Abstract
Purpose
Assessment of individual VTE risk in cancer patients prior to chemotherapy is critical for determining necessity of interventions. Risk assessment models (RAM) are available but have not been validated for haematological malignancy. We aimed to assess the validity of the Vienna Cancer and Thrombosis Study (V-CATS) score in prediction of VTE in a variety of haematological malignancies.
Methods
This is a prospective cohort study conducted on 81 newly diagnosed cancer patients undergoing chemotherapy. Demographic, clinical and cancer related data were collected, patients were followed up for 6 months, and VTE events were recorded. Khorana score (KS) was calculated. Plasma D-dimer and sP-selectin were measured, and then, V-CATS score was calculated. Receiver operator curve (ROC) was used to assess the sensitivity and specificity of RAMs. A modified V-CATS was generated and subsequently assessed by using new cut-off levels of d-dimer and sP-selectin based on ROC curve of the patients’ results and compared the probability of VTE occurrence using all three RAMs.
Results
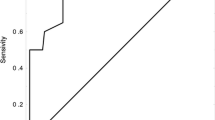
Among the 81 patients included in this study, a total of 2.7% were diagnosed with advanced metastatic cancer. The most frequent cancer was non-Hodgkin lymphoma (39.5%), and 8 patients (9.8%) developed VTE events. The calculated probability of VTE occurrence using KS, V-CATS and modified V-CATS scores at cut-off levels ≥ 3 was 87.5%, 87.5% and 100%, respectively. The AUC in ROC curve of modified Vienna CATS score showed significant difference when compared to that of V-CATS and KS (P = 0.047 and 0.029, respectively).
Conclusion
The findings of our study highlight the value of three VTE risk assessment models in haematological malignancies. The modified V-CATS score demonstrated higher specificity compared to both V-CATS and KS, while all three scores exhibited similar sensitivity. We encourage the implementation of RAMs in haematological cancers for an appropriate use of thromboprophylaxis.




Similar content being viewed by others
Data availability
The research data associated with this paper are available and can be accessed when needed.
Abbreviations
- RAM:
-
Risk assessment models
- V-CATS:
-
Vienna Cancer and Thrombosis Study
- KS:
-
Khorana score
- VTE:
-
Venous thromboembolism
- BMI:
-
Body Mass Index
- sP-sel:
-
Soluble P-selectin
- IRB:
-
Institutional Review Board
- CBC:
-
Complete Blood Count
- INR:
-
International Normalized Ratio
- NLR:
-
Neutrophil Lymphocyte Ratio
- PLR:
-
Platelet Lymphocyte Ratio
- DVT:
-
Deep vein thrombosis
- PE:
-
Pulmonary embolism
- ELISA:
-
Enzyme-linked immunosorbent assay
- MM:
-
Multiple myeloma
- CLL:
-
Chronic lymphocytic leukaemia
- CML:
-
Chronic myeloid leukaemia
References
Adrián S et al (2016) PO-05-Incidence of venous thromboembolism (VTE) in bile duct tumors (BDT) treated with chemotherapy in ambulatory setting. Thromb Res 140:S178
Arpaia G et al (2009) D-dimer before chemotherapy might predict venous thromboembolism. Blood Coag Fibrinol 20(3):170–175
Ates H et al (2017) Diagnostic validity of hematologic parameters in evaluation of massive pulmonary embolism. J Clin Lab Anal 31(5):e22072
Ay C et al (2007) High concentrations of soluble P-selectin are associated with risk of venous thromboembolism and the P-selectin Thr715 variant. Clin Chem 53(7):1235–1243
Ay C et al (2008) High plasma levels of soluble P-selectin are predictive of venous thromboembolism in cancer patients: results from the Vienna Cancer and Thrombosis Study (CATS). Blood 112(7):2703–2708
Colombo R, Gallipoli P, Castelli R (2014) Thrombosis and hemostatic abnormalities in hematological malignancies. Clin Lymphoma Myeloma Leuk 14(6):441–450
Connolly GC et al (2010) Leukocytosis, thrombosis and early mortality in cancer patients initiating chemotherapy. Thromb Res 126(2):113–118
Di Nisio M et al (2007) Diagnostic accuracy of D-dimer test for exclusion of venous thromboembolism: a systematic review. J Thromb Haemost 5(2):296–304
Donnellan E, Khorana AA (2017) Cancer and venous thromboembolic disease: a review. Oncologist 22(2):199–207
El-sayed HA et al (2022) Validity of modified Vienna-CATS score for prediction of venous thromboembolism in Egyptian cancer cases. Egyptian J Hospital Med 89(2):6831–6838
Falanga A (2011) The cancer-thrombosis connection. Hematologist. https://doi.org/10.1182/hem.V8.4.1244
Ferroni P et al (2015) Venous thromboembolism risk prediction in ambulatory cancer patients: clinical significance of neutrophil/lymphocyte ratio and platelet/lymphocyte ratio. Int J Cancer 136(5):1234–1240
Ferroni P et al (2017) Validation of a machine learning approach for venous thromboembolism risk prediction in oncology. Dis Markers 2017:1
Fitzmauric C et al (2019) Global, regional, and national cancer incidence, mortality, years of life lost, years lived with disability, and disability-adjusted life-years for 29 cancer groups, 1990 to 2017: a systematic analysis for the global burden of disease study. JAMA Oncol 5(12):1749–1768
