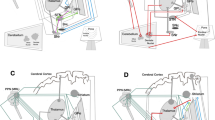
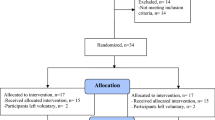
Abstract
Galvanic vestibular stimulation (GVS) has increasingly been used to stimulate the vestibular system in health and disease. While perceptible supra-threshold GVS destabilizes postural control in healthy control (HC) subjects, imperceptible ‘noisy’ GVS (nGVS) is reported to improve postural control in patients with bilateral vestibulopathy (BV) and therapeutic devices using nGVS are currently under development. We questioned (1) whether perceptible GVS destabilizes postural control of BV patients, expecting any effect to be smaller than in healthy subjects due to the patients’ vestibulopathy, and (2) whether imperceptible nGVS improves postural control in comparison to an active sham stimulus in context-dependent conditions, hypothesizing that it fades off once postural control becomes more challenging with respect to its sensory (standing on foam) or cognitive (dual task) complexity. We tested postural responses of 30 BV patients to bimastoidal perceptible (lowGVS, highGVS) or imperceptible (nGVS, sham, noGVS) GVS in comparison to 24 age-matched HC. Perceptible GVS intensities were applied according to the participants’ individual motion perception thresholds. Postural sway speed (PSS) was analyzed in a 4-factorial experimental design with the factors group (BV, HC), vision (eyes open/closed), condition (baseline, proprioception, cognition) and stimulation (noGVS, sham, nGVS, lowGVS, highGVS). With eyes open (EO), there were no group-related PSS differences in the baseline and cognition condition in response to either stimulations. With EO on foam and with eye closed (EC) in all conditions, patients showed larger PSS than HC, irrespective of the stimulation type. PSS differed with GVS intensities within each group but not between the groups. PSS under nGVS on EC was only smaller in patients when compared to perceptible GVS, but it was not different from noGVS or sham stimulation. Moreover, this nGVS effect was only found in the baseline but not in the more challenging dual task and foam condition. Almost half of the patients showed higher individual thresholds of motion perception of GVS compared to HC. Interestingly, this high-threshold subgroup showed significantly larger PSS with EC as compared to HC and the low-threshold patient subgroup, although both patient subgroups did not differ in vestibular parameters. We conclude, first, that perceptible GVS is able to destabilize BV patients similarly to HC subjects, suggesting sufficient vestibular afferent processing of GVS during vestibulo-spinal postural control. Second, the effect of the hitherto observed improved postural control by nGVS appears to be small during more demanding postural control conditions (foam, cognitive distraction) that are closer to the patients’ everyday life, when active sham stimuli are used as control stimuli. These findings underline the meaning of active control conditions when the efficacy of nGVS is tested, e.g. in portable GVS devices in the attempt to improve postural control in BV patients. However, differential GVS effects on vestibulo-perceptional and vestibulo-spinal thresholds should be taken into account. Finally, our data suggest that individual motion perception thresholds for GVS could potentially serve as a predictor of postural control safety and falling risk in BV.




Similar content being viewed by others
Abbreviations
- BV:
-
Bilateral vestibulopathy
- HC:
-
Healthy control
- VOR:
-
Vestibulo-ocular reflex
- VSS:
-
Vertigo symptom scale
- DHI:
-
Dizziness handicap score
- CVS:
-
Clinical vestibular score
- SVV:
-
Subjective visual vertical
- LTG:
-
Low-threshold group
- HTG:
-
High-threshold group
- EO:
-
Eyes open
- EC:
-
Eyes closed
- GVS:
-
Galvanic vestibular stimulation
- nGVS:
-
Noisy GVS
References
Asslander L, Peterka RJ (2016) Sensory reweighting dynamics following removal and addition of visual and proprioceptive cues. J Neurophysiol 116:272–285
Bense S, Stephan T, Yousry TA, Brandt T, Dieterich M (2001) Multisensory cortical signal increases and decreases during vestibular galvanic stimulation (fMRI). J Neurophysiol 85:886–899
Brandt T, Dieterich M (2019) 'Excess anxiety' and 'less anxiety': both depend on vestibular function. Curr Opin Neurol 33:136–141
