Abstract
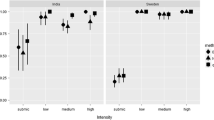
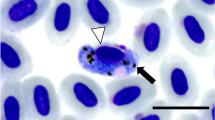
Avian malaria, caused by Plasmodium spp., has been linked to the mortality and population-level declines in native birds in some regions. While molecular diagnostic methods have greatly improved our ability to detect infections of both human and bird malaria, failing to identify false negatives remains an important handicap, particularly for avian malaria due to host DNA presence in the bird blood cells. In an attempt to improve the accuracy of diagnostics by PCR, we evaluated the performance of a commercial silica-membrane-based DNA extraction kit by modifying the protocol with four unpooled elution volume alternatives. Our results suggest that the best template is the DNA extract obtained from the second eluate of a first 50 μL elution step. In one case, the only band visible was from this second eluate and, thus, may not have been identified as positive for Plasmodium spp. if a different elution protocol had been followed. Our results are likely explained by the concept of size exclusion chromatography by which particles of different sizes will elute at different rates. Overall, first elution templates may consist of a lower ratio of parasite to host DNA, while second eluates may contain a higher parasite to host DNA ratio. A low ratio of parasite to host DNA is a concern in detecting chronic infections, in which birds typically carry low levels of parasitemia, making accurate diagnostics imperative when identifying reservoirs of disease that could lead to spillback events.



Similar content being viewed by others
References
Akane A, Matsubara K, Nakamura H, Takahashi S, Kimura K (1994) Identification of the heme compound copurified with deoxyribonucleic acid (DNA) from bloodstains, a major inhibitor of polymerase chain reaction (PCR) amplification. J Forensic Sci 39:362–372. doi:10.1520/JFS13607J
Alaeddini R (2012) Forensic implications of PCR inhibition—a review. Forensic Sci Int Genet 6:297–305. doi:10.1016/j.fsigen.2011.08.006
Alley M, Fairley R, Martin D, Howe L, Atkinson T (2008) An outbreak of avian malaria in captive yellowheads/mohua (Mohoua ochrocephala). N Z Vet J 56:247–251. doi:10.1080/00480169.2008.36842
Alley M, Hale K, Cash W, Ha H, Howe L (2010) Concurrent avian malaria and avipox virus infection in translocated South Island saddlebacks (Philesturnus carunculatus carunculatus). N Z Vet J 58:218–223. doi:10.1080/00480169.2010.68868
Al-soud WA, Rådström P (2001) Purification and characterization of PCR-inhibitory components in blood cells. J Clin Microbiol 39:485–493. doi:10.1128/JCM.39.2.485
Atkinson CT, Samuel MD (2010) Avian malaria Plasmodium relictum in native Hawaiian forest birds: epizootiology and demographic impacts on’apapane Himatione sanguinea. J Avian Biol 41:357–366. doi:10.1111/j.1600-048X.2009.04915.x
Atkinson CT, Utzurrum RB, LaPointe DA, Camp RJ, Crampton LH, Foster JT, Giambelluca TW (2014) Changing climate and the altitudinal range of avian malaria in the Hawaiian Islands—an ongoing conservation crisis on the island of Kaua’i. Glob Chang Biol 20:2426–2436. doi:10.1111/gcb.12535
Atkinson CT, Van Riper C (1991) Pathogenicity and epizootiology of avian haematozoa: Plasmodium, Leucocytozoan, and Haemoproteus. In: Loye JE, Zuk M (eds) Bird-parasite interactions: ecology, evolution, and behaviour. Oxford Univ Press, Oxford, pp 19–48
Bélec L, Authier J, Eliezer-Vanerot MC, Piédouillet C, Mohamed AS, Gherardi RK (1998) Myoglobin as a polymerase chain reaction (PCR) inhibitor: a limitation for PCR from skeletal muscle tissue avoided by the use of thermus thermophilus polymerase. Muscle Nerve 21:1064–1067. doi:10.1002/(SICI)1097-4598(199808)21:8<1064::AID-MUS11>3.0.CO;2-U
