Abstract
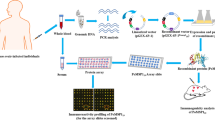
Plasmodium knowlesi utilizes the Duffy binding protein alpha (PkDBPα) to facilitate its invasion into human erythrocytes. PkDBPα region II (PkDBPαII) from Peninsular Malaysia and Malaysian Borneo has been shown to occur as distinct haplotypes, and the predominant haplotypes from these geographical areas demonstrated differences in binding activity to human erythrocytes in erythrocyte binding assays. This study aimed to determine the effects of genetic polymorphisms in PkDBPαII to immune responses in animal models. The recombinant PkDBPαII (~ 45 kDa) of Peninsular Malaysia (PkDBPαII-H) and Malaysian Borneo (PkDBPαII-S) were expressed in a bacterial expression system, purified, and used in mice and rabbit immunization. The profile of cytokines IL-1ra, IL-2, IL-6, IL-10, TNF-α, and IFN-γ in immunized mice spleen was determined via ELISA. The titer and IgG subtype distribution of raised antibodies was characterized. Immunized rabbit sera were purified and used to perform an in vitro merozoite invasion inhibition assay. The PkDBPαII-immunized mice sera of both groups showed high antibody titer and a similar IgG subtype distribution pattern: IgG2b > IgG1 > IgG2a > IgG3. The PkDBPαII-H group was shown to have higher IL-1ra (P = 0.141) and IL-6 (P = 0.049) concentrations, with IL-6 levels significantly higher than that of the PkDBPαII-S group (P ≤ 0.05). Merozoite invasion inhibition assay using purified anti-PkDBPαII antibodies showed a significantly higher inhibition rate in the PkDBPαII-H group than the PkDBPαII-S group (P ≤ 0.05). Besides, anti-PkDBPαII-H antibodies were able to exhibit inhibition activity at a lower concentration than anti-PkDBPαII-S antibodies. PkDBPαII was shown to be immunogenic, and the PkDBPαII haplotype from Peninsular Malaysia exhibited higher responses in cytokines IL-1ra and IL-6, antibody IgM level, and merozoite invasion inhibition assay than the Malaysian Borneo haplotype. This suggests that polymorphisms in the PkDBPαII affect the level of immune responses in the host.




Similar content being viewed by others
References
Adams JH, Hudson DE, Torii M, Ward GE, Wellems TE, Aikawa M, Miller LH (1990) The duffy receptor family of Plasmodium knowlesi is located within the micronemes of invasive malaria merozoites. Cell 63:141–153. https://doi.org/10.1016/0092-8674(90)90295-p
Adams JH, Kim LS, Dolan SA, Fang X, Kaslow DC, Miller LH (1992) A family of erythrocyte binding proteins of malaria parasites. Proc Natl Acad Sci USA 89:7085–7089
Angulo I, Fresno M (2002) Cytokines in the pathogenesis of and protection against malaria. Clin Diag Lab Immunol 9(6):1145–1152. https://doi.org/10.1128/CDLI.9.6.1145-1152.2002
Bachmann MF, Oxenius A (2007) Interleukin 2: from immunostimulation to immunoregulation and back again. EMBO Rep 8:1142–1148. https://doi.org/10.1038/sj.embor.7401099
Berger A (2000) Science commentary - Th1 and Th2 responses: what are they? BMJ 321(7258):424. https://doi.org/10.1136/bmj.321.7258.424
Cheong FW, Fong MY, Lau YL, Mahmud R (2013) Immunogenicity of bacterial-expressed recombinant Plasmodium knowlesi merozoite surface protein-142 (MSP-142). Malar J 12(454):1–9. https://doi.org/10.1186/1475-2875-12-454
Chitnis CE (2001) Molecular insights into receptors used by malaria parasites for erythrocyte invasion. Curr Opin Hematol 8:85–91. https://doi.org/10.1097/00062752-200103000-00005
Chitnis C, Miller LH (1994) Identification of the erythrocyte binding domains of Plasmodium vivax and Plasmodium knowlesi proteins involved in erythrocyte invasion. J Exp Med 180:497–506. https://doi.org/10.1084/jem.180.2.497
Corrigan RA, Rowe JA (2010) Strain variation in early innate cytokine induction by Plasmodium falciparum. Parasit Immunol 32(7):512–527. https://doi.org/10.1111/j.1365-3024.2010.01225.x
Cox-Singh J, Davis TME, Lee KS, Shamsul SSG, Matusop A, Ratnam S, Rahman HA, Conway DJ, Singh B (2008) Plasmodium knowlesi malaria in humans is widely distributed and potentially life-threatening. Clin Infect Dis 46(2):165–171. https://doi.org/10.1086/524888