Cai J, Lee S, Ba F, Garg S, Kim LJ, Liu A, Kim D, Wang ZJ, McKeown MJ (2018) Galvanic vestibular stimulation (GVS) augments deficient pedunculopontine nucleus (PPN) connectivity in mild Parkinson's disease: fMRI effects of different stimuli. Front Neurosci 12:101
Cathers I, Day BL, Fitzpatrick RC (2005) Otolith and canal reflexes in human standing. J Physiol 563:229–234
Collins JJ, Chow CC, Imhoff TT (1995) Stochastic resonance without tuning. Nature 376:236–238
Cutfield NJ, Scott G, Waldman AD, Sharp DJ, Bronstein AM (2014) Visual and proprioceptive interaction in patients with bilateral vestibular loss. Neuroimage Clin 4:274–282
Cyran CA, Boegle R, Stephan T, Dieterich M, Glasauer S (2016) Age-related decline in functional connectivity of the vestibular cortical network. Brain Struct Funct 221:1443–1463
Dlugaiczyk J, Gensberger KD, Straka H (2019) Galvanic vestibular stimulation: from basic concepts to clinical applications. J Neurophysiol 121:2237–2255
Donker SF, Roerdink M, Greven AJ, Beek PJ (2007) Regularity of center-of-pressure trajectories depends on the amount of attention invested in postural control. Exp Brain Res 181:1–11
Faul F, Erdfelder E, Lang AG, Buchner A (2007) G*Power 3: a flexible statistical power analysis program for the social, behavioral, and biomedical sciences. Behav Res Methods 39:175–191
Fitzpatrick RC, Day BL (2004) Probing the human vestibular system with galvanic stimulation. J Appl Physiol (1985) 96:2301–2316
Fitzpatrick RC, Marsden J, Lord SR, Day BL (2002) Galvanic vestibular stimulation evokes sensations of body rotation. NeuroReport 13:2379–2383
Fitzpatrick RC, Wardman DL, Taylor JL (1999) Effects of galvanic vestibular stimulation during human walking. J Physiol 517(Pt 3):931–939
Fujimoto C, Egami N, Kawahara T, Uemura Y, Yamamoto Y, Yamasoba T, Iwasaki S (2018) Noisy galvanic vestibular stimulation sustainably improves posture in bilateral vestibulopathy. Front Neurol 9:900
Fujimoto C, Egami N, Kinoshita M, Sugasawa K, Yamasoba T, Iwasaki S (2014) Postural stability in vestibular neuritis: age, disease duration, and residual vestibular function. Laryngoscope 124:974–979
Fujimoto C, Murofushi T, Chihara Y, Ushio M, Suzuki M, Yamaguchi T, Yamasoba T, Iwasaki S (2013) Effect of severity of vestibular dysfunction on postural instability in idiopathic bilateral vestibulopathy. Acta Otolaryngol 133:454–461
Fujimoto C, Yagi M, Murofushi T (2019) Recent advances in idiopathic bilateral vestibulopathy: a literature review. Orphanet J Rare Dis 14:202
Fujimoto C, Yamamoto Y, Kamogashira T, Kinoshita M, Egami N, Uemura Y, Togo F, Yamasoba T, Iwasaki S (2016) Noisy galvanic vestibular stimulation induces a sustained improvement in body balance in elderly adults. Sci Rep 6:37575
Ghofrani M, Olyaei G, Talebian S, Bagheri H, Malmir K (2017) Test–retest reliability of linear and nonlinear measures of postural stability during visual deprivation in healthy subjects. J Phys Ther Sci 29:1766–1771
Goel R, Kofman I, Jeevarajan J, De Dios Y, Cohen HS, Bloomberg JJ, Mulavara AP (2015) Using low levels of stochastic vestibular stimulation to improve balance function. PLoS ONE 10:e0136335
Goel R, Rosenberg MJ, Cohen HS, Bloomberg JJ, Mulavara AP (2019) Calibrating balance perturbation using electrical stimulation of the vestibular system. J Neurosci Methods 311:193–199
Gottlich M, Jandl NM, Wojak JF, Sprenger A, von der Gablentz J, Munte TF, Kramer UM, Helmchen C (2014) Altered resting-state functional connectivity in patients with chronic bilateral vestibular failure. Neuroimage Clin 4:488–499
Guerraz M, Day BL (2005) Expectation and the vestibular control of balance. J Cogn Neurosci 17:463–469
Haburcakova C, Lewis RF, Merfeld DM (2012) Frequency dependence of vestibuloocular reflex thresholds. J Neurophysiol 107:973–983
Helmchen C, Kirchhoff JB, Gottlich M, Sprenger A (2017) Postural ataxia in cerebellar downbeat nystagmus: its relation to visual, proprioceptive and vestibular signals and cerebellar atrophy. PLoS ONE 12:e0168808
Helmchen C, Knauss J, Trillenberg P, Frendl A, Sprenger A (2017) Role of the patient's history of vestibular symptoms in the clinical evaluation of the Bedside Head-Impulse Test. Front Neurol 8:51
