Abstract
Background
Since the introduction of cold corneoscleral segment storage prior to keratoplasty there have been continuous efforts to ameliorate the preservation media in order to better maintain the quality of the corneal epi- and endothelium. Recent studies have shown that basic fibroblast growth factor (FGF-2) preserves the viability of, for example, retinal ganglion cells and pigment epithelium cells. Therefore, we investigated the effect of different concentrations of FGF-2 added to a modified Optisol storage medium on endothelial damage after corneal storage at 4°C.
Methods
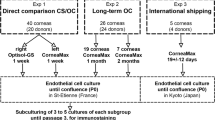
Bovine corneas were stored at 4°C for 14 days and for another 24 h at 34°C. Various FGF-2 concentrations (4, 20 and 40 ng/ml) were added to the medium either at day (D) 1, D14, or both D1 and D14. Quantitative evaluation of corneal damage after 14+1 days of storage was conducted by means of the Janus green photometry assay. Histological and ultrastructural investigations of the preserved endothelium were also performed. Bovine cell culture experiments using the TUNEL assay aimed to elucidate the role of FGF-2 on prevention of endothelial apoptosis.
Results
The mean endothelial damage in control corneas increased from 4.9±1.8% (fresh corneas) to 13.4±2.4% after 14+1 days of storage. FGF-2 at 20 ng/ml or 40 ng/ml added at any of the indicated time points significantly reduced the overall endothelial damage by 5.1–7.3%, corresponding to 38–54% less endothelial damage than in control corneas (P<0.001). Light- and electron microscopic investigations confirmed this protective effect of FGF-2 on corneal endothelial cells. The TUNEL assay revealed a true anti-apoptotic effect of FGF-2 on endothelial cells in culture.
Conclusion
Our study clearly demonstrates the effectiveness of FGF-2 to enhance cell survival of the corneal endothelium after storage at 4°C. A clinical interest could be seen in the potential future application of FGF-2 as an adjuvant to corneal preservation media in order to better maintain endothelial viability during corneal storage.






Similar content being viewed by others
References
Akimoto M, Miyatake S, Kogishi J, et al (1999) Adenovirally expressed basic fibroblast growth factor rescues photoreceptor cells in RCS rats. Invest Ophthalmol Vis Sci 40:273–279
Anderson KJ, Dam D, Lee S, Cotman CW (1988) Basic fibroblast growth factor prevents death of lesioned cholinergic neurons in vivo. Nature 332:360–361
Araki T, Taniwaki T, Becerra SP, Chader GJ, Schwartz JP (1998) Pigment epithelium-derived factor (PEDF) differentially protects immature but not mature cerebellar granule cells against apoptotic cell death. J Neurosci Res 53:7–15
Bourne WM (1986) Endothelial cell survival in transplanted human corneas preserved at 4°C in 2.5% chondroitin sulfate for one to 13 days. Am J Ophthalmol 102:382–386
Briat B, David T, Serdarevic O, Gordon J, Renard G, Pouliquen Y (1994) Comparative study on the preservation of human corneal endothelium during organ culture storage with Optisol and Optisol plus epidermal growth factor (EGF). Invest Ophthalmol Vis Sci [Suppl] 35:1860
Esch F, Ueno N, Baird A, Hill F, Demoroy L, Ling N, Gospodarowicz D, Guillemin R (1985) Primary structure of bovine brain acidic FGF. Biochem Biophys Res Commun 133:554–562
Factorovich EG, Steinberg RH, Yasumura D, Matthes MT, LaVail MM (1990) Photoreceptor degeneration in inherited retinal dystrophy delayed by bFGF. Nature 347:83–86
Giguère L, Cheng J, Gospodarowicz D (1982) Factors involved in the control of proliferation of bovine corneal endothelial cells maintained in serum-free medium. J Cell Physiol 110:72–80
Gospodarowicz D, Mescher A, Birdwell CR (1977) Stimulation of corneal endothelial cell proliferation in vitro by fibroblast and epidermal growth factor. Exp Eye Res 25:75–89
Gospodarowicz D, Neufeld G, Schweigerer L (1987) Fibroblast growth factor: structural and biological properties. J Cell Physiol (Suppl) 5:15–26
Guillonneau X, Bryckaert M, Launay-Longo C, Courtois Y, Mascarelli F (1998) Endogenous FGF1-induced activation and synthesis of extracellular signal-regulated kinase 2 reduce cell apoptosis in retinal pigmented epithelial cells. J Biol Chem 273:22367–22373
Hartmann C, Rieck P (1989) A new test for endothelial viability: the Janus green photometry technique. Arch Ophthalmol 107:1511–1515
Hoppenreijs VP, Pels E, Vrensen GF, Treffers WF (1994) Basic fibroblast growth factor stimulates corneal endothelial cell growth and endothelial wound healing of human corneas. Invest Ophthalmol Vis Sci 35:931–44