Cox-Singh J, Singh B, Daneshvar C, Planche T, Parker-Williams J, Krishna S (2011) Anti-Inflammatory cytokines predominate in acute human Plasmodium knowlesi infections. PLoS One 6(6):020541. https://doi.org/10.1371/journal.pone.0020541
Dobaño C, Santano R, Vidal M, Jimenez A, Jairoce C, Ubillos I, Dosoo D, Aguilar R, Williams NA, Ayestaran A, Valim C, Asante KP et al (2019) Differential patterns of IgG subclass responses to Plasmodium falciparum antigens in relation to malaria protection and RTS, S vaccination. Front Immunol 10:1–19. https://doi.org/10.3389/fimmu.2019.00439
Farrington L, Vance H, Rek J, Prahl M, Jagannathan P, Katureebe A, Arinaitwe E, Kamya MR, Dorsey G, Feeney ME (2017) Both inflammatory and regulatory cytokine responses to malaria are blunted with increasing age in highly exposed children. Malar J 16(1):1–11. https://doi.org/10.1186/s12936-017-2148-6
Fong MY, Rashdi SAA, Yusof R, Lau YL (2015) Distinct genetic difference between the Duffy binding protein (PkDBPαII) of Plasmodium knowlesi clinical isolates from North Borneo and Peninsular Malaysia. Malar J 14:1–7. https://doi.org/10.1186/s12936-015-0610-x
Ho M, Sexton MM, Tongtawe P, Looareesuwan S, Suntharasamai P, Webster HK (1995) Interleukin-10 inhibits tumor necrosis factor production but not antigen-specific lymphoproliferation in acute Plasmodium falciparum malaria. J Infect Dis 172(3):838–844. https://doi.org/10.1093/infdis/172.3.838
Hussain R, Dawood G, Abrar N, Toossi Z, Minai A, Dojki M, Ellner JJ (1995) Selective increases in antibody isotypes and immunoglobulin G subclass responses to secreted antigens in tuberculosis patients and healthy household contacts of the patients. Clin Diagn Lab Immunol 2(6):726–32. https://doi.org/10.1128/cdli.2.6.726-732.1995
Hussin N, Lim YAL, Goh PP, William T, Jelip J, Mudin RN (2020) Updates on malaria incidence and profile in Malaysia from 2013 to 2017. Malar J 19(55):1–14. https://doi.org/10.1186/s12936-020-3135-x
Jason J, Archibald LK, Nwanyanwu OC, Bell M, Buchanan I, Larned J, Kazembe PN, Dobbie H, Parekh B, Byrd MG, Eick A, Han A, Jarvis WR (2001) Cytokines and malaria parasitemia. Clin Immunol 100(2):208–218. https://doi.org/10.1006/clim.2001.5057
Kawano Y, Noma T, Yata J (1994) Regulation of human IgG subclass production by cytokines IFN-gamma and IL-6 act antagonistically in the induction of human IgG1 but additively in the induction of IgG2. J Immunol 153(11):4948–58
Kwenti TE, Kukwah TA, Dizzle T, Kwenti B, Nyassa BR, Dilonga MH, Enow-Orock G, Tendongfor N, Anong ND, Wanji S, Njunda LA, Nkuo-Akenji T (2019) Comparative analysis of IgG and IgG subclasses against Plasmodium falciparum MSP - 1 19 in children from five contrasting bioecological zones of Cameroon. Malar J 18(16):1–13. https://doi.org/10.1186/s12936-019-2654-9
Lim KL, Amir A, Lau YL, Fong MY (2017) The Duffy binding protein (PkDBPαII) of Plasmodium knowlesi from Peninsular Malaysia and Malaysian Borneo show different binding activity levels to human erythrocytes. Malar J 16(331):1–8. https://doi.org/10.1186/s12936-017-1984-8
Moon RW, Hall J, Rangkuti F, Ho YS, Almond N, Mitchell GH, Pain A, Holder AA, Blackman MJ (2013) Adaptation of the genetically tractable malaria pathogen Plasmodium knowlesi to continuous culture in human erythrocytes. Proc Natl Acad Sci USA 110(2):531–536. https://doi.org/10.1073/pnas.1216457110
Narum DL, Haynes JD, Fuhrmann S, Moch K, Liang H, Hoffmann HL, Kim BK (2000) Antibodies against the plasmodium falciparum receptor binding domain of EBA-175 block invasion pathways that do not involve sialic acids. Infect Immun 68(4):1964–1966. https://doi.org/10.1128/IAI.68.4.1964-1966.2000
Oyegue-Liabagui SL, Bouopda-Tuedom AG, Kouna LC, Maghendji-Nzondo S, Nzoughe H, Tchitoula-Makaya N, Pegha-Moukandja I, Lekana-Douki J-B (2017) Pro- and anti-inflammatory cytokines in children with malaria in Franceville. Gabon Am J Clin Exp Immunol 6(2):9–20
