Abstract
Background
Breast cancer patients with brain metastasis (BM) have a poor prognosis. This study aims to identify the risk factors of BM in patients with metastatic breast cancer (MBC) and establish a competing risk model for predicting the risk of brain metastases at different time points along the course of disease.
Methods
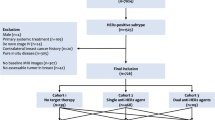
Patients with MBC admitted to the breast disease center of Peking University First Hospital from 2008 to 2019 were selected and retrospectively analyzed to establish a risk prediction model for brain metastases. Patients with MBC admitted to eight breast disease centers from 2015 to 2017 were selected for external validation of the competing risk model. The competing risk approach was used to estimate cumulative incidence. Univariate Fine-Gray competing risk regression, optimal subset regression, and LASSO Cox regression were used to screen potential predictors of brain metastases. Based on the results, a competing risk model for predicting brain metastases was established. The discrimination of the model was evaluated using AUC, Brier score, and C-index. The calibration was evaluated by the calibration curves. The model was assessed for clinical utility by decision curve analysis (DCA), as well as by comparing the cumulative incidence of brain metastases between groups with different predicted risks.
Results
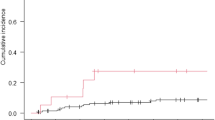
From 2008 to 2019, a total of 327 patients with MBC in the breast disease center of Peking University First Hospital were admitted into the training set for this study. Among them, 74 (22.6%) patients developed brain metastases. From 2015 to 2017, a total of 160 patients with MBC in eight breast disease centers were admitted into the validation set for this study. Among them, 26 (16.3%) patients developed brain metastases. BMI, age, histological type, breast cancer subtype, and extracranial metastasis pattern were included in the final competing risk model for BM. The C-index of the prediction model in the validation set was 0.695, and the AUCs for predicting the risk of brain metastases within 1, 3, and 5 years were 0.674, 0.670, and 0.729, respectively. Time-dependent DCA curves demonstrated a net benefit of the prediction model with thresholds of 9–26% and 13–40% when predicting the risk of brain metastases at 1 and 3 years, respectively. Significant differences were observed in the cumulative incidence of brain metastases between groups with different predicted risks (P < 0.05 by Gray’s test).
Conclusions
In this study, a competing risk model for BM was innovatively established, with the multicenter data being used as an independent external validation set to confirm the predictive efficiency and universality of the model. The C-index, calibration curves, and DCA of the prediction model indicated good discrimination, calibration, and clinical utility, respectively. Considering the high risk of death in patients with metastatic breast cancer, the competing risk model of this study is more accurate in predicting the risk of brain metastases compared with the traditional Logistic and Cox regression models.











Similar content being viewed by others

Data availability
The datasets generated during and/or analysed during the current study are available from the corresponding author on reasonable request.
References
Andrews DW et al (2004) Whole brain radiation therapy with or without stereotactic radiosurgery boost for patients with one to three brain metastases: phase III results of the RTOG 9508 randomised trial. Lancet 363:1665–1672. https://doi.org/10.1016/s0140-6736(04)16250-8
Arvold ND et al (2012) Brain metastases after breast-conserving therapy and systemic therapy: incidence and characteristics by biologic subtype. Breast Cancer Res Treat 136:153–160. https://doi.org/10.1007/s10549-012-2243-x
Awada A et al (2016) Neratinib plus paclitaxel vs trastuzumab plus paclitaxel in previously untreated metastatic ERBB2-positive breast cancer: the NEfERT-T randomized clinical trial. JAMA Oncol 2:1557–1564. https://doi.org/10.1001/jamaoncol.2016.0237
Azim HA, Abdel-Malek R, Kassem L (2018) Predicting brain metastasis in breast cancer patients: stage versus biology. Clin Breast Cancer 18:e187–e195. https://doi.org/10.1016/j.clbc.2017.08.004
Barnholtz-Sloan JS, Sloan AE, Davis FG, Vigneau FD, Lai P, Sawaya RE (2004) Incidence proportions of brain metastases in patients diagnosed (1973–2001) in the Metropolitan Detroit Cancer Surveillance System. J Clin Oncol 22:2865–2872. https://doi.org/10.1200/jco.2004.12.149