Khorana AA et al (2005) Risk factors for chemotherapy-associated venous thromboembolism in a prospective observational study. Cancer 104(12):2822–2829
Khorana AA et al (2008) Development and validation of a predictive model for chemotherapy-associated thrombosis. Blood 111(10):4902–4907
Kröger K et al (2006) Risk factors for venous thromboembolic events in cancer patients. Ann Oncol 17(2):297–303
Mirza A-S et al (2019) Validation of the Khorana score in acute myeloid leukemia patients: a single-institution experience. Thromb J 17(1):1–6
Montoro J et al (2020) Uniform graft-versus-host disease prophylaxis with posttransplant cyclophosphamide, sirolimus, and mycophenolate mofetil following hematopoietic stem cell transplantation from haploidentical, matched sibling and unrelated donors. Bone Marrow Transplant 55(11):2147–2159
Mulder FI et al (2019) The Khorana score for prediction of venous thromboembolism in cancer patients: a systematic review and meta-analysis. Haematologica 104(6):1277
Noble S et al (2017) Predictors of active cancer thromboembolic outcomes: validation of the Khorana score among patients with lung cancer: comment. J Thromb Haemost 15(3):590–591
Pabinger I, Ay C (2009) Biomarkers and venous thromboembolism. Arterioscler Thromb Vasc Biol 29(3):332–336
Peng L et al (2022) High level of neutrophil to lymphocyte ratio increases the risk of deep venous thrombosis in intensive care unit patients after oral cancer surgery: a retrospective study. Ann Transl Med. https://doi.org/10.21037/atm-22-2453
Polgar J, Matuskova J, Wagner D (2005) The P-selectin, tissue factor, coagulation triad. J Thromb Haemost 3(8):1590–1596
Rickles FR et al (2007) Bleeding and thrombosis in acute leukemia: what does the future of therapy look like? Thromb Res 120:S99–S106
Riondino S et al (2019) Predicting VTE in cancer patients: candidate biomarkers and risk assessment models. Cancer 11(1):95
Sallah S, Wan JY, Nguyen NP (2002) Venous thrombosis in patients with solid tumors: determination of frequency and characteristics. Thromb Haemost 87(04):575–579
Simanek R et al (2010) High platelet count associated with venous thromboembolism in cancer patients: results from the Vienna Cancer and Thrombosis Study (CATS). J Thromb Haemost 8(1):114–120
Streiff MB et al (2015) Cancer-associated venous thromboembolic disease, version1. 2015. J Natl Compr Cancer Netw 13(9):1079–1095
Timp JF et al (2013) Epidemiology of cancer-associated venous thrombosis Blood. J Am Soc Hematol 122(10):1712–1723
Trujillo-Santos J et al (2008) Elevated white blood cell count and outcome in cancer patients with venous thromboembolism. Thromb Haemost 100(05):905–911
Uaprasert N et al (2010) Venous thromboembolism in multiple myeloma: current perspectives in pathogenesis. Eur J Cancer 46(10):1790–1799
Verhovsek M et al (2008) Systematic review: D-dimer to predict recurrent disease after stopping anticoagulant therapy for unprovoked venous thromboembolism. Ann Intern Med 149(7):481–490
Wu G et al (2015) Combination of platelet to lymphocyte ratio and neutrophil to lymphocyte ratio is a useful prognostic factor in advanced non-small cell lung cancer patients. Thoracic Cancer 6(3):275–287
Wun T, White RH (2010) Venous thromboembolism in patients with acute leukemia, lymphoma, and multiple myeloma. Thromb Res 125:S96–S102
Funding
This study was primarily personally funded. Mansoura University facilitated patient recruitment and provided research laboratory facilities.
Author information
Authors and Affiliations
Contributions
HE conducted all practical work, tabulated and analyzed data and wrote the first draft of the manuscript. RB critically evaluated and wrote the manuscript. MO critically evaluated the data, guided data interpretation and data presentation, and critically reviewed and wrote the manuscript. AE, ME, YT, DS, HG reviewed the data and manuscript. TS designed the study and supervised the study. The contributions of authors were in accordance with the journal's guidelines.
Corresponding author
Ethics declarations
Conflict of interest
All authors have no conflict of interest related to this work.
Ethical approval
The research adhered to the Code of Ethics of the World Medical Association (Declaration of Helsinki) for studies involving human subjects. The project was approved by Mansoura University Board of ethics.
Patient consent statement
All the patients involved in the study provided the written consent for participation and publication of scientific data.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
EL-Sayed, H.A., Othman, M., Azzam, H. et al. Assessing the risk of venous thromboembolism in patients with haematological cancers using three prediction models. J Cancer Res Clin Oncol 149, 17771–17780 (2023). https://doi.org/10.1007/s00432-023-05475-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00432-023-05475-7