Abstract
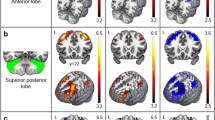
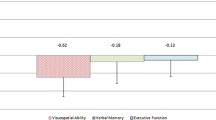
Spinocerebellar ataxia type 6 (SCA6) is a genetic disease that causes degeneration of Purkinje cells, and recent evidence points to degeneration of Betz cells in the motor cortex. The relation between functional activity of motor cortex and symptom severity during a hand-grip motor control in vivo has not yet been investigated. This study explored both functional changes in the sensorimotor cortex and cerebellar regions and structural alterations in the cerebellum for SCA6 patients as compared to age-matched healthy controls using a multimodal imaging approach (task-based fMRI, task-based functional connectivity, and free-water diffusion MRI). Further, we tested their relation with the severity of ataxia symptoms. SCA6 patients had reduced functional activity in the sensorimotor cortex, supplementary motor area (SMA), cerebellar vermis, and cerebellar lobules I-VI (corrected P < 0.05). Reduced task-based functional connectivity between cortical motor regions (i.e., primary motor cortex and SMA) and cerebellar regions (i.e., vermis and lobules I–VI) was found in SCA6 (corrected P < 0.05). SCA6 had elevated free-water values throughout the cerebellum as compared with controls (corrected P < 0.05). Importantly, reduced functional activity in the sensorimotor cortex and SMA and increased free-water in the superior cerebellar peduncle and cerebellar lobule V were related to more severe symptoms in SCA6 (all pairs: R 2 ≥ 0.4 and corrected P < 0.05). Current results demonstrate that impaired functional activity in sensorimotor cortex and SMA and elevated free-water of lobule V and superior cerebellar peduncle are both related to symptom severity, and may provide candidate biomarkers for SCA6.





Similar content being viewed by others
References
Bastian AJ (2006) Learning to predict the future: the cerebellum adapts feedforward movement control. Curr Opin Neurobiol 16:645–649. doi:10.1016/j.conb.2006.08.016
Benjamini Y, Hochberg Y (1995) Controlling the false discovery rate: a practical and powerful approach to multiple testing. J Roy Stat Soc B Met 57:289–300
Benjamini Y, Yekutieli D (2001) The control of the false discovery rate in multiple testing under dependency. Ann Stat 29:1165–1188. doi:10.1214/aos/1013699998
Bernard JA et al (2012) Resting state cortico-cerebellar functional connectivity networks: a comparison of anatomical and self-organizing map approaches. Front Neuroanat 6:31. doi:10.3389/Fnana.2012.00031
Bernard JA et al (2014) Dissociable functional networks of the human dentate nucleus. Cereb Cortex 24:2151–2159. doi:10.1093/cercor/bht065
Burciu RG et al (2015) Distinct patterns of brain activity in progressive supranuclear palsy and Parkinson’s disease. Mov Disord 30:1248–1258. doi:10.1002/mds.26294
Diedrichsen J (2006) A spatially unbiased atlas template of the human cerebellum. Neuroimage 33:127–138. doi:10.1016/j.neuroimage.2006.05.056
Diedrichsen J, Balsters JH, Flavell J, Cussans E, Ramnani N (2009) A probabilistic MR Atlas of the human cerebellum. Neuroimage 46:39–46. doi:10.1016/j.neuroimage.2009.01.045
Eichler L, Bellenberg B, Hahn HK, Koster O, Schols L, Lukas C (2011) Quantitative assessment of brain stem and cerebellar atrophy in spinocerebellar ataxia types 3 and 6: impact on clinical status. Am J Neuroradiol 32:890–897. doi:10.3174/ajnr.A2387
