Abstract
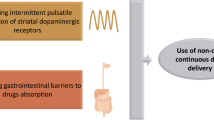
Motor fluctuations and dyskinesias occur in the majority of patients with Parkinson’s disease (PD) and are likely to result from changes in dopamine production, storage and release, occurring as consequences of the nigrostriatal degenerative process. All studies comparing levodopa versus dopamine agonist early therapy indicate that initiation with agonists is associated with a reduced risk of motor complications —in particular, dyskinesias— possibly because agonists’ longer half-lives provide continuous dopaminergic delivery. In advanced PD patients, switching from a pulsatile to continuous dopaminergic delivery may widen patients’ therapeutic window. Currently, this can be accomplished only with subcutaneous apomorphine or duodenal levodopa infusions. Apomorphine is a highly soluble agonist whose effect is similar to dopamine. Conversely, replacing whole oral therapy with levodopa infusion bypasses gastric emptying and avoids peaks and troughs in plasma by releasing levodopa in the duodenum/jejunum.
Similar content being viewed by others
References
Fabbrini G, Brotchie JM, Grandas F, Nomoto M, Goetz CG (2007) Levodopa-induced dyskinesias. Mov Disord 22:1379–1389
Schrag A, Quinn N (2000) Dyskinesias and motor fluctuations in Parkinson’s disease. A community-based study. Brain 123:2297–2305
Chapuis S, Ouchchane L, Metz O, Gerbaud L, Durif F (2005) Impact of the motor complications of Parkinson’s disease on the quality of life. Mov Disord 20:224–230
Antonini A, Barone P (2008) Dopamine agonist-based strategies in the treatment of Parkinson’s disease. Neurol Sci 29(Suppl 5):S371–S374
Hardie RJ, Lees AJ, Stern GM (1984) On-off fluctuations in Parkinson’s disease. A clinical and neuropharmacological study. Brain 107:487–506
Grandas F, Luquin MR, Rodriguez M, Vaamonde J, Lera G, Obeso JA (1992) Motor fluctuations in Parkinson disease: risk factors. Neurologia 7:89–93
Jenner P (2004) Avoidance of dyskinesia: preclinical evidence for continuous dopaminergic stimulation. Neurology 62(1 Suppl 1):S47–S55
Stock Exchange Release: Primary objective of STRIDE-PD study was not achieved. Orion Corporation (Finland) 2009
Antonini A, Tolosa E, Mizuno Y, Yamamoto M, Poewe W (2009) A reassessment of risks and benefits of dopamine agonists in Parkinson’s disease. Lancet Neurol 8:929–937
Rascol O, Brooks DJ, Korczyn AD, De Deyn PP, Clarke CE, Lang AE (2000) A five-year study of the incidence of dyskinesia in patients with early Parkinson’s disease who were treated with ropinirole or levodopa. 056 Study Group. N Engl J Med 342(20):1484–1491
Parkinson Study Group (2009) Long-term effect of initiating Pramipexole vs. Levodopa in early Parkinson disease. Arch Neurol 66(5):563–570
Hauser RA, Rascol O, Korczyn AD, Jon Stoessl A, Watts RL, Poewe W et al (2007) Ten-year follow-up of Parkinson’s disease patients randomized to initial therapy with ropinirole or levodopa. Mov Disord 22(16):2409–2417
Katzenschlager R, Head J, Schrag A, Ben-Shlomo Y, Evans A, Lees AJ (2008) Fourteen-year final report of the randomized PDRG-UK trial comparing three initial treatments in PD. Neurology 71(7):474–480
Poewe W, Kleedorfer B, Wagner M, Bosch S, Schelosky L (1993) Continuous subcutaneous apomorphine infusions for fluctuating Parkinson’s disease. Long-term follow-up in 18 patients. Adv Neurol 60:656–659
Poewe WH, Rascol O, Quinn N, Tolosa E, Oertel WH, Martignoni E et al (2007) Efficacy of pramipexole and transdermal rotigotine in advanced Parkinson’s disease: a double-blind, double-dummy, randomised controlled trial. Lancet Neurol 6(6):513–520
Katzenschlager R, Hughes A, Evans A, Manson AJ, Hoffman M, Swinn L et al (2005) Continuous subcutaneous apomorphine therapy improves dyskinesias in Parkinson’s disease: a prospective study using single-dose challenges. Mov Disord 20(2):151–157
Stocchi F, Ruggieri S, Vacca L, Olanow CW (2002) Prospective randomized trial of lisuride infusion versus oral levodopa in patients with Parkinson’s disease. Brain 125(Pt 9):2058–2066
Antonini A, Tolosa E (2009) Apomorphine and levodopa infusion therapies for advanced Parkinson’s disease: selection criteria and patient management. Expert Rev Neurother 9:859–867
Kurlan R, Rothfield KP, Woodward WR et al (1988) Erratic gastric emptying may cause “random” fluctuations of parkinsonian mobility. Neurology 38:419–421
Antonini A (2007) New strategies in motor parkinsonism. Parkinsonism Relat Disord 13(Suppl 3):S446–S449
Mouradian MM, Juncos J, Fabbrini G, Chase TN (1987) Motor fluctuations in Parkinson’s disease: pathogenetic and therapeutic studies. Ann Neurol 22:475–479
Kurlan R, Nutt JG, Woodward WR et al (1988) Duodenal and gastric delivery of levodopa in Parkinsonism. Ann Neurol 23:589–595
Sage JI, Sonsalla PK, McHale DM et al (1990) Clinical experience with duodenal infusions of levodopa for the treatment of motor fluctuations in Parkinson’s disease. Adv Neurol 53:383–386
Nyholm D, Nilsson Remahl AI, Dizdar N et al (2005) Duodenal levodopa infusion monotherapy vs. oral polypharmacy in advanced Parkinson disease. Neurology 64:216–223
Nyholm D (2006) Enteral levodopa/carbidopa gel infusion for the treatment of motor fluctuations and dyskinesias in advanced Parkinson’s disease. Expert Rev Neurother 6:1403–1411
Antonini A, Isaias IU, Canesi M, Zibetti M, Mancini F, Manfredi L et al (2007) Duodenal levodopa infusion for advanced Parkinson’s disease: 12-month treatment outcome. Mov Disord 22(8):1145–1149
Honig H, Antonini A, Martinez-Martin P et al (2009) Intrajejunal levodopa infusion in Parkinson’s disease: a pilot multicenter study of effects on nonmotor symptoms and quality of life. Mov Disord 24:1468–1474
Antonini A, Chaudhuri KR, Martinez-Martin P, Odin P (2010) Oral and infusion based strategies for managing motor complications in patients with Parkinson’s disease. CNS Drugs 24(2):119–129
Conflict of interest
Dr. Antonini has received honoraria from Solvay and Chiesi Pharmaceuticals.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Antonini, A., Ursino, G., Calandrella, D. et al. Continuous dopaminergic delivery in Parkinson’s disease. J Neurol 257 (Suppl 2), 305–308 (2010). https://doi.org/10.1007/s00415-010-5714-1
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00415-010-5714-1