Bartsch R et al (2022) Trastuzumab deruxtecan in HER2-positive breast cancer with brain metastases: a single-arm, phase 2 trial. Nat Med 28:1840–1847. https://doi.org/10.1038/s41591-022-01935-8
Brown PD et al (2017) Postoperative stereotactic radiosurgery compared with whole brain radiotherapy for resected metastatic brain disease (NCCTG N107C/CEC·3): a multicentre, randomised, controlled, phase 3 trial. Lancet Oncol 18:1049–1060. https://doi.org/10.1016/s1470-2045(17)30441-2
Brown PD et al (2020) Hippocampal avoidance during whole-brain radiotherapy plus memantine for patients with brain metastases: phase III trial NRG oncology CC001. J Clin Oncol 38:1019–1029. https://doi.org/10.1200/jco.19.02767
Cheng X, Xia L, Sun S (2021) A pre-operative MRI-based brain metastasis risk-prediction model for triple-negative breast cancer. Gland Surg 10:2715–2723. https://doi.org/10.21037/gs-21-537
Cho SY, Choi HY (1980) Causes of death and metastatic patterns in patients with mammary cancer. Ten-year autopsy study. Am J Clin Pathol 73:232–234. https://doi.org/10.1093/ajcp/73.2.232
Darlix A et al (2019) Impact of breast cancer molecular subtypes on the incidence, kinetics and prognosis of central nervous system metastases in a large multicentre real-life cohort. Br J Cancer 121:991–1000. https://doi.org/10.1038/s41416-019-0619-y
Duchnowska R et al (2015) Predicting early brain metastases based on clinicopathological factors and gene expression analysis in advanced HER2-positive breast cancer patients. J Neurooncol 122:205–216. https://doi.org/10.1007/s11060-014-1704-y
Freedman RA et al (2019) TBCRC 022: a phase II trial of neratinib and capecitabine for patients with human epidermal growth factor receptor 2-positive breast cancer and brain metastases. J Clin Oncol 37:1081–1089. https://doi.org/10.1200/jco.18.01511
Frisk G, Svensson T, Bäcklund LM, Lidbrink E, Blomqvist P, Smedby KE (2012) Incidence and time trends of brain metastases admissions among breast cancer patients in Sweden. Br J Cancer 106:1850–1853. https://doi.org/10.1038/bjc.2012.163
Gabani P et al (2019) Treatment response as predictor for brain metastasis in triple negative breast cancer: a score-based model. Breast J 25:363–372. https://doi.org/10.1111/tbj.13230
Gauthier H et al (2010) Survival of breast cancer patients with meningeal carcinomatosis. Ann Oncol 21:2183–2187. https://doi.org/10.1093/annonc/mdq232
Gradishar WJ et al (2022) Breast cancer, version 3.2022, NCCN clinical practice guidelines in oncology. J Natl Compr Cancer Netw JNCCN 20:691–722. https://doi.org/10.6004/jnccn.2022.0030
Graesslin O et al (2010) Nomogram to predict subsequent brain metastasis in patients with metastatic breast cancer. J Clin Oncol 28:2032–2037. https://doi.org/10.1200/jco.2009.24.6314
Hammond ME et al (2010) American Society of Clinical Oncology/College Of American Pathologists guideline recommendations for immunohistochemical testing of estrogen and progesterone receptors in breast cancer. J Clin Oncol 28:2784–2795. https://doi.org/10.1200/jco.2009.25.6529
Jerusalem G et al (2022) Trastuzumab deruxtecan in HER2-positive metastatic breast cancer patients with brain metastases: a DESTINY-Breast01 subgroup analysis. Cancer Discov 12:2754–2762. https://doi.org/10.1158/2159-8290.CD-22-0837
Kim YJ, Kim JS, Kim IA (2018) Molecular subtype predicts incidence and prognosis of brain metastasis from breast cancer in SEER database. J Cancer Res Clin Oncol 144:1803–1816. https://doi.org/10.1007/s00432-018-2697-2
Klimov S et al (2017) Novel immunohistochemistry-based signatures to predict metastatic site of triple-negative breast cancers. Br J Cancer 117:826–834. https://doi.org/10.1038/bjc.2017.224
Koniali L et al (2020) Risk factors for breast cancer brain metastases: a systematic review. Oncotarget 11:650–669. https://doi.org/10.18632/oncotarget.27453
Krop IE et al (2015) Trastuzumab emtansine (T-DM1) versus lapatinib plus capecitabine in patients with HER2-positive metastatic breast cancer and central nervous system metastases: a retrospective, exploratory analysis in EMILIA. Ann Oncol 26:113–119. https://doi.org/10.1093/annonc/mdu486
Lin NU, Bellon JR, Winer EP (2004) CNS metastases in breast cancer. J Clin Oncol 22:3608–3617. https://doi.org/10.1200/jco.2004.01.175
Lin NU et al (2020) Intracranial efficacy and survival with tucatinib plus trastuzumab and capecitabine for previously treated HER2-positive breast cancer with brain metastases in the HER2CLIMB trial. J Clin Oncol 38:2610–2619. https://doi.org/10.1200/JCO.20.00775