Helmchen C, Rother M, Spliethoff P, Sprenger A (2019) Increased brain responsivity to galvanic vestibular stimulation in bilateral vestibular failure. Neuroimage Clin 24:101942
Horslen BC, Dakin CJ, Inglis JT, Blouin JS, Carpenter MG (2014) Modulation of human vestibular reflexes with increased postural threat. J Physiol Lond 592:3671–3685
Inukai Y, Otsuru N, Masaki M, Saito K, Miyaguchi S, Kojima S, Onishi H (2018) Effect of noisy galvanic vestibular stimulation on center of pressure sway of static standing posture. Brain Stimul 11:85–93
Ivanenko Y, Gurfinkel VS (2018) Human postural control. Front Neurosci 12:171
Iwasaki S, Karino S, Kamogashira T, Togo F, Fujimoto C, Yamamoto Y, Yamasoba T (2017) Effect of noisy galvanic vestibular stimulation on ocular vestibular-evoked myogenic potentials to bone-conducted vibration. Front Neurol 8:26
Iwasaki S, Yamamoto Y, Togo F, Kinoshita M, Yoshifuji Y, Fujimoto C, Yamasoba T (2014) Noisy vestibular stimulation improves body balance in bilateral vestibulopathy. Neurology 82:969–975
Jorns-Haderli M, Straumann D, Palla A (2007) Accuracy of the bedside head impulse test in detecting vestibular hypofunction. J Neurol Neurosurg Psychiatry 78:1113–1118
Keywan A, Badarna H, Jahn K, Wuehr M (2020) No evidence for after-effects of noisy galvanic vestibular stimulation on motion perception. Sci Rep 10:2545
Keywan A, Jahn K, Wuehr M (2019) Noisy galvanic vestibular stimulation primarily affects otolith-mediated motion perception. Neuroscience 399:161–166
Keywan A, Wuehr M, Pradhan C, Jahn K (2018) Noisy galvanic stimulation improves roll-tilt vestibular perception in healthy subjects. Front Neurol 9:83
Kwan A, Forbes PA, Mitchell DE, Blouin JS, Cullen KE (2019) Neural substrates, dynamics and thresholds of galvanic vestibular stimulation in the behaving primate. Nat Commun 10:1904
Lim SB, Cleworth TW, Horslen BC, Blouin JS, Inglis JT, Carpenter MG (2017) Postural threat influences vestibular-evoked muscular responses. J Neurophysiol 117:604–611
Lobel E, Kleine JF, Bihan DL, Leroy-Willig A, Berthoz A (1998) Functional MRI of galvanic vestibular stimulation. J Neurophysiol 80:2699–2709
Lund S, Broberg C (1983) Effects of different head positions on postural sway in man induced by a reproducible vestibular error signal. Acta Physiol Scand 117:307–309
MacDougall HG, Moore ST, Curthoys IS, Black FO (2006) Modeling postural instability with galvanic vestibular stimulation. Exp Brain Res 172:208–220
Machner B, Sprenger A, Fullgraf H, Trillenberg P, Helmchen C (2013) Video-based head impulse test. Importance for routine diagnostics of patients with vertigo. Der Nervenarzt 84:975–983
Massot C, Schneider AD, Chacron MJ, Cullen KE (2012) The vestibular system implements a linear-nonlinear transformation in order to encode self-motion. PLoS Biol 10:e1001365
Mian OS, Dakin CJ, Blouin JS, Fitzpatrick RC, Day BL (2010) Lack of otolith involvement in balance responses evoked by mastoid electrical stimulation. J Physiol 588:4441–4451
Moss F, Ward LM, Sannita WG (2004) Stochastic resonance and sensory information processing: a tutorial and review of application. Clin Neurophysiol 115:267–281
Nooristani M, Maheu M, Houde MS, Bacon BA, Champoux F (2019) Questioning the lasting effect of galvanic vestibular stimulation on postural control. PLoS ONE 14:e0224619
Potvin-Desrochers A, Richer N, Lajoie Y (2017) Cognitive tasks promote automatization of postural control in young and older adults. Gait Posture 57:40–45
Priesol AJ, Valko Y, Merfeld DM, Lewis RF (2014) Motion perception in patients with idiopathic bilateral vestibular hypofunction. Otolaryngol Head Neck Surg 150:1040–1042
Ramos Macias A, Ramos de Miguel A, Rodriguez Montesdeoca I, Borkoski Barreiro S, Falcon Gonzalez JC (2019) Chronic electrical stimulation of the otolith organ: preliminary results in humans with bilateral vestibulopathy and sensorineural hearing loss. Audiol Neurootol 25(1–2):79–90
Raymakers JA, Samson MM, Verhaar HJ (2005) The assessment of body sway and the choice of the stability parameter(s). Gait Posture 21:48–58
Richer N, Lajoie Y (2020) Automaticity of postural control while dual-tasking revealed in young and older adults. Exp Aging Res 46:1–21
Rinalduzzi S, Cipriani AM, Capozza M, Accornero N (2011) Postural responses to low-intensity, short-duration, galvanic vestibular stimulation as a possible differential diagnostic procedure. Acta Neurol Scand 123:111–116
