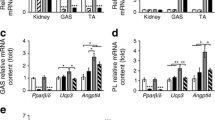
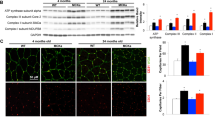
Abstract
Lifestyle-related diseases are rapidly increasing at least in part due to less physical activity. The health beneficial effects of regular physical activity include metabolic adaptations in skeletal muscle, which are thought to be elicited by cumulative effects of transient gene responses to each single exercise, but how is this regulated? A potential candidate in this is the transcriptional coactivator peroxisome proliferator-activated receptor-γ coactivator (PGC)-1α, which has been identified as a master regulator of mitochondrial biogenesis, but also been shown to regulate proteins involved in angiogenesis and the anti-oxidant defence as well as to affect expression of inflammatory markers. Exercise increases PGC-1α transcription and potentially PGC-1α activity through post-translational modifications, and concomitant PGC-1α-mediated gene regulation is suggested to be an underlying mechanism for adaptations in skeletal muscle, when exercise is repeated. The current review presents some of the key findings in PGC-1α-mediated regulation of metabolically related, anti-oxidant and inflammatory proteins in skeletal muscle in the basal state and in response to exercise training, and describes functional significance of PGC-1α-mediated effects in skeletal muscle. In addition, regulation of PGC-1α expression and activity in skeletal muscle is described. The impact of changes in PGC-1α expression in mouse skeletal muscle and the ability of PGC-1α to regulate multiple pathways and functions underline the potential importance of PGC-1α in skeletal muscle adaptations in humans. The absence of exercise-induced PGC-1α-mediated gene regulation during a physical inactive lifestyle is suggested to lead to reduced oxidative capacity of skeletal muscle and concomitant impaired metabolism.



Similar content being viewed by others
References
Adhihetty PJ, Uguccioni G, Leick L et al (2009) The role of PGC-1{alpha} on mitochondrial function and apoptotic susceptibility in muscle. Am J Physiol Cell Physiol 297:C217–C225
Arany Z, Foo SY, Ma Y et al (2008) HIF-independent regulation of VEGF and angiogenesis by the transcriptional coactivator PGC-1alpha. Nature 451:1008–1012
Arany Z, He L, Lin J (2005) Transcriptional coactivator PGC-1α controls the energy state and contractile function of cardiac muscle. Cell Metabol 1:259–271
Baar K, Wende AR, Jones TE et al (2002) Adaptations of skeletal muscle to exercise: rapid increase in the transcriptional coactivator PGC-1. FASEB J 16:1879–1886
Barres R, Osler ME, Rune A et al (2009) Non-CpG methylation of the PGC-1a promoter through DNMT3B controls mitochondrial density. Cell Metabol 10:189–198
Booth FW, Baldwin KM (1996) Muscle plasticity: energy demand and supply processes. In: Rowell LB, Shepherd JT (eds) The handbook of physiology. Exercise: regulation and integration of multiple systems. American Physiological Society, Bethesda, pp 1075–1123
Calvo JA, Daniels TG, Wang X et al (2008) Muscle-specific expression of PPARgamma coactivator-1alpha improves exercise performance and increases peak oxygen uptake. J Appl Physiol 104:1304–1312
Canto C, Gerhart-Hines Z, Feige JN et al (2009) AMPK regulates energy expenditure by modulating NAD + metabolism and SIRT1 activity. Nature 458:1056–1062
Chinsomboon J, Ruas J, Gupta R et al (2009) The transcriptional coactivator PGC-1alpha mediates exercise-induced angiogenesis in skeletal muscle. PNAS 106:21401–21406
Conley KE, Jubrias SA, Esselman PC (2000) Oxidative capacity and ageing in human muscle. J Physiol 526:203–210