Falcon MI, Gomez CM, Chen EE, Shereen A, Solodkin A (2015) Early cerebellar network shifting in spinocerebellar ataxia type 6. Cereb Cortex. doi:10.1093/cercor/bhv154
Fonov V, Evans AC, Botteron K, Almli CR, McKinstry RC, Collins DL (2011) Unbiased average age-appropriate atlases for pediatric studies. Neuroimage 54:313–327. doi:10.1016/j.neuroimage.2010.07.033
Gierga K et al (2009) Spinocerebellar ataxia type 6 (SCA6): neurodegeneration goes beyond the known brain predilection sites. Neuropathol Appl Neurobiol 35:515–527. doi:10.1111/j.1365-2990.2009.01015.x
Ilg W, Golla H, Thier P, Giese MA (2007) Specific influences of cerebellar dysfunctions on gait. Brain 130:786–798. doi:10.1093/brain/awl376
Jo HJ, Saad ZS, Simmons WK, Milbury LA, Cox RW (2010) Mapping sources of correlation in resting state FMRI, with artifact detection and removal. Neuroimage 52:571–582. doi:10.1016/j.neuroimage.2010.04.246
Kelly RM, Strick PL (2003) Cerebellar loops with motor cortex and prefrontal cortex of a nonhuman primate. J Neurosci 23:8432–8444
Maier-Hein KH et al (2015) Widespread white matter degeneration preceding the onset of dementia. Alzheimers Dement 11:485–493. doi:10.1016/j.jalz.2014.04.518
Mayka MA, Corcos DM, Leurgans SE, Vaillancourt DE (2006) Three-dimensional locations and boundaries of motor and premotor cortices as defined by functional brain imaging: a meta-analysis. Neuroimage 31:1453–1474. doi:10.1016/j.neuroimage.2006.02.004
Middleton FA, Strick PL (2000) Basal ganglia and cerebellar loops: motor and cognitive circuits. Brain Res Rev 31:236–250. doi:10.1016/S0165-0173(99)00040-5
Morton SM, Bastian AJ (2004) Cerebellar control of balance and locomotion. Neuroscientist 10:247–259. doi:10.1177/1073858404263517
Neely KA et al (2015) Functional brain activity relates to 0–3 and 3–8 Hz force oscillations in essential tremor. Cereb Cortex 25:4191–4202. doi:10.1093/cercor/bhu142
Ofori E, Du G, Babcock D, Huang X, Vaillancourt DE (2015a) Parkinson’s disease biomarkers program brain imaging repository. Neuroimage 124:1120–1124. doi:10.1016/j.neuroimage.2015.05.005
Ofori E et al (2015b) Increased free water in the substantia nigra of Parkinson’s disease: a single-site and multi-site study. Neurobiol Aging 36:1097–1104. doi:10.1016/j.neurobiolaging.2014.10.029
Ofori E et al (2015c) Longitudinal changes in free-water within the substantia nigra of Parkinson’s disease. Brain 138:2322–2331. doi:10.1093/brain/awv136
Pasternak O et al (2012) Excessive extracellular volume reveals a neurodegenerative pattern in schizophrenia onset. J Neurosci 32:17365–17372. doi:10.1523/Jneurosci.2904-12.2012
Planetta PJ et al (2015a) Distinct functional and macrostructural brain changes in Parkinson’s disease and multiple system atrophy. Hum Brain Mapp 36:1165–1179. doi:10.1002/hbm.22694
Planetta PJ et al (2015b) Free-water imaging in Parkinson’s disease and atypical parkinsonism. Brain. doi:10.1093/brain/awv361
Prodoehl J et al (2013) Diffusion tensor imaging of Parkinson’s disease, atypical parkinsonism, and essential tremor. Mov Disord 28:1816–1822. doi:10.1002/mds.25491
Purves D, Augustine GJ, Fitzpatrick D, Hall WC, LaMantia AS, White LE (2012) Neuroscience, 5th edn. Sinauer Association Inc, Sunderland
Reetz K et al (2013) Genotype-specific patterns of atrophy progression are more sensitive than clinical decline in SCA1, SCA3, and SCA6. Brain 136:905–917. doi:10.1093/brain/aws369