Rovira-Vallbona E, Moncunill G, Bassat Q, Aguilar R, Machevo S, Puyol L, Quintó L, Menéndez C, Chitnis CE, Alonso PL, Dobaño C, Mayor A (2012) Low antibodies against Plasmodium falciparum and imbalanced pro-inflammatory cytokines are associated with severe malaria in Mozambican children: a case – control study. Malar J 11(181):1–11. https://doi.org/10.1186/1475-2875-11-181
Singh S, Pandey K, Chattopadhayay R, Yazdani SS, Lynn A, Bharadwaj A, Ranjan A, Chitnis C (2001) Biochemical, biophysical, and functional characterization of bacterially expressed and refolded receptor binding domain of Plasmodium vivax Duffy-binding protein. J Biol Chem 276(20):17111–17116. https://doi.org/10.1074/jbc.M101531200
Singh AP, Puri SK, Chitnis CE (2002) Antibodies raised against receptor-binding domain of Plasmodium knowlesi Duffy binding protein inhibit erythrocyte invasion. Mol Biochem Parasitol 121(1):21–31. https://doi.org/10.1016/s0166-6851(02)00017-8
Singh SK, Singh AP, Pandey S, Yazdani SS, Chitnis CE, Sharma A (2003) Definition of structural elements in Plasmodium vivax and P. knowlesi Duffy-binding domains necessary for erythrocyte invasion. Biochem J 374(1):193–8. https://doi.org/10.1042/BJ20030622
Singh SK, Hora R, Belrhali H, Chitnis CE, Sharma A (2006) Structural basis for Duffy recognition by the malaria parasite Duffy-binding-like domain. Nature 439:741–744. https://doi.org/10.1038/nature04443
Smith EC, Taylor-Robinson AW (2003) Parasite-specific immunoglobulin isotypes during lethal and non-lethal murine malaria infections. Parasitol Res 89(1):26–33. https://doi.org/10.1007/s00436-002-0687-3
Teo A, Feng G, Brown GV, Beeson JG, Rogerson SJ (2016) Functional antibodies and protection against blood-stage malaria. Trends Parasitol 32(11):887–898. https://doi.org/10.1016/j.pt.2016.07.003
Vanbuskirk KM, Sevova E, Adams JH (2004) Conserved residues in the Plasmodium vivax Duffy-binding protein ligand domain are critical for erythrocyte receptor recognition. Proc Natl Acad Sci USA 101(44):15754–15759. https://doi.org/10.1073/pnas.0405421101
White NJ (2008) Plasmodium knowlesi: The fifth human malaria parasite. Clin Infect Dis 46(2):172–3. https://doi.org/10.1086/524889
William T, Menon J, Rajahram G, Chan L, Ma G, Donaldson S, Khoo S, Frederick C, Jelip J, Anstey NM, Yeo TW (2011) Severe Plasmodium knowlesi Malaria in a tertiary care Hospital Sabah. Malaysia Emerging Infect Dis 17(7):1248–1255. https://doi.org/10.3201/eid1707.101017
World Health Organization, WHO (2021) Malaria World Report 2021. Available online: https://www.who.int/teams/global-malaria-programme/reports/world-malaria-report-2021. Accessed 28 Feb 2022
Acknowledgements
Special thanks to the Department of Parasitology, Faculty of Medicine, Universiti Malaya, for providing the samples and facilities.
Funding
The study was supported by the Fundamental Research Grant Scheme (FRGS), FRGS/1/2018/SKK12/UM/02/1 (FP030-2018A) from the Ministry of Higher Education, Malaysia.
Author information
Authors and Affiliations
Contributions
FWC, YLL, and MYF designed the project and experiments. UKA prepared the manuscript and carried out the experimental procedures, including recombinant protein preparation, animal immunization, PkA1H1 culture preparation, and related assays. FWC was involved in hands-on experiment training. UKA, FWC, YLL, and MYF were involved in the data analysis. The authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Ethics approval
Animal ethics and all experimental protocols have been reviewed and approved by the Faculty of Medicine Institutional Animal Care and Use Committee, University Malaya (2018–211218/PARA/R/CFW).
Conflict of interest
The authors declare no competing interests.
Additional information
Handling Editor: Una Ryan
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Azlan, U.K., Cheong, F.W., Lau, Y.L. et al. Plasmodium knowlesi Duffy binding protein alpha region II (PkDBPαII) in clinical isolates from Peninsular Malaysia and Malaysian Borneo exhibit different immune responses in animal models. Parasitol Res 121, 3443–3454 (2022). https://doi.org/10.1007/s00436-022-07665-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00436-022-07665-7