Constantin-Teodosiu D, Baker DJ, Constantin D, Greenhaff PL (2009) PPARdelta agonism inhibits skeletal muscle PDC activity, mitochondrial ATP production and force generation during prolonged contraction. J Physiol 587:231–239
De FE, Alvarez G, Berria R et al (2008) Insulin-resistant muscle is exercise resistant: evidence for reduced response of nuclear-encoded mitochondrial genes to exercise. Am J Physiol 294:E607–E614
Dominy JE Jr, Lee Y, Gerhart-Hines Z, Puigserver P (2010) Nutrient-dependent regulation of PGC-1alpha's acetylation state and metabolic function through the enzymatic activities of Sirt1/GCN5. Biochem Biophys doi:10.1016/j.bbapap.2009.11.023
Geng T, Li P, Okutsu M et al (2010) PGC-1α plays a functional role in exercise-induced mitochondrial biogenesis and angiogenesis but not fiber-type transformation in mouse skeletal muscle. Am J Physiol Cell Physiol 298:572–579
Gerhart-Hines Z, Rodgers JT, Bare O et al (2007) Metabolic control of mitochondrial function and fatty acid oxidation through PGC-1alpha/SIRT1. EMBO J 26:1902–1912
Handschin C, Chin S, Li P et al (2007) Skeletal muscle fiber-type switching, exercise intolerance, and myopathy in PGC-1alpha muscle-specific knock-out animals. J Biol Chem 282:30014–30021
Handschin C, Choi CS, Chin S et al (2007) Abnormal glucose homeostasis in skeletal muscle-specific PGC-1α knockout mice reveals skeletal muscle-pancreatic βcell crosstalk. J Clin Invest 117:3463–3474
Handschin C, Rhee J, Lin J et al (2003) An autoregulatory loop controls peroxisome proliferator-activated receptor gamma coactivator 1alpha expression in muscle. PNAS 100:7111–7116
Handschin C, Spiegelman BM (2008) The role of exercise and PGC1α in inflammation and chronic disease. Nature 454:463–469
Irrcher I, Adhihetty JP, Sheehan T et al (2003) PPARγ coactivator-1α expression during thyroid hormone- and contractile activity-induced mitochondrial adaptations. Am J Physiol 284:C1669–C1677
Jager S, Handschin C, St-Pierre J, Spiegelman BM (2007) AMP-activated protein kinase (AMPK) action in skeletal muscle via direct phosphorylation of PGC-1{alpha}. PNAS 104:12017–12022
Jorgensen SB, Treebak JT, Viollet B et al (2007) Role of AMPKalpha2 in basal, training-, and AICAR-induced GLUT4, hexokinase II, and mitochondrial protein expression in mouse muscle. Am J Physiol 292:E331–E339
Jorgensen SB, Wojtaszewski JFP, Viollet B et al (2005) Effects of alpha-AMPK knockout on exercise-induced gene activation in mouse skeletal muscle. FASEB J 19:1146–1148
Kelly DP, Scarpulla RC (2007) Transcriptional regulatory circuits controlling mitochondrial biogenesis and function. Genes Dev 18:357–368
Kiilerich K, Adser H, Jakobsen AH et al (2009) Role of PGC-1α in PDH regulation in mouse skeletal muscle. Biochem Exerc (in press)
Koval JA, DeFronzo RA, O'Doherty RM et al (1998) Regulation of hexokinase II activity and expression in human muscle by moderate exercise. Am J Physiol 274:E304–E308
Kraniou Y, Cameron-Smith D, Misso M et al (2000) Effects of exercise on GLUT-4 and glycogenin gene expression in human skeletal muscle. J Appl Physiol 88:794–796
Kusuhara K, Madsen K, Jensen L et al (2007) Calcium signalling in the regulation of PGC-1alpha, PDK4 and HKII mRNA expression. Biol Chem 388:481–488
Leick L, Hellsten Y, Fentz J et al (2009) PGC-1{alpha} mediates exercise-induced skeletal muscle VEGF expression in mice. Am J Physiol 297:E92–E103
Leick L, Lyngby SS, Wojtaszewski JFP, Pilegaard H (2010) PGC-1α is required for training-induced prevention of age-associated decline in mitochondrial enzymes in mouse skeletal muscle. Exp Gerontol 45:336–342
Leick L, Plomgaard P, Wojtaszewski JFP, Pilegaard H (2009) Endurance exercise induces mRNA of oxidative proteins in human skeletal muscle late in recovery. Scan J Sports Med (in press)