Sato K, Ishigame K, Ying SH, Oishi K, Miller MI, Mori S (2015) Macro- and microstructural changes in patients with spinocerebellar ataxia type 6: assessment of phylogenetic subdivisions of the cerebellum and the brain stem. AJNR Am J Neuroradiol 36:84–90. doi:10.3174/ajnr.A4085
Schurz M, Wimmer H, Richlan F, Ludersdorfer P, Klackl J, Kronbichler M (2015) Resting-state and task-based functional brain connectivity in developmental dyslexia. Cereb Cortex 25:3502–3514. doi:10.1093/cercor/bhu184
Seidel K, Siswanto S, Brunt ERP, den Dunnen W, Korf HW, Rub U (2012) Brain pathology of spinocerebellar ataxias. Acta Neuropathol 124:1–21. doi:10.1007/s00401-012-1000-x
Shadmehr R, Krakauer JW (2008) A computational neuroanatomy for motor control. Exp Brain Res 185:359–381. doi:10.1007/s00221-008-1280-5
Solodkin A, Gomez CM (2012) Spinocerebellar ataxia type 6. Handb Clin Neurol 103:461–473. doi:10.1016/B978-0-444-51892-7.00029-2
Spraker MB, Prodoehl J, Corcos DM, Comella CL, Vaillancourt DE (2010) Basal ganglia hypoactivity during grip force in drug naive Parkinson’s disease. Hum Brain Mapp 31:1928–1941. doi:10.1002/hbm.20987
Stefanescu MR et al (2015) Structural and functional MRI abnormalities of cerebellar cortex and nuclei in SCA3, SCA6 and Friedreich’s ataxia. Brain 138:1182–1197. doi:10.1093/brain/awv064
Tuite PJ, Dagher A (2013) Magnetic resonance imaging in movement disorders: a guide for clinicians and scientists. Cambridge University, Cambridge
Tzourio-Mazoyer N et al (2002) Automated anatomical labeling of activations in SPM using a macroscopic anatomical parcellation of the MNI MRI single-subject brain. Neuroimage 15:273–289. doi:10.1006/nimg.2001.0978
Vaillancourt DE, Mayka MA, Corcos DM (2006) Intermittent visuomotor processing in the human cerebellum, parietal cortex, and premotor cortex. J Neurophysiol 95:922–931. doi:10.1152/jn.00718.2005
Wang Y et al (2011) Quantification of increased cellularity during inflammatory demyelination. Brain 134:3587–3598. doi:10.1093/brain/awr307
Watson TC, Becker N, Apps R, Jones MW (2014) Back to front: cerebellar connections and interactions with the prefrontal cortex. Front Syst Neurosci 8:4. doi:10.3389/fnsys.2014.00004
Acknowledgments
The authors would like to thank the participants and their families for their time and commitment to this research. This work was Supported by the National Institutes of Health (R01 NS075012, R01 NS058487, R21 NS093695, and T32 NS082168).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Electronic supplementary material
Below is the link to the electronic supplementary material.
429_2016_1263_MOESM1_ESM.tif
Supplementary material 1 Supplementary Data Fig. 1. Functional activity for control and SCA6 groups during a hand-grip force control. Data are thresholded at P < 0.005 (cluster size correction using the AFNI’s 3dClustSim, providing a corrected P < 0.05). Warm (yellow) colors indicate where BOLD (blood oxygen level-dependent) activity was positive. C: contralateral hemisphere, I: ipsilateral hemisphere (TIFF 1741 kb)
Rights and permissions
About this article
Cite this article
Kang, N., Christou, E.A., Burciu, R.G. et al. Sensory and motor cortex function contributes to symptom severity in spinocerebellar ataxia type 6. Brain Struct Funct 222, 1039–1052 (2017). https://doi.org/10.1007/s00429-016-1263-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00429-016-1263-4