Montemurro F et al (2020) Trastuzumab emtansine (T-DM1) in patients with HER2-positive metastatic breast cancer and brain metastases: exploratory final analysis of cohort 1 from KAMILLA, a single-arm phase IIIb clinical trial☆. Ann Oncol 31:1350–1358. https://doi.org/10.1016/j.annonc.2020.06.020
Murthy RK et al (2020) Tucatinib, trastuzumab, and capecitabine for HER2-positive metastatic breast cancer. N Engl J Med 382:597–609. https://doi.org/10.1056/NEJMoa1914609
Pestalozzi BC et al (2006) Identifying breast cancer patients at risk for central nervous system (CNS) metastases in trials of the international breast cancer study group (IBCSG). Ann Oncol 17:935–944. https://doi.org/10.1093/annonc/mdl064
Saura C et al (2020) Neratinib plus capecitabine versus lapatinib plus capecitabine in HER2-positive metastatic breast cancer previously treated with ≥ 2 HER2-directed regimens: phase III NALA trial. J Clin Oncol 38:3138–3149. https://doi.org/10.1200/jco.20.00147
Siegel RL, Miller KD, Wagle NS, Jemal A (2023) Cancer statistics, 2023. CA Cancer J Clin 73:17–48. https://doi.org/10.3322/caac.21763
Smedby KE, Brandt L, Bäcklund ML, Blomqvist P (2009) Brain metastases admissions in Sweden between 1987 and 2006. Br J Cancer 101:1919–1924. https://doi.org/10.1038/sj.bjc.6605373
Sperduto PW et al (2012) Effect of tumor subtype on survival and the graded prognostic assessment for patients with breast cancer and brain metastases. Int J Radiat Oncol Biol Phys 82:2111–2117. https://doi.org/10.1016/j.ijrobp.2011.02.027
Subbiah IM et al (2015) Validation and development of a modified breast graded prognostic assessment as a tool for survival in patients with breast cancer and brain metastases. J Clin Oncol 33:2239–2245. https://doi.org/10.1200/jco.2014.58.8517
Sun M-S, Liu Y-H, Ye J-M, Liu Q, Cheng Y-J, Xin L, Xu L (2021) A nomogram for predicting brain metastasis in patients with de novo stage IV breast cancer. Ann Transl Med 9:853. https://doi.org/10.21037/atm-21-1808
Takada M et al (2018) Prediction of postoperative disease-free survival and brain metastasis for HER2-positive breast cancer patients treated with neoadjuvant chemotherapy plus trastuzumab using a machine learning algorithm. Breast Cancer Res Treat 172:611–618. https://doi.org/10.1007/s10549-018-4958-9
Tsukada Y, Fouad A, Pickren JW, Lane WW (1983) Central nervous system metastasis from breast carcinoma. Autopsy study. Cancer 52:2349–2354. https://doi.org/10.1002/1097-0142(19831215)52:12%3c2349::aid-cncr2820521231%3e3.0.co;2-b
Wolff AC et al (2013) Recommendations for human epidermal growth factor receptor 2 testing in breast cancer: American Society of Clinical Oncology/College of American Pathologists clinical practice guideline update. J Clin Oncol 31:3997–4013. https://doi.org/10.1200/jco.2013.50.9984
Xue J, Peng G, Yang JS, Ding Q, Cheng J (2013) Predictive factors of brain metastasis in patients with breast cancer. Med Oncol 30:337. https://doi.org/10.1007/s12032-012-0337-2
Funding
This study was supported by National Key R&D Program of China (2016YFC0901302) and Interdisciplinary Clinical Research Project of Peking University First Hospital (2019CR38).
Author information
Authors and Affiliations
Contributions
All authors contributed to the study conception and design. Material preparation, data collection and analysis were performed by QW and M-SS. The first draft of the manuscript was written by QW and M-SS, and all authors commented on previous versions of the manuscript. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors have no relevant financial or non-financial interests to disclose.
Ethical approval
This study was performed in line with the principles of the Declaration of Helsinki. The ethical approval of this retrospective study was divided into two parts. First, for patients from the Breast Disease Center of Peking University First Hospital, this study was approved by the Ethics Review Committee of Peking University First Hospital (No.2021-283), and informed consent was obtained by telephone during the follow-up. For patients collected from the Breast Cancer Multicenter Clinical Cohort (2016YFC0901302), the study was approved by each participating institution's ethics review committee. Given the retrospective and anonymous nature of the study, additional informed consent specific to this study was not required.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Wu, Q., Sun, MS., Liu, YH. et al. Development and external validation of a prediction model for brain metastases in patients with metastatic breast cancer. J Cancer Res Clin Oncol 149, 12333–12353 (2023). https://doi.org/10.1007/s00432-023-05125-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00432-023-05125-y