Leick L, Wojtaszewski JF, Johansen ST et al (2008) PGC-1alpha is not mandatory for exercise- and training-induced adaptive gene responses in mouse skeletal muscle. Am J Physiol 294:E463–E474
Leone TC, Lehman JJ, Finck BN et al (2005) PGC-1α deficiency causes multi-system energy metabolic dearrangements: muscle dysfunction, abnormal weight control and hepatic steatosis. PLoS Biol 3:e101
Lerin C, Rodgers JT, Kalume DE et al (2006) GCN5 acetyltransferase complex controls glucose metabolism through transcriptional repression of PGC-1alpha. Cell Metabol 3:429–438
Lin J, Handschin C, Spiegelman BM (2005) Metabolic control through the PGC-1 family of transcription coactivators. Cell Metabol 1:361–370
Lin J, Wu H, Tarr PT et al (2002) Transcriptional co-activator PGC-1 alpha drives the formation of slow-twitch muscle fibres. Nature 418:797–801
Lin J, Wu PH, Tarr PT et al (2004) Defects in adaptive energy metabolism with CNS-linked hyperactivity in PGC-1alpha null mice. Cell 119:121–135
Ling C, Poulsen P, Carlsson E et al (2004) Multiple environmental and genetic factors influence skeletal muscle PGC-1alpha and PGC-1beta gene expression in twins. J Clin Invest 114:1518–1526
Michael LF, Wu Z, Cheatham RB et al (2001) Restoration of insulin-sensitive glucose transporter (GLUT4) gene expression in muscle cells by the transcriptional coactivator PGC-1. PNAS 98:3820–3825
Neufer PD, Dohm GL (1993) Exercise induces a transient increase in transcription of the GLUT-4 gene in skeletal muscle. Am J Physiol 265:C1597–C1603
O'Doherty RM, Bracy DP, Granner DK, Wasserman DH (1996) Transcription of the rat skeletal muscle hexokinase II gene is increased by acute exercise. J Appl Physiol 81:789–793
Ojuka EO, Jones TE, Han DH et al (2003) Raising Ca2+ in L6 myotubes mimics effects of exercise on mitochondrial biogenesis in muscle. FASEB J 17:675–681
Pilegaard H, Ordway GA, Saltin B, Neufer PD (2000) Transcriptional regulation of gene expression in human skeletal muscle during recovery from exercise. Am J Physiol 279:E806–E814
Pilegaard H, Osada T, Andersen L et al (2005) Influence of substrate availability on transcriptional regulation of metabolic genes in human skeletal muscle. Metab 54:1048–1055
Pilegaard H, Saltin B, Neufer PD (2003) Exercise induces transient transcriptional activation of the PGC-1alpha gene in human skeletal muscle. J Physiol 546:851–858
Puigserver P, Rhee J, Lin J et al (2001) Cytokine stimulation of energy expenditure through p38 MAP kinase activation of PPARgamma coactivator-1. Mol Cell 8:971–982
Puigserver P, Wu Z, Park CW, Graves R, Wright M, Spiegelman BM (1998) A cold-inducible coactivator of nuclear receptors linked to adaptive thermogenesis. Cell 92(6):829–839
Röckl KSC, Witczak CA, Goodyear LJ (2008) Signaling mechanisms in skeletal muscle: acute responses and chronic adaptations to exercise. Life 60:145–153
Rodgers JT, Lerin C, Gerhart-Hines Z, Puigserver P (2008) Metabolic adaptations through the PGC-1α and SIRT1 pathways. FEBS Lett 582:46–53
Rodgers JT, Lerin C, Haas W et al (2005) Nutrient control of glucose homeostasis through a complex of PGC-1alpha and SIRT1. Nature 434:113–118
Seale P, Kajumuras S, Yang W et al (2007) Transcriptional control of brown fat determination by PRDM16. Cell Metabol 6:38–54
Seip RL, Mair K, Cole TG, Semenkovich CF (1997) Induction of human skeletal muscle lipoprotein lipase gene expression by short-term exercise is transient. Am J Physiol 272:E255–E261
Silveira LR, Pilegaard H, Kusuhara K et al (2006) The effect of reactive oxygen species and antioxidants on basal and contraction-induced gene expression of PGC-1α, UCP3 and HKII in primary rat skeletal muscle cells. Biochem Biophys Acta 1763:969–976
