Abstract
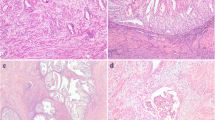
This study was aimed at determining whether high-grade endometrioid carcinomas (grade 3 International Federation of Gynecology and Obstetrics) might overlap, at least partially, non-endometrioid carcinomas (type II). To this end, a panel of clinical–pathological and immunohistochemical parameters was evaluated in three different populations: low-grade endometrioid carcinomas (LGECs; n = 57), high-grade endometrioid carcinomas (HGECs; n = 26), and non-endometrioid carcinomas (NECs; n = 30). Besides morphological appearance, HGECs appeared similar to LGECs in p53 immunostaining profile; features different from LGECs included a higher local aggressiveness, a higher invasion of lymph-vascular spaces, a lower expression of ERα and PR, and a higher proliferative index. HGECs were similar to NECs for local aggressiveness, invasion rate of lymph-vascular spaces, lymph node metastasis incidence, and proliferative index. HGECs, however, showed a lower rate of extra-nodal metastases, a lower incidence of p53 overexpression, and a higher positivity for ERα and PR. In conclusion, results from this study show that HGECs exhibit overlapping morphological and immunohistochemical features of both type I and type II endometrial carcinomas. Further research is needed to clarify the clinical value of this observation.



Similar content being viewed by others
References
Stewart BW, Kleihues P (eds) (2003) World cancer report. IARC, Lyon
Lax SF (2004) Molecular genetic pathways in various types of endometrial carcinoma: from a phenotypical to a molecular-based classification. Virchows Arch 444:213–223, Review
Bokhman JV (1983) Two pathogenetic types of endometrial carcinoma. Gynecol Oncol 15:10–17
Bansal N, Yendluri V, Wenham RM (2009) The molecular biology of endometrial cancers and the implications for pathogenesis, classification, and targeted therapies. Cancer Control 16:8–13, Review
Burton JL, Wells M (1998) Recent advances in the histopathology and molecular pathology of carcinoma of the endometrium. Histopathology 33:297–303, Review
Zorn KK, Bonome T, Gangi L et al (2005) Gene expression profiles of serous, endometrioid, and clear cell subtypes of ovarian and endometrial cancer. Clin Cancer Res 11:6422–6430
Bakkum-Gamez JN, Gonzalez-Bosquet J, Laack NN et al (2008) Current issues in the management of endometrial cancer. Mayo Clin Proc 83:97–112, Review
Amant F, Moerman P, Neven P et al (2005) Endometrial cancer. Lancet 366:491–505, Review
International Federation of Gynecology and Obstetrics (FIGO) (1999) Announcements. FIGO stages: 1998 revision. Gynecol Oncol 35:125–127
Matias-Guiu X (2009) Endometrial neoplasia. In: Nucci MR, Oliva E (eds) Gynecologic pathology. Churchill and Livingstone, London, pp 235–259
Alkushi A, Lim P, Coldman A, Huntsman D, Miller D, Gilks CB (2004) Interpretation of p53 immunoreactivity in endometrial carcinoma: establishing a clinically relevant cut-off level. Int J Gynecol Pathol 23:129–137
Jeczen R, Skomra D, Cybulski M, Schneider-Stock R, Szewczuk W, Roessner A, Rechberger T, Semczuk A (2007) P53/MDM2 overexpression in metastatic endometrial cancer: correlation with clinicopathological features and patient outcome. Clin Exp Metastasis 24:503–511
Mendivil A, Schuler KM, Gehrig PA (2009) Non-endometrioid adenocarcinoma of the uterine corpus: a review of selected histological subtypes. Cancer Control 16:46–52
Ronnet BM, Zaino RJ, Ellenson LH et al (2002) Endometrial carcinoma. In: Kurman RJ (ed) Blaustein’s pathology of the female genital tract. Springer, New York, pp 501–559
Doll A, Abal M, Rigau M et al (2008) Novel molecular profiles of endometrial cancer-new light through old windows. J Steroid Biochem Mol Biol 108:221–229
Sherman ME, Bur ME, Kurman RJ (1995) p53 in endometrial cancer and its putative precursors: evidence for diverse pathways of tumorigenesis. Hum Pathol 26:1268–1274
Rose PG (1996) Endometrial carcinoma. N Engl J Med 335:640–649
Salvesen HB, Iversen OE, Akslen LA (1999) Prognostic significance of angiogenesis and Ki-67, p53, and p21 expression: a population-based endometrial carcinoma study. J Clin Oncol 17:1382–1390
Carter J, Pather S (2006) An overview of uterine cancer and its management. Expert Rev Anticancer Ther 6:33–42, Review
Lax SF, Pizer ES, Ronnett BM, Kurman RJ (1998) Comparison of estrogen and progesterone receptor, Ki-67, and p53 immunoreactivity in uterine endometrioid carcinoma and endometrioid carcinoma with squamous, mucinous, secretory, and ciliated cell differentiation. Hum Pathol 29:924–931
Oreskovic S, Babic D, Kalafatic D et al (2004) A significance of immunohistochemical determination of steroid receptors, cell proliferation factor Ki-67 and protein p53 in endometrial carcinoma. Gynecol Oncol 93:34–40
Barrena Medel NI, Bansal S, Miller DS et al (2009) Pharmacotherapy of endometrial cancer. Expert Opin Pharmacother 10:1939–1951, Review
Chaudhry P, Asselin E (2009) Resistance to chemotherapy and hormone therapy in endometrial cancer. Endocr Relat Cancer 16:363–380
Clarke BA, Gilks CB (2010) Endometrial carcinoma: controversies in histopathological assessment of grade and tumour cell type. J Clin Pathol 63:410–415
Conflict of interest statement
We declare that we have no conflict of interest.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Zannoni, G.F., Vellone, V.G., Arena, V. et al. Does high-grade endometrioid carcinoma (grade 3 FIGO) belong to type I or type II endometrial cancer? A clinical–pathological and immunohistochemical study. Virchows Arch 457, 27–34 (2010). https://doi.org/10.1007/s00428-010-0939-z
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00428-010-0939-z