Castro I, Howe L, Tompkins DM, Barraclough RK, Slaney D (2011) Presence and seasonal prevalence of Plasmodium spp. in a rare endemic New Zealand passerine (tieke or Saddleback, Philesturnus carunculatus). J Wildl Dis 47:860–7. doi:10.7589/0090-3558-47.4.860
Concepcion GT, Medina M, Toonen RJ (2006) Noncoding mitochondrial loci for corals. Mol Ecol Notes 6:1208–1211. doi:10.1111/j.1471-8286.2006.01493.x
Desneux J, Pourcher A (2014) Comparison of DNA extraction kits and modification of DNA elution procedure for the quantitation of subdominant bacteria from piggery effluents with real-time PCR. Microbiologyopen 3:437–445. doi:10.1002/mbo3.178
Fredricks DN, Relman DA (1998) Improved amplification of microbial DNA from blood cultures by removal of the PCR inhibitor sodium polyanetholesulfonate. J Clin Microbiol 36:2810–2816
Freed L, Cann R (2006) DNA quality and accuracy of avian malaria PCR diagnostics: a review. Condor 108:459–473. doi:10.1650/0010-5422(2006)108[459:DQAAOA]2.0.CO;2
Gillen A, Strausbaugh C, Tindall K (2008) Evaluation of Beta corolliflora for resistance to curly top in Idaho. J Sugar Beet Res 45:99–118
Goodheart J, Valdés Á (2012) Re-evaluation of the Doriopsilla areolata Bergh, 1880 (Mollusca: Opisthobranchia) subspecies complex in the eastern Atlantic Ocean and its relationship to South African Doriopsilla miniata (Alder & Hancock, 1864) based on molecular data. Mar Biodivers 43:113–120. doi:10.1007/s12526-012-0136-1
Hellgren O, Waldenstrom J, Bensch S (2004) A new PCR assay for simultaneous studies of Leucocytozoon, Plasmodium, and Haemoproteus from avian blood. J Parasitol 90:797–802. doi:10.1645/GE-184R1
Hilario E (2004) End labeling procedures: an overview. Mol Biotechnol 28:77–80. doi:10.1385/MB:28:1:77
Howe L, Castro IC, Schoener ER, Hunter S, Barraclough RK, Alley MR (2012) Malaria parasites (Plamodium spp.) infecting introduced, native and endemic New Zealand birds. Parasitol Res 110:913–23. doi:10.1007/s00436-011-2577-z
Khan G, Kangro HO, Coates PJ, Heath RB (1991) Inhibitory effects of urine on the polymerase chain reaction for cytomegalovirus DNA. J Clin Pathol 44:360–365. doi:10.1136/jcp.44.5.360
Kreader CA (1996) Relief of amplification inhibition in PCR with bovine serum albumin or T4 gene 32 protein. Appl Environ Microbiol 62:1102–1106
LaPointe DA, Atkinson CT, Samuel MD (2012) Ecology and conservation biology of avian malaria. Ann N Y Acad Sci 1249:211–26. doi:10.1111/j.1749-6632.2011.06431.x
Massey B, Gleeson D, Slaney D, Tompkins D (2007) PCR detection of Plasmodium and blood meal identification in a native New Zealand mosquito. J Vector Ecol 32:154–156. doi:10.3376/1081-1710(2007)32[154:PDOPAB]2.0.CO;2
Ohmori K, Nakamura K, Kasahara M, Takabatake R, Kitta K, Fujimaki T, Kondo K, Teshima R, Akiyama H (2013) A DNA extraction and purification method using an ion-exchange resin type kit for the detection of genetically modified papaya in processed papaya products. Food Control 32:728–735. doi:10.1016/j.foodcont.2013.01.013
Palinauskas V, Dolnik OV, Valkiūnas G, Bensch S (2010) Laser microdissection microscopy and single cell PCR of avian hemosporidians. J Parasitol 96:420–424. doi:10.1645/GE-2247.1
Regan JF, Furtado MR, Brevnov MG, Jordan JA (2012) A sample extraction method for faster, more sensitive PCR-based detection of pathogens in blood culture. J Mol Diagnostics 14:120–129. doi:10.1016/j.jmoldx.2011.10.001
Ricklefs RE, Fallon SM (2002) Diversification and host switching in avian malaria parasites. Proc Biol Sci 269:885–92. doi:10.1098/rspb.2001.1940