Schniepp R, Boerner JC, Decker J, Jahn K, Brandt T, Wuehr M (2018) Noisy vestibular stimulation improves vestibulospinal function in patients with bilateral vestibulopathy. J Neurol 265:57–62
Sprenger A, Steinhaus S, Helmchen C (2017) Postural control during recall of vestibular sensation in patients with functional dizziness and unilateral vestibulopathy. J Neurol 264:42–44
Sprenger A, Wojak JF, Jandl NM, Helmchen C (2017) Postural control in bilateral vestibular failure: its relation to visual, proprioceptive, vestibular, and cognitive input. Front Neurol 8:444
Sprenger A, Wojak JF, Jandl NM, Hertel S, Helmchen C (2014) Predictive mechanisms improve the vestibulo-ocular reflex in patients with bilateral vestibular failure. J Neurol 261:628–631
St-Amant G, Rahman T, Polskaia N, Fraser S, Lajoie Y (2020) Unveilling the cerebral and sensory contributions to automatic postural control during dual-task standing. Hum Mov Sci 70:102587
Stephan T, Deutschlander A, Nolte A, Schneider E, Wiesmann M, Brandt T, Dieterich M (2005) Functional MRI of galvanic vestibular stimulation with alternating currents at different frequencies. Neuroimage 26:721–732
Strupp M, Kim JS, Murofushi T, Straumann D, Jen JC, Rosengren SM, Della Santina CC, Kingma H (2017) Bilateral vestibulopathy: diagnostic criteria consensus document of the classification committee of the Barany Society. J Vestib Res 27:177–189
Tax CM, Bom AP, Taylor RL, Todd N, Cho KK, Fitzpatrick RC, Welgampola MS (2013) The galvanic whole-body sway response in health and disease. Clin Neurophysiol 124:2036–2045
Tschan R, Wiltink J, Best C, Bense S, Dieterich M, Beutel ME, Eckhardt-Henn A (2008) Validation of the German version of the Vertigo Symptom Scale (VSS) in patients with organic or somatoform dizziness and healthy controls. J Neurol 255:1168–1175
Volkening K, Bergmann J, Keller I, Wuehr M, Muller F, Jahn K (2014) Verticality perception during and after galvanic vestibular stimulation. Neurosci Lett 581:75–79
Welgampola MS, Ramsay E, Gleeson MJ, Day BL (2013) Asymmetry of balance responses to monaural galvanic vestibular stimulation in subjects with vestibular schwannoma. Clin Neurophysiol 124:1835–1839
Woll J, Sprenger A, Helmchen C (2019) Postural control during galvanic vestibular stimulation in patients with persistent perceptual–postural dizziness. J Neurol 266:1236–1249
Wuehr M, Boerner JC, Pradhan C, Decker J, Jahn K, Brandt T, Schniepp R (2018) Stochastic resonance in the human vestibular system—noise-induced facilitation of vestibulospinal reflexes. Brain Stimul 11:261–263
Wuehr M, Decker J, Schniepp R (2017) Noisy galvanic vestibular stimulation: an emerging treatment option for bilateral vestibulopathy. J Neurol 264:81–86
Wuehr M, Nusser E, Krafczyk S, Straube A, Brandt T, Jahn K, Schniepp R (2016) Noise-enhanced vestibular input improves dynamic walking stability in healthy subjects. Brain Stimul 9:109–116
Yardley L, Masson E, Verschuur C, Haacke N, Luxon L (1992) Symptoms, anxiety and handicap in dizzy patients: development of the Vertigo Symptom Scale. J Psychosom Res 36:731–741
Zingler VC, Cnyrim C, Jahn K, Weintz E, Fernbacher J, Frenzel C, Brandt T, Strupp M (2007) Causative factors and epidemiology of bilateral vestibulopathy in 255 patients. Ann Neurol 61:524–532
Zingler VC, Weintz E, Jahn K, Mike A, Huppert D, Rettinger N, Brandt T, Strupp M (2008) Follow-up of vestibular function in bilateral vestibulopathy. J Neurol Neurosurg Psychiatry 79:284–288
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflicts of interest
None.
Ethical standard
The studies involving human participants were reviewed and approved by Ethics Committee of the Universität zu Lübeck (#17-036). The patients/participants provided their written informed consent to participate in this study.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Sprenger, A., Spliethoff, P., Rother, M. et al. Effects of perceptible and imperceptible galvanic vestibular stimulation on the postural control of patients with bilateral vestibulopathy. J Neurol 267, 2383–2397 (2020). https://doi.org/10.1007/s00415-020-09852-x
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00415-020-09852-x