St-Pierre J, Drori S, Uldry M et al (2006) Suppression of reactive oxygen species and neurodegeneration by the PGC-1 transcriptional coactivators. Cell 127:397–408
St-Pierre J, Lin J, Krauss S et al (2003) Bioenergetic analysis of peroxisome proliferator-activated receptor gamma coactivators 1alpha and 1beta (PGC-1alpha and PGC-1beta) in muscle cells. J Biol Chem 278:26597–26603
Sugden MC, Holness MJ (2003) Recent advances in mechanisms regulating glucose oxidation at the level of the pyruvate dehydrogenase complex by PDK's. Am J Physiol 284:E855–E862
Suwa M, Nakano H, Radak Z, Kumagai S (2008) Endurance exercise increases the SIRT1 and peroxisome proliferator-activated receptor gamma coactivator-1alpha protein expression in rat skeletal muscle. Metab 57:986–998
Terada S, Goto M, Kato M et al (2002) Effects of low-intensity prolonged exercise on PGC-1 mRNA expression in rat epitrochlearis muscle. Biochem Biophys Res Commun 296:350–354
Vega RB, Huss JM, Kelly DP (2000) The coactivator PGC-1 cooperates with peroxisome proliferator-activated receptor alpha in transcriptional control of nuclear genes encoding mitochondrial fatty acid oxidation enzymes. Mol Cell Biol 20:1868–1876
Wallberg AE, Yamamurra S, Malik D et al (2003) Coordination of p300-mediated chromatin remodelling and TRAP/mediator function through coactivator PGC-1α. Mol Cell 12:1137–1149
Wellen KE, Hotamisligil GS (2005) Inflammation, stress, and diabetes. J Clin Invest 115:1111–1119
Wende AR, Huss JM, Schaeffer PJ et al (2005) PGC-1α coactivates PDK4 gene expression via the orphan nuclear receptor ERRα: a mechanism for transcriptional control of muscle glucose metabolism. Mol Cell Biol 25:10684–10694
Wende AR, Schaeffer PJ, Parker GJ et al (2007) A role for the transcriptional coactivator PGC-1α in muscle refueling. J Biol Chem 50:36642–36651
Wenz T, Rossi S, Rotundo RL et al (2009) Increased muscle PGC-1alpha expression protects from sarcopenia and metabolic disease during aging. PNAS 106:20405–20410
Williams RS, Neufer PD (1996) Regulation of gene expression in skeletal muscle by contractile activity. In: Rowell LB, Shepherd JT (eds) The handbook of physiology. Exercise: regulation and integration of multiple systems. American Physiological Society, Bethesda, pp 1124–1150
Wright DC, Geiger PC, Han DH et al (2007) Calcium induces increases in peroxisome proliferator-activated receptor gamma coactivator-1alpha and mitochondrial biogenesis by a pathway leading to p38 mitogen-activated protein kinase activation. J Biol Chem 282:18793–18799
Wu H, Kanatous SB, Thurmond FA et al (2002) Regulation of mitochondrial biogenesis in skeletal muscle by CaMK. Science 296:349–352
Wu Z, Puigserver P, Andersson U et al (1999) Mechanisms controlling mitochondrial biogenesis and respiration through the thermogenic coactivator PGC-1. Cell 98:115–124
Acknowledgements
The PGC-1α-related work in the authors' laboratory is supported by the Lundbeck Foundation, the Novo Nordisk Foundation and the Danish Medical Research Council. The Centre of Inflammation and Metabolism is supported by The Danish National Research Foundation (grant 02-512-555). CIM is part of the UNIK Project: Food, Fitness and Pharma for Health and Disease, supported by the Danish Ministry of Science, Technology and Innovation. The Copenhagen Muscle Research Centre is supported by a grant from the Capital Region of Denmark.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Olesen, J., Kiilerich, K. & Pilegaard, H. PGC-1α-mediated adaptations in skeletal muscle. Pflugers Arch - Eur J Physiol 460, 153–162 (2010). https://doi.org/10.1007/s00424-010-0834-0
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00424-010-0834-0