Ricklefs RE, Swanson BL, Fallon SM, Martinez-Abrain A, Scheuerlein A, Gray J, Latta S (2005) Community relationships of avian malaria parasites in southern Missouri. Ecol Monogr 75:543–559. doi:10.1890/04-1820
Toader V, Moldovan I, Sofletea N, Abrudan IV, Curtu AL (2009) DNA isolation and amplification in oak species (Quercus spp.). Bull Transilv Univ Brasov 2:45–50
Tompkins DM, Gleeson DM (2006) Relationship between avian malaria distribution and an exotic invasive mosquito in New Zealand. J R Soc New Zeal 36:51–62. doi:10.1080/03014223.2006.9517799
Tsai Y, Olson BH (1992) Rapid method for separation of bacterial DNA from humic substances in sediments for polymerase chain reaction. Appl Environ Microbiol 58:2292–2295
Ulstrup KE, Van Oppen MJH (2003) Geographic and habitat partitioning of genetically distinct zooxanthellae (Symbiodinium) in Acropora corals on the Great Barrier Reef. Mol Ecol 12:3477–3484. doi:10.1046/j.1365-294X.2003.01988.x
Valkiunas G, Zehtindjiev P, Hellgren O, Ilieva M, Iezhova TA, Bensch S (2007) Linkage between mitochondrial cytochrome b lineages and morphospecies of two avian malaria parasites, with a description of Plasmodium (Novyella) ashfordi sp. nov. Parasitol Res 100:1311–22. doi:10.1007/s00436-006-0409-3
van Riper C, van Riper SG, Goff ML, Laird M (1986) The epizootiology and ecological significance of malaria in Hawaiian land birds. Ecol Monogr 56:327–344. doi:10.2307/1942550
Wang Y, Teraoka I, Hansen FY, Peters GH, Hassager O (2010) A theoretical study of the separation principle in size exclusion chromatography. Macromolecules 43:1651–1659. doi:10.1021/ma902377g
Wilson IG (1997) Inhibition and facilitation of nucleic acid amplification. Appl Environ Microbiol 63:3741–3751
Wood MJ, Cosgrove CL, Wilkin TA, Knowles SCL, Day KP, Sheldon BC (2007) Within-population variation in prevalence and lineage distribution of avian malaria in blue tits, Cyanistes caeruleus. Mol Ecol 16:3263–73. doi:10.1111/j.1365-294X.2007.03362.x
Woodworth BL, Atkinson CT, LaPointe DA, Hart PJ, Spiegel CS, Tweed EJ, Henneman C, Lebrun J, Denette T, Demots R et al (2005) Host population persistence in the face of introduced vector-borne diseases: Hawaii amakihi and avian malaria. Proc Natl Acad Sci U S A 102:1531–6. doi:10.1073/pnas.0409454102
Yang DY, Eng B, Dudar JC, Saunders SR, Waye JS (1997) Removal of PCR inhibitors using silica-based spin columns: application to ancient bones. J Can Soc Forensic Sci 30:1–5. doi:10.1080/00085030.1997.10757080
Ye J, Ji A, Parra EJ, Zheng X, Jiang C, Zhao X, Hu L, Tu Z (2004) A simple and efficient method for extracting DNA from old and burned bone. J Forensic Sci 49:754–759. doi:10.1520/JFS2003275
Zhang Y, Isaacman DJ, Wadowsky RM, Rydquist-White J, Post JC, Ehrlich GD (1995) Detection of Streptococcus pneumoniae in whole blood by PCR. J Clin Microbiol 33:596–601
Acknowledgments
Thanks to Robert Poulin and Daniel Tompkins for providing feedback on earlier drafts of the manuscript as well as Tania King for laboratory support. Additional thanks to the New Zealand Department of Conservation and the staff of Nelson Lakes National Park for their continued support of wildlife disease research. Funding was provided by the Department of Zoology, University of Otago and Landcare Research. Birds were sampled under Otago University Animal Ethics Approval 2/13 and New Zealand Department of Conservation permit 35873-FAU.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Niebuhr, C.N., Blasco-Costa, I. Improving detection of avian malaria from host blood: a step towards a standardised protocol for diagnostics. Parasitol Res 115, 3905–3911 (2016). https://doi.org/10.1007/s00436-016-5157-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00436-016-5157-4