sjogren’s syndrome; supplementation with dehydroepiandrosterone restores the concentrations. J Clin Endocrinol Metab 94:2044–2051. doi:10.1210/jc.2009-0106
Fox RI (2005) Sjogren’s syndrome. Lancet 366:321–331. doi:10.1016/S0140-6736(05)66990-5
Hartkamp A, Geenen R, Godaert GL, Bootsma H, Kruize AA, Bijlsma JW, Derksen RH (2008) Effect of dehydroepiandrosterone administration on fatigue, well-being, and functioning in women with primary Sjogren syndrome: a randomised controlled trial. Ann Rheum Dis 67:91–97. doi:10.1136/ard.2007.071563
Helmick CG, Felson DT, Lawrence RC, Gabriel S, Hirsch R, Kwoh CK, Liang MH, Kremers HM, Mayes MD, Merkel PA, Pillemer SR, Reveille JD, Stone JH, National Arthritis Data W (2008) Estimates of the prevalence of arthritis and other rheumatic conditions in the United States. Part I. Arthritis Rheum 58:15–25. doi:10.1002/art.23177
Howie BN, Donnelly P, Marchini J (2009) A flexible and accurate genotype imputation method for the next generation of genome-wide association studies. PLoS Genet 5:e1000529. doi:10.1371/journal.pgen.1000529
Howie B, Marchini J, Stephens M (2011) Genotype imputation with thousands of genomes. G3 (Bethesda) 1:457–470. doi:10.1534/g3.111.001198
Howie B, Fuchsberger C, Stephens M, Marchini J, Abecasis GR (2012) Fast and accurate genotype imputation in genome-wide association studies through pre-phasing. Nat Genet 44:955–959. doi:10.1038/ng.2354
Ice JA, Li H, Adrianto I, Lin PC, Kelly JA, Montgomery CG, Lessard CJ, Moser KL (2012) Genetics of Sjogren’s syndrome in the genome-wide association era. J Autoimmun 39:57–63. doi:10.1016/j.jaut.2012.01.008
Ishimaru N, Arakaki R, Watanabe M, Kobayashi M, Miyazaki K, Hayashi Y (2003) Development of autoimmune exocrinopathy resembling Sjogren’s syndrome in estrogen-deficient mice of healthy background. Am J Pathol 163:1481–1490. doi:10.1016/S0002-9440(10)63505-5
Ishimaru N, Arakaki R, Yoshida S, Yamada A, Noji S, Hayashi Y (2008) Expression of the retinoblastoma protein RbAp48 in exocrine glands leads to Sjogren’s syndrome-like autoimmune exocrinopathy. J Exp Med 205:2915–2927. doi:10.1084/jem.20080174
Jayasena CS, Bronner ME (2012) Rbms3 functions in craniofacial development by posttranscriptionally modulating TGF-beta signaling. J Cell Biol 199:453–466. doi:10.1083/jcb.201204138
Kong L, Ogawa N, Nakabayashi T, Liu GT, D’Souza E, McGuff HS, Guerrero D, Talal N, Dang H (1997) Fas and Fas ligand expression in the salivary glands of patients with primary Sjogren’s syndrome. Arthritis Rheum 40:87–97
Kong L, Robinson CP, Peck AB, Vela-Roch N, Sakata KM, Dang H, Talal N, Humphreys-Beher MG (1998) Inappropriate apoptosis of salivary and lacrimal gland epithelium of immunodeficient NOD-scid mice. Clin Exp Rheumatol 16:675–681
Konttinen YT, Fuellen G, Bing Y, Porola P, Stegaev V, Trokovic N, Falk SS, Liu Y, Szodoray P, Takakubo Y (2012) Sex steroids in Sjogren’s syndrome. J Autoimmun 39:49–56. doi:10.1016/j.jaut.2012.01.004
Kuo C-F, Grainge MJ, Yu K-H, See L-C, Luo S-F, Valdes AM, Chou IJ (2012) Sibling relative risk and heritability of Sjgren’s syndrome: a nationwide population study in Taiwan. Arthritis Rheum 64:2673
Labrie F (2010) DHEA, important source of sex steroids in men and even more in women. Prog Brain Res 182:97–148. doi:10.1016/S0079-6123(10)82004-7