Houssaint E, Blanquet P, Champion-Arnaud P, Gesnel MC, Torriglia A, Courtois Y, Breathnach P (1990) Related fibroblast growth factor receptor genes exist in the human genome. Proc Natl Acad Sci 87:8180–8184
Kaufman HE, Beuerman RW, Steinemann TL, et al. (1991) Optisol corneal storage medium. Arch Ophthalmol 109:864–868
Komuro A, Hodge DO, Gores GJ, Bourne WM (1999) Cell death during corneal storage at 4°C. Invest Ophthalmol Vis Sci 40:2827–2832
Lass JH, Bourne WM, Musch DC, Sugar A, Gordon JF, Reinhart WJ, Meyer RF, Patel DI, Bruner WE, Cano DB, et al (1992) A randomized, double masked clinical trial of Optisol vs DexSol corneal storage media. Arch Ophthalmol 110:1404–1408
Lass JH, Musch DC, Gordon JF, Laing RA, The Corneal Preservation Study Group (1994) Epidermal growth factor and insulin in corneal preservation. Ophthalmology 101:352–359
Lee PL, Johnson DE, Cousens LS, Fried VA, Williams LT (1989) Purification and complementary DNA cloning of a receptor for basic fibroblast growth factor. Science 245:57–60
Lindstrom RL, Kaufman HE, Skelnik DL, et al (1992) Optisol corneal storage medium. Am J Ophthalmol 114:345–356
McCarey BE, Kaufman HE (1974) Improved corneal storage. Invest Ophthalmol Vis Sci 13:165–173
Means TL, Geroski DH, Hadley A, Lynn MJ, Edelhauser HF (1995) Viability of human corneal endothelium following Optisol-GS storage. Arch Ophthalmol 113:805–809
Naor J, Slomovic AR, Chipman M, Rootman DS (2002) A randomized, double-masked clinical trial of Optisol-GS vs Chen Medium for human corneal storage. Arch Ophthalmol 120:1280–1285
Pharmakakis N, Hartmann C, Hilgers RD, Bergmann L, Koliopoulos I (1995) Correlation study of two methods for evaluating corneal endothelial damage in vitro: Janus green photometry technique versus cell counting. Ophthalmic Res 27:67–73
Plouet J, Courty J, Olivié M, Courtois Y, Barritault D (1983) A highly reliable and sensitive assay for the purification of cellular growth factor. Cell Mol Biol 30:105–110
Renaud F, Desset S, Oliver L, Giminez-Galego G, Van Oberghen E, Courtois Y, Laurent M (1996) The neurotrophic activity of FGF-1 depends on FGF-1 expression and is independant of the MAP kinase cascade pathway. J Biol Chem 271:2801–2811
Rieck P, Hartmann C, Pouliquen Y, Courtois Y (1992) Recombinant human bFGF stimulates corneal endothelial wound healing in rabbits. Curr Eye Res 19:153–160
Rieck P, Oliver L, Engelmann K, Fuhrmann G, Hartmann C, Courtois Y (1995) Role of exogenous / endogenous FGF-2 and TGFß on human corneal endothelial cells proliferation in vitro. Exp Cell Res 220:36–46
Rieck P, Cholidis S, Hartmann C (2001) Intracellular signaling pathway of corneal endothelial cell migration during wound healing in vitro. Exp Eye Res 73:723–733
Rogelj S, Klagsbrun M, Atzmon R, Kurokawa M, Halmovitz A, Fuks Z, Vlodaysky I (1989) Basic fibroblast growth factor is an extracellular matrix component required for supporting the proliferation of vascular endothelial cells and the differentiation of PC 12 cells. J Cell Biol 109:823–831
Sabatier P, Rieck P, Daumer ML, Courtois Y, Pouliquen Y, Hartmann C (1996) Effects of human recombinant basic fibroblast growth factor on endothelial wound healing in organ culture of human cornea. J Fr Ophtalmol 19:200–207
Saksela O, Moscatelli D, Sommer A, Rifkin DB (1988) Endothelial cell-derived heparan sulfate binds basic fibroblast growth factor and protects it from proteolytic degradation. J Cell Biol 107:743–751
Schilling-Schön A, Pleyer U, Hartmann C, Rieck P (2000) The role of endogenous growth factors to support endothelial migration after wounding in vitro. Exp Eye Res 71:583–589
Sievers J, Hausmann B, Unsicker K, Berry M (1987) Fibroblast growth factors promote the survival of adult rat retinal ganglion cells after transection of the optic nerve. Neurosci Lett 76:157–162
Stainer GA, Brightbill FS, Calkins B (1981) A comparison of corneal storage in moist chamber and McCarey-Kaufman medium in human keratoplasty. Ophthalmology 88:46–49
Zhang C, Takahashi K, Lam TT, Tso MOM (1994) Effects of basic fibroblast growth factor in retinal ischemia. Invest Ophthalmol Vis Sci 35:3163–3168
Acknowledgement
Supported by a research grant (Ri 568/3-2) to P.R. from the Deutsche Forschungsgemeinschaft (DFG), Bonn, Germany.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Rieck, P.W., von Stockhausen, R.M., Metzner, S. et al. Fibroblast growth factor-2 protects endothelial cells from damage after corneal storage at 4°C. Graefe's Arch Clin Exp Ophthalmol 241, 757–764 (2003). https://doi.org/10.1007/s00417-003-0687-8
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00417-003-0687-8