Labrie F (2015) All sex steroids are made intracellularly in peripheral tissues by the mechanisms of intracrinology after menopause. J Steroid Biochem Mol Biol 145:133–138. doi:10.1016/j.jsbmb.2014.06.001
Li Y, Zhang K, Chen H, Sun F, Xu J, Wu Z, Li P, Zhang L, Du Y, Luan H, Li X, Wu L, Li H, Wu H, Li X, Li X, Zhang X, Gong L, Dai L, Sun L, Zuo X, Xu J, Gong H, Li Z, Tong S, Wu M, Li X, Xiao W, Wang G, Zhu P, Shen M, Liu S, Zhao D, Liu W, Wang Y, Huang C, Jiang Q, Liu G, Liu B, Hu S, Zhang W, Zhang Z, You X, Li M, Hao W, Zhao C, Leng X, Bi L, Wang Y, Zhang F, Shi Q, Qi W, Zhang X, Jia Y, Su J, Li Q, Hou Y, Wu Q, Xu D, Zheng W, Zhang M, Wang Q, Fei Y, Zhang X, Li J, Jiang Y, Tian X, Zhao L, Wang L, Zhou B, Li Y, Zhao Y, Zeng X, Ott J, Wang J, Zhang F (2013) A genome-wide association study in Han Chinese identifies a susceptibility locus for primary Sjogren’s syndrome at 7q11.23. Nat Genet 45:1361–1365. doi:10.1038/ng.2779
Manoussakis MN, Tsinti M, Kapsogeorgou EK, Moutsopoulos HM (2012) The salivary gland epithelial cells of patients with primary Sjogren’s syndrome manifest significantly reduced responsiveness to 17beta-estradiol. J Autoimmun 39:64–68. doi:10.1016/j.jaut.2012.01.005
Marchini J, Howie B, Myers S, McVean G, Donnelly P (2007) A new multipoint method for genome-wide association studies by imputation of genotypes. Nat Genet 39:906–913. doi:10.1038/ng2088
McCartney-Francis NL, Mizel DE, Redman RS, Frazier-Jessen M, Panek RB, Kulkarni AB, Ward JM, McCarthy JB, Wahl SM (1996) Autoimmune Sjogren’s-like lesions in salivary glands of TGF-beta1-deficient mice are inhibited by adhesion-blocking peptides. J Immunol 157:1306–1312
Pan WH, Fann CS, Wu JY, Hung YT, Ho MS, Tai TH, Chen YJ, Liao CJ, Yang ML, Cheng AT, Chen YT (2006) Han Chinese cell and genome bank in Taiwan: purpose, design and ethical considerations. Hum Hered 61:27–30. doi:10.1159/000091834
Ponten F, Jirstrom K, Uhlen M (2008) The Human Protein Atlas–a tool for pathology. J Pathol 216:387–393. doi:10.1002/path.2440
Porola P, Virkki L, Przybyla BD, Laine M, Patterson TA, Pihakari A, Konttinen YT (2008) Androgen deficiency and defective intracrine processing of dehydroepiandrosterone in salivary glands in Sjogren’s syndrome. J Rheumatol 35:2229–2235
Rahimi Darabad R, Suzuki T, Richards SM, Jakobiec FA, Zakka FR, Barabino S, Sullivan DA (2014) Does estrogen deficiency cause lacrimal gland inflammation and aqueous-deficient dry eye in mice? Exp Eye Res 127:153–160. doi:10.1016/j.exer.2014.07.017
Rajaiya J, Nixon JC, Ayers N, Desgranges ZP, Roy AL, Webb CF (2006) Induction of immunoglobulin heavy-chain transcription through the transcription factor Bright requires TFII-I. Mol Cell Biol 26:4758–4768. doi:10.1128/MCB.02009-05
Robinson CP, Yamachika S, Alford CE, Cooper C, Pichardo EL, Shah N, Peck AB, Humphreys-Beher MG (1997) Elevated levels of cysteine protease activity in saliva and salivary glands of the nonobese diabetic (NOD) mouse model for Sjogren syndrome. Proc Natl Acad Sci USA 94:5767–5771
Schot LP, Verheul HA, Schuurs AH (1984) Effect of nandrolone decanoate on Sjogren’s syndrome like disorders in NZB/NZW mice. Clin Exp Immunol 57:571–574
Shim GJ, Warner M, Kim HJ, Andersson S, Liu L, Ekman J, Imamov O, Jones ME, Simpson ER, Gustafsson JA (2004) Aromatase-deficient mice spontaneously develop a lymphoproliferative autoimmune disease resembling Sjogren’s syndrome. Proc Natl Acad Sci USA 101:12628–12633. doi:10.1073/pnas.0405099101
Spaan M, Porola P, Laine M, Rozman B, Azuma M, Konttinen YT (2009) Healthy human salivary glands contain a DHEA-sulphate processing intracrine machinery, which is deranged in primary Sjogren’s syndrome. J Cell Mol Med 13:1261–1270. doi:10.1111/j.1582-4934.2009.00727.x
Sullivan RM, Cermak JM, Papas AS, Dana MR, Sullivan DA (2002) Economic and quality of life impact of dry eye symptoms in women with Sjogren’s syndrome. Adv Exp Med Biol 506:1183–1188
Sullivan DA, Belanger A, Cermak JM, Berube R, Papas AS, Sullivan RM, Yamagami H, Dana MR, Labrie F (2003) Are women with Sjogren’s syndrome androgen-deficient? J Rheumatol 30:2413–2419
Taiym S, Haghighat N, Al-Hashimi I (2004) A comparison of the hormone levels in patients with Sjogren’s syndrome and healthy controls. Oral Surg Oral Med Oral Pathol Oral Radiol Endod 97:579–583. doi:10.1016/S107921040400068X
Valtysdottir ST, Wide L, Hallgren R (2001) Low serum dehydroepiandrosterone sulfate in women with primary Sjogren’s syndrome as an isolated sign of impaired HPA axis function. J Rheumatol 28:1259–1265
Virkki LM, Porola P, Forsblad-d’Elia H, Valtysdottir S, Solovieva SA, Konttinen YT (2010) Dehydroepiandrosterone (DHEA) substitution treatment for severe fatigue in DHEA-deficient patients with primary Sjogren’s syndrome. Arthritis Care Res (Hoboken) 62:118–124. doi:10.1002/acr.20022
Vitali C, Bombardieri S, Jonsson R, Moutsopoulos HM, Alexander EL, Carsons SE, Daniels TE, Fox PC, Fox RI, Kassan SS, Pillemer SR, Talal N, Weisman MH, European Study Group on Classification Criteria for Sjogren’s S (2002) Classification criteria for Sjogren’s syndrome: a revised version of the European criteria proposed by the American-European Consensus Group. Ann Rheum Dis 61:554–558
Zheng J, Huang R, Huang Q, Deng F, Chen Y, Yin J, Chen J, Wang Y, Shi G, Gao X, Liu Z, Petersen F, Yu X (2015) The GTF2I rs117026326 polymorphism is associated with anti-SSA-positive primary Sjogren’s syndrome. Rheumatology (Oxford) 54:562–564. doi:10.1093/rheumatology/keu466
Acknowledgments
We thank the Academia Sinica Genomic Medicine Multicenter Study (40-05-GMM) and the National Center for Genome Medicine at Academia Sinica, Taiwan, for technical/bioinformatics support. This Center was supported by grants from the National Core Facility Program for Biotechnology of National Science Council, Taiwan (NSC102-2319-B-001-001). We thank the Translational Resource Center for Genomic Medicine (TRC, NSC102-2325-B-001-040) of National Research Program for Biopharmaceuticals (NRPB) for the support in sample management. We thank the Taiwan Biobank for providing the allele frequency and NGS data of controls for our replication.
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare that there are no conflicts of interest.
Additional information
I.-W. Song and H.-C. Chen contribute equally to this work.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Song, IW., Chen, HC., Lin, YF. et al. Identification of susceptibility gene associated with female primary Sjögren’s syndrome in Han Chinese by genome-wide association study. Hum Genet 135, 1287–1294 (2016). https://doi.org/10.1007/s00439-016-1716-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00